+検索条件
-Structure paper
| タイトル | TRPV1 structures in nanodiscs reveal mechanisms of ligand and lipid action. |
|---|---|
| ジャーナル・号・ページ | Nature, Vol. 534, Issue 7607, Page 347-351, Year 2016 |
| 掲載日 | 2016年6月16日 |
 著者 著者 | Yuan Gao / Erhu Cao / David Julius / Yifan Cheng /  |
| PubMed 要旨 | When integral membrane proteins are visualized in detergents or other artificial systems, an important layer of information is lost regarding lipid interactions and their effects on protein structure. ...When integral membrane proteins are visualized in detergents or other artificial systems, an important layer of information is lost regarding lipid interactions and their effects on protein structure. This is especially relevant to proteins for which lipids have both structural and regulatory roles. Here we demonstrate the power of combining electron cryo-microscopy with lipid nanodisc technology to ascertain the structure of the rat TRPV1 ion channel in a native bilayer environment. Using this approach, we determined the locations of annular and regulatory lipids and showed that specific phospholipid interactions enhance binding of a spider toxin to TRPV1 through formation of a tripartite complex. Furthermore, phosphatidylinositol lipids occupy the binding site for capsaicin and other vanilloid ligands, suggesting a mechanism whereby chemical or thermal stimuli elicit channel activation by promoting the release of bioactive lipids from a critical allosteric regulatory site. |
 リンク リンク |  Nature / Nature /  PubMed:27281200 / PubMed:27281200 /  PubMed Central PubMed Central |
| 手法 | EM (単粒子) |
| 解像度 | 2.95 - 3.8 Å |
| 構造データ | EMDB-8117, PDB-5irx: EMDB-8119, PDB-5is0:  EMDB-8120: |
| 化合物 | 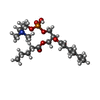 ChemComp-6O8:  ChemComp-6OE: 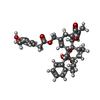 ChemComp-6EU: 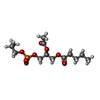 ChemComp-6O9: 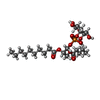 ChemComp-6ES:  ChemComp-6ET: |
| 由来 |
|
 キーワード キーワード | TRANSPORT PROTEIN / TRP / ion channel / nanodisc / vanilloid / lipid / interaction |
 ムービー
ムービー コントローラー
コントローラー 構造ビューア
構造ビューア 万見文献について
万見文献について










 haplopelma schmidti (クモ)
haplopelma schmidti (クモ)