+検索条件
-Structure paper
| タイトル | Filament formation drives catalysis by glutaminase enzymes important in cancer progression. |
|---|---|
| ジャーナル・号・ページ | Nat Commun, Vol. 15, Issue 1, Page 1971, Year 2024 |
| 掲載日 | 2024年3月4日 |
 著者 著者 | Shi Feng / Cody Aplin / Thuy-Tien T Nguyen / Shawn K Milano / Richard A Cerione /  |
| PubMed 要旨 | The glutaminase enzymes GAC and GLS2 catalyze the hydrolysis of glutamine to glutamate, satisfying the 'glutamine addiction' of cancer cells. They are the targets of anti-cancer drugs; however, their ...The glutaminase enzymes GAC and GLS2 catalyze the hydrolysis of glutamine to glutamate, satisfying the 'glutamine addiction' of cancer cells. They are the targets of anti-cancer drugs; however, their mechanisms of activation and catalytic activity have been unclear. Here we demonstrate that the ability of GAC and GLS2 to form filaments is directly coupled to their catalytic activity and present their cryo-EM structures which provide a view of the conformational states essential for catalysis. Filament formation guides an 'activation loop' to assume a specific conformation that works together with a 'lid' to close over the active site and position glutamine for nucleophilic attack by an essential serine. Our findings highlight how ankyrin repeats on GLS2 regulate enzymatic activity, while allosteric activators stabilize, and clinically relevant inhibitors block, filament formation that enables glutaminases to catalyze glutaminolysis and support cancer progression. |
 リンク リンク |  Nat Commun / Nat Commun /  PubMed:38438397 / PubMed:38438397 /  PubMed Central PubMed Central |
| 手法 | EM (らせん対称) / EM (単粒子) |
| 解像度 | 3.12 - 3.69 Å |
| 構造データ | EMDB-40918, PDB-8szj: EMDB-40920, PDB-8szl: EMDB-40950, PDB-8t0z:  EMDB-43533: Human liver-type glutaminase, bound with inhibitor Compound 968 |
| 化合物 |  ChemComp-PO4: 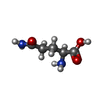 ChemComp-GLN: |
| 由来 |
|
 キーワード キーワード | HYDROLASE / Cancer / Filament / Metabolism / Metabolic |
 ムービー
ムービー コントローラー
コントローラー 構造ビューア
構造ビューア 万見文献について
万見文献について








 homo sapiens (ヒト)
homo sapiens (ヒト)