+検索条件
-Structure paper
| タイトル | Molecular basis of Wnt biogenesis, secretion, and Wnt7-specific signaling. |
|---|---|
| ジャーナル・号・ページ | Cell, Vol. 186, Issue 23, Page 5028-5040.e14, Year 2023 |
| 掲載日 | 2023年11月9日 |
 著者 著者 | Xiaofeng Qi / Qinli Hu / Nadia Elghobashi-Meinhardt / Tao Long / Hongwen Chen / Xiaochun Li /   |
| PubMed 要旨 | Wnt proteins are enzymatically lipidated by Porcupine (PORCN) in the ER and bind to Wntless (WLS) for intracellular transport and secretion. Mechanisms governing the transfer of these low-solubility ...Wnt proteins are enzymatically lipidated by Porcupine (PORCN) in the ER and bind to Wntless (WLS) for intracellular transport and secretion. Mechanisms governing the transfer of these low-solubility Wnts from the ER to the extracellular space remain unclear. Through structural and functional analyses of Wnt7a, a crucial Wnt involved in central nervous system angiogenesis and blood-brain barrier maintenance, we have elucidated the principles of Wnt biogenesis and Wnt7-specific signaling. The Wnt7a-WLS complex binds to calreticulin (CALR), revealing that CALR functions as a chaperone to facilitate Wnt transfer from PORCN to WLS during Wnt biogenesis. Our structures, functional analyses, and molecular dynamics simulations demonstrate that a phospholipid in the core of Wnt-bound WLS regulates the association and dissociation between Wnt and WLS, suggesting a lipid-mediated Wnt secretion mechanism. Finally, the structure of Wnt7a bound to RECK, a cell-surface Wnt7 co-receptor, reveals how RECK engages the N-terminal domain of Wnt7a to activate Wnt7-specific signaling. |
 リンク リンク |  Cell / Cell /  PubMed:37852257 / PubMed:37852257 /  PubMed Central PubMed Central |
| 手法 | EM (単粒子) |
| 解像度 | 3.1 - 3.84 Å |
| 構造データ | EMDB-41764, PDB-8tzo: EMDB-41765, PDB-8tzp: EMDB-41768, PDB-8tzs: |
| 化合物 | 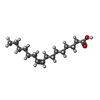 ChemComp-PAM:  ChemComp-POV:  ChemComp-CA: |
| 由来 |
|
 キーワード キーワード | SIGNALING PROTEIN / MEMBRANE PROTEIN |
 ムービー
ムービー コントローラー
コントローラー 構造ビューア
構造ビューア 万見文献について
万見文献について








 homo sapiens (ヒト)
homo sapiens (ヒト)