+検索条件
-Structure paper
| タイトル | Structural and functional studies of LRP6 ectodomain reveal a platform for Wnt signaling. |
|---|---|
| ジャーナル・号・ページ | Dev Cell, Vol. 21, Issue 5, Page 848-861, Year 2011 |
| 掲載日 | 2011年11月15日 |
 著者 著者 | Shuo Chen / Doryen Bubeck / Bryan T MacDonald / Wen-Xue Liang / Jian-Hua Mao / Tomas Malinauskas / Oscar Llorca / A Radu Aricescu / Christian Siebold / Xi He / E Yvonne Jones /  |
| PubMed 要旨 | LDL-receptor-related protein 6 (LRP6), alongside Frizzled receptors, transduces Wnt signaling across the plasma membrane. The LRP6 ectodomain comprises four tandem β-propeller-EGF-like domain (PE) ...LDL-receptor-related protein 6 (LRP6), alongside Frizzled receptors, transduces Wnt signaling across the plasma membrane. The LRP6 ectodomain comprises four tandem β-propeller-EGF-like domain (PE) pairs that harbor binding sites for Wnt morphogens and their antagonists including Dickkopf 1 (Dkk1). To understand how these multiple interactions are integrated, we combined crystallographic analysis of the third and fourth PE pairs with electron microscopy (EM) to determine the complete ectodomain structure. An extensive inter-pair interface, conserved for the first-to-second and third-to-fourth PE interactions, contributes to a compact platform-like architecture, which is disrupted by mutations implicated in developmental diseases. EM reconstruction of the LRP6 platform bound to chaperone Mesd exemplifies a binding mode spanning PE pairs. Cellular and binding assays identify overlapping Wnt3a- and Dkk1-binding surfaces on the third PE pair, consistent with steric competition, but also suggest a model in which the platform structure supports an interplay of ligands through multiple interaction sites. |
 リンク リンク |  Dev Cell / Dev Cell /  PubMed:22000855 / PubMed:22000855 /  PubMed Central PubMed Central |
| 手法 | EM (単粒子) / X線回折 |
| 解像度 | 1.9 - 26.0 Å |
| 構造データ | 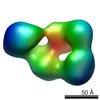 EMDB-1964: 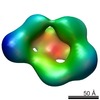 EMDB-1965:  PDB-4a0p: |
| 化合物 |  ChemComp-NAG:  ChemComp-CA:  ChemComp-CL:  ChemComp-EDO:  ChemComp-HOH: |
| 由来 |
|
 キーワード キーワード |  SIGNALING / LRP6 / SIGNALING / LRP6 /  WNT SIGNALLING (Wntシグナル経路) / WNT3A / DKK1 / WNT SIGNALLING (Wntシグナル経路) / WNT3A / DKK1 /  MESD MESD |
 ムービー
ムービー コントローラー
コントローラー 構造ビューア
構造ビューア 万見文献について
万見文献について




