+検索条件
-Structure paper
| タイトル | Structural identification of a hotspot on CFTR for potentiation. |
|---|---|
| ジャーナル・号・ページ | Science, Vol. 364, Issue 6446, Page 1184-1188, Year 2019 |
| 掲載日 | 2019年6月21日 |
 著者 著者 | Fangyu Liu / Zhe Zhang / Anat Levit / Jesper Levring / Kouki K Touhara / Brian K Shoichet / Jue Chen /  |
| PubMed 要旨 | Cystic fibrosis is a fatal disease caused by mutations in the cystic fibrosis transmembrane conductance regulator (CFTR). Two main categories of drugs are being developed: correctors that improve ...Cystic fibrosis is a fatal disease caused by mutations in the cystic fibrosis transmembrane conductance regulator (CFTR). Two main categories of drugs are being developed: correctors that improve folding of CFTR and potentiators that recover the function of CFTR. Here, we report two cryo-electron microscopy structures of human CFTR in complex with potentiators: one with the U.S. Food and Drug Administration (FDA)-approved drug ivacaftor at 3.3-angstrom resolution and the other with an investigational drug, GLPG1837, at 3.2-angstrom resolution. These two drugs, although chemically dissimilar, bind to the same site within the transmembrane region. Mutagenesis suggests that in both cases, hydrogen bonds provided by the protein are important for drug recognition. The molecular details of how ivacaftor and GLPG1837 interact with CFTR may facilitate structure-based optimization of therapeutic compounds. |
 リンク リンク |  Science / Science /  PubMed:31221859 / PubMed:31221859 /  PubMed Central PubMed Central |
| 手法 | EM (単粒子) |
| 解像度 | 3.2 - 3.3 Å |
| 構造データ | |
| 化合物 | 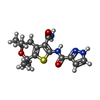 ChemComp-LJP:  ChemComp-MG:  ChemComp-ATP:  ChemComp-POV:  ChemComp-CLR:  ChemComp-AJP: 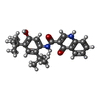 ChemComp-VX7: |
| 由来 |
|
 キーワード キーワード | HYDROLASE / ABC transporter / anion channel / cystic fibrosis / membrane protein / GLPG1837 / ivacaftor |
 ムービー
ムービー コントローラー
コントローラー 構造ビューア
構造ビューア 万見文献について
万見文献について







 homo sapiens (ヒト)
homo sapiens (ヒト)