[English] 日本語
 Yorodumi
Yorodumi- EMDB-2236: Negative Staining Structure of Human Polycomb Repressive Complex ... -
+ Open data
Open data
- Basic information
Basic information
| Entry | Database: EMDB / ID: EMD-2236 | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Title | Negative Staining Structure of Human Polycomb Repressive Complex 2 bound to the co-factor AEBP2 | |||||||||
 Map data Map data | Negative Staining Reconstruction of the Polycomb Repressive Complex 2 bound to the co-factor AEBP2 | |||||||||
 Sample Sample |
| |||||||||
 Keywords Keywords | Polycomb / PRC2 / epigenetic / gene silencing / nucleosome / labeling / chemical cross-linking / chromatin | |||||||||
| Function / homology | ESC/E(Z) complex / SET domain Function and homology information Function and homology information | |||||||||
| Biological species |  Homo sapiens (human) Homo sapiens (human) | |||||||||
| Method | single particle reconstruction / negative staining / Resolution: 21.0 Å | |||||||||
 Authors Authors | Ciferri C / Lander GC / Maiolica A / Herzog F / Aebersold R / Nogales E | |||||||||
 Citation Citation |  Journal: Elife / Year: 2012 Journal: Elife / Year: 2012Title: Molecular architecture of human polycomb repressive complex 2. Authors: Claudio Ciferri / Gabriel C Lander / Alessio Maiolica / Franz Herzog / Ruedi Aebersold / Eva Nogales /  Abstract: Polycomb Repressive Complex 2 (PRC2) is essential for gene silencing, establishing transcriptional repression of specific genes by tri-methylating Lysine 27 of histone H3, a process mediated by ...Polycomb Repressive Complex 2 (PRC2) is essential for gene silencing, establishing transcriptional repression of specific genes by tri-methylating Lysine 27 of histone H3, a process mediated by cofactors such as AEBP2. In spite of its biological importance, little is known about PRC2 architecture and subunit organization. Here, we present the first three-dimensional electron microscopy structure of the human PRC2 complex bound to its cofactor AEBP2. Using a novel internal protein tagging-method, in combination with isotopic chemical cross-linking and mass spectrometry, we have localized all the PRC2 subunits and their functional domains and generated a detailed map of interactions. The position and stabilization effect of AEBP2 suggests an allosteric role of this cofactor in regulating gene silencing. Regions in PRC2 that interact with modified histone tails are localized near the methyltransferase site, suggesting a molecular mechanism for the chromatin-based regulation of PRC2 activity.DOI:http://dx.doi.org/10.7554/eLife.00005.001. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Movie |
 Movie viewer Movie viewer |
|---|---|
| Structure viewer | EM map:  SurfView SurfView Molmil Molmil Jmol/JSmol Jmol/JSmol |
| Supplemental images |
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_2236.map.gz emd_2236.map.gz | 3.5 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-2236-v30.xml emd-2236-v30.xml emd-2236.xml emd-2236.xml | 14.7 KB 14.7 KB | Display Display |  EMDB header EMDB header |
| Images |  emd_2236.jpg emd_2236.jpg | 482.1 KB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-2236 http://ftp.pdbj.org/pub/emdb/structures/EMD-2236 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-2236 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-2236 | HTTPS FTP |
-Validation report
| Summary document |  emd_2236_validation.pdf.gz emd_2236_validation.pdf.gz | 186.2 KB | Display |  EMDB validaton report EMDB validaton report |
|---|---|---|---|---|
| Full document |  emd_2236_full_validation.pdf.gz emd_2236_full_validation.pdf.gz | 185.3 KB | Display | |
| Data in XML |  emd_2236_validation.xml.gz emd_2236_validation.xml.gz | 5.4 KB | Display | |
| Arichive directory |  https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-2236 https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-2236 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-2236 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-2236 | HTTPS FTP |
-Related structure data
| Similar structure data |
|---|
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|
- Map
Map
| File |  Download / File: emd_2236.map.gz / Format: CCP4 / Size: 5.2 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_2236.map.gz / Format: CCP4 / Size: 5.2 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Negative Staining Reconstruction of the Polycomb Repressive Complex 2 bound to the co-factor AEBP2 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Projections & slices | Image control
Images are generated by Spider. | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 3.14 Å | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
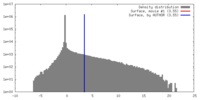
| Density |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
CCP4 map header:
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
-Supplemental data
- Sample components
Sample components
-Entire : Negative Staining Reconstruction of Polycomb repressive Complex 2...
| Entire | Name: Negative Staining Reconstruction of Polycomb repressive Complex 2 bound to the co-factor AEBP2 |
|---|---|
| Components |
|
-Supramolecule #1000: Negative Staining Reconstruction of Polycomb repressive Complex 2...
| Supramolecule | Name: Negative Staining Reconstruction of Polycomb repressive Complex 2 bound to the co-factor AEBP2 type: sample / ID: 1000 / Oligomeric state: Pentameric Complex / Number unique components: 5 |
|---|---|
| Molecular weight | Theoretical: 265 MDa |
-Macromolecule #1: RbAp48
| Macromolecule | Name: RbAp48 / type: protein_or_peptide / ID: 1 / Number of copies: 1 / Oligomeric state: monomer / Recombinant expression: Yes |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) / synonym: Human / Location in cell: Nucleus Homo sapiens (human) / synonym: Human / Location in cell: Nucleus |
| Recombinant expression | Organism:  |
| Sequence | GO: ESC/E(Z) complex / InterPro: SET domain |
-Macromolecule #2: Ezh2
| Macromolecule | Name: Ezh2 / type: protein_or_peptide / ID: 2 / Number of copies: 1 / Oligomeric state: monomer / Recombinant expression: Yes |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) / synonym: Human / Location in cell: Nucleus Homo sapiens (human) / synonym: Human / Location in cell: Nucleus |
| Recombinant expression | Organism:  |
| Sequence | GO: ESC/E(Z) complex / InterPro: SET domain |
-Macromolecule #3: Suz12
| Macromolecule | Name: Suz12 / type: protein_or_peptide / ID: 3 / Number of copies: 1 / Oligomeric state: monomer / Recombinant expression: Yes |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) / synonym: Human / Location in cell: Nucleus Homo sapiens (human) / synonym: Human / Location in cell: Nucleus |
| Recombinant expression | Organism:  |
| Sequence | GO: ESC/E(Z) complex / InterPro: SET domain |
-Macromolecule #4: AEBP2
| Macromolecule | Name: AEBP2 / type: protein_or_peptide / ID: 4 / Number of copies: 1 / Oligomeric state: monomer / Recombinant expression: Yes |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) / synonym: Human / Location in cell: Nucleus Homo sapiens (human) / synonym: Human / Location in cell: Nucleus |
| Recombinant expression | Organism:  |
| Sequence | GO: ESC/E(Z) complex / InterPro: SET domain |
-Macromolecule #5: EED
| Macromolecule | Name: EED / type: protein_or_peptide / ID: 5 / Number of copies: 1 / Oligomeric state: monomer / Recombinant expression: Yes |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) / synonym: Human / Location in cell: Nucleus Homo sapiens (human) / synonym: Human / Location in cell: Nucleus |
| Recombinant expression | Organism:  |
| Sequence | GO: ESC/E(Z) complex / InterPro: SET domain |
-Experimental details
-Structure determination
| Method | negative staining |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Concentration | 0.1 mg/mL |
|---|---|
| Buffer | pH: 7.6 Details: 25 mM Hepes pH 7.5, 138 mM NaCl, 10% Glycerol, 0.05% NP40, 1 mM Tris(2-carboxyethyl) phosphine hydrochloride [TCEP] |
| Staining | Type: NEGATIVE / Details: 2% Uranyl Formate |
| Grid | Details: 400 mesh copper carbon support |
| Vitrification | Cryogen name: NONE / Instrument: OTHER |
- Electron microscopy
Electron microscopy
| Microscope | FEI TECNAI 20 |
|---|---|
| Temperature | Average: 298 K |
| Alignment procedure | Legacy - Astigmatism: Objective lens astigmatism was corrected at 150,000 times magnification |
| Specialist optics | Energy filter - Name: FEI |
| Date | Apr 11, 2011 |
| Image recording | Category: CCD / Film or detector model: GENERIC GATAN (4k x 4k) / Number real images: 500 / Average electron dose: 20 e/Å2 |
| Tilt angle min | 0 |
| Tilt angle max | 0 |
| Electron beam | Acceleration voltage: 120 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | Calibrated magnification: 80000 / Illumination mode: FLOOD BEAM / Imaging mode: DARK FIELD / Cs: 2.2 mm / Nominal defocus max: 1.3 µm / Nominal defocus min: 0.6 µm / Nominal magnification: 80000 |
| Sample stage | Specimen holder model: OTHER |
- Image processing
Image processing
| Details | Material and Methods section of Paper |
|---|---|
| CTF correction | Details: whole micrograph |
| Final reconstruction | Applied symmetry - Point group: C1 (asymmetric) / Algorithm: OTHER / Resolution.type: BY AUTHOR / Resolution: 21.0 Å / Resolution method: FSC 0.5 CUT-OFF Software - Name: Appion, Leginon, Spider, Imagic, EMAN2, SPARX Number images used: 39527 |
| Final two d classification | Number classes: 1000 |
-Atomic model buiding 1
| Initial model | PDB ID: |
|---|---|
| Software | Name:  Chimera Chimera |
| Details | Protocol: local rigid-body fitting algorithm |
| Refinement | Space: REAL / Protocol: RIGID BODY FIT |
-Atomic model buiding 2
| Initial model | PDB ID: |
|---|---|
| Software | Name:  Chimera Chimera |
| Details | Protocol: local rigid-body fitting algorithm |
| Refinement | Space: REAL / Protocol: RIGID BODY FIT |
 Movie
Movie Controller
Controller











 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)