[English] 日本語
 Yorodumi
Yorodumi- EMDB-14804: Structure of an endogenous human TREX complex bound to mRNA, comp... -
+ Open data
Open data
- Basic information
Basic information
| Entry |  | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Title | Structure of an endogenous human TREX complex bound to mRNA, composite map | |||||||||
 Map data Map data | TREX-mRNA composite map generated from sharpened focused maps A (EMD 14805),B (EMD-14806) and C (EMD-14807 ) | |||||||||
 Sample Sample |
| |||||||||
 Keywords Keywords | transcription and export complex / TREX / RNA export / RNA packaging / RNA binding protein / gene expression / GENE REGULATION | |||||||||
| Function / homology |  Function and homology information Function and homology informationTHO complex / THO complex part of transcription export complex / primitive hemopoiesis / transcription export complex / C5-methylcytidine-containing RNA reader activity / regulation of mRNA export from nucleus / U6 snRNP / Transport of the SLBP independent Mature mRNA / Transport of the SLBP Dependant Mature mRNA / ATP-dependent activity, acting on RNA ...THO complex / THO complex part of transcription export complex / primitive hemopoiesis / transcription export complex / C5-methylcytidine-containing RNA reader activity / regulation of mRNA export from nucleus / U6 snRNP / Transport of the SLBP independent Mature mRNA / Transport of the SLBP Dependant Mature mRNA / ATP-dependent activity, acting on RNA / mRNA 3'-end processing / Transport of Mature mRNA Derived from an Intronless Transcript / ATP-dependent protein binding / Transport of Mature mRNA derived from an Intron-Containing Transcript / RNA export from nucleus / U4 snRNA binding / RNA Polymerase II Transcription Termination / U4 snRNP / stem cell division / poly(A)+ mRNA export from nucleus / generation of neurons / spliceosomal complex assembly / monocyte differentiation / blastocyst development / U6 snRNA binding / neuron development / RHOBTB2 GTPase cycle / mRNA export from nucleus / catalytic step 2 spliceosome / mRNA Splicing - Major Pathway / RNA splicing / central nervous system development / spliceosomal complex / mRNA splicing, via spliceosome / cell morphogenesis / mRNA processing / nuclear matrix / Signaling by CSF1 (M-CSF) in myeloid cells / osteoblast differentiation / negative regulation of neuron projection development / regulation of gene expression / RNA helicase activity / nuclear speck / nuclear body / RNA helicase / mRNA binding / apoptotic process / signal transduction / ATP hydrolysis activity / DNA binding / RNA binding / extracellular exosome / nucleoplasm / ATP binding / membrane / identical protein binding / nucleus / cytoplasm / cytosol Similarity search - Function | |||||||||
| Biological species |  Homo sapiens (human) Homo sapiens (human) | |||||||||
| Method | single particle reconstruction / cryo EM / Resolution: 3.9 Å | |||||||||
 Authors Authors | Pacheco-Fiallos FB / Vorlaender MK / Plaschka C | |||||||||
| Funding support | European Union, 2 items
| |||||||||
 Citation Citation |  Journal: Nature / Year: 2023 Journal: Nature / Year: 2023Title: mRNA recognition and packaging by the human transcription-export complex. Authors: Belén Pacheco-Fiallos / Matthias K Vorländer / Daria Riabov-Bassat / Laura Fin / Francis J O'Reilly / Farja I Ayala / Ulla Schellhaas / Juri Rappsilber / Clemens Plaschka /    Abstract: Newly made mRNAs are processed and packaged into mature ribonucleoprotein complexes (mRNPs) and are recognized by the essential transcription-export complex (TREX) for nuclear export. However, the ...Newly made mRNAs are processed and packaged into mature ribonucleoprotein complexes (mRNPs) and are recognized by the essential transcription-export complex (TREX) for nuclear export. However, the mechanisms of mRNP recognition and three-dimensional mRNP organization are poorly understood. Here we report cryo-electron microscopy and tomography structures of reconstituted and endogenous human mRNPs bound to the 2-MDa TREX complex. We show that mRNPs are recognized through multivalent interactions between the TREX subunit ALYREF and mRNP-bound exon junction complexes. Exon junction complexes can multimerize through ALYREF, which suggests a mechanism for mRNP organization. Endogenous mRNPs form compact globules that are coated by multiple TREX complexes. These results reveal how TREX may simultaneously recognize, compact and protect mRNAs to promote their packaging for nuclear export. The organization of mRNP globules provides a framework to understand how mRNP architecture facilitates mRNA biogenesis and export. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Supplemental images |
|---|
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_14804.map.gz emd_14804.map.gz | 8.2 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-14804-v30.xml emd-14804-v30.xml emd-14804.xml emd-14804.xml | 24.4 KB 24.4 KB | Display Display |  EMDB header EMDB header |
| Images |  emd_14804.png emd_14804.png | 118.2 KB | ||
| Filedesc metadata |  emd-14804.cif.gz emd-14804.cif.gz | 8.5 KB | ||
| Others |  emd_14804_additional_1.map.gz emd_14804_additional_1.map.gz | 8.3 MB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-14804 http://ftp.pdbj.org/pub/emdb/structures/EMD-14804 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-14804 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-14804 | HTTPS FTP |
-Related structure data
| Related structure data |  7znkMC  7znjC  7znlC C: citing same article ( M: atomic model generated by this map |
|---|---|
| Similar structure data | Similarity search - Function & homology  F&H Search F&H Search |
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|---|
| Related items in Molecule of the Month |
- Map
Map
| File |  Download / File: emd_14804.map.gz / Format: CCP4 / Size: 274.6 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_14804.map.gz / Format: CCP4 / Size: 274.6 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | TREX-mRNA composite map generated from sharpened focused maps A (EMD 14805),B (EMD-14806) and C (EMD-14807 ) | ||||||||||||||||||||||||||||||||||||
| Projections & slices | Image control
Images are generated by Spider. | ||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 1.41 Å | ||||||||||||||||||||||||||||||||||||
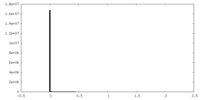
| Density |
| ||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
|
-Supplemental data
-Additional map: TREX-mRNA composite map generated from unsharpened focused maps...
| File | emd_14804_additional_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | TREX-mRNA composite map generated from unsharpened focused maps A (EMD 14805),B (EMD-14806) and C (EMD-14807 | ||||||||||||
| Projections & Slices |
| ||||||||||||
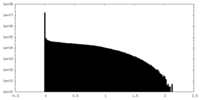
| Density Histograms |
- Sample components
Sample components
+Entire : endogenous human TREX-mRNA complex
+Supramolecule #1: endogenous human TREX-mRNA complex
+Macromolecule #1: RNA
+Macromolecule #2: THO complex subunit 1
+Macromolecule #3: THO complex subunit 2
+Macromolecule #4: THO complex subunit 3
+Macromolecule #5: THO complex subunit 5 homolog
+Macromolecule #6: THO complex subunit 6 homolog
+Macromolecule #7: THO complex subunit 7 homolog
+Macromolecule #8: Spliceosome RNA helicase DDX39B
+Macromolecule #9: THO complex subunit 4
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Concentration | 0.2 mg/mL |
|---|---|
| Buffer | pH: 7.9 |
| Vitrification | Cryogen name: ETHANE |
- Electron microscopy
Electron microscopy
| Microscope | FEI TITAN KRIOS |
|---|---|
| Image recording | Film or detector model: GATAN K3 (6k x 4k) / Average electron dose: 60.0 e/Å2 |
| Electron beam | Acceleration voltage: 300 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD / Nominal defocus max: 2.0 µm / Nominal defocus min: 0.5 µm |
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
- Image processing
Image processing
| Startup model | Type of model: EMDB MAP EMDB ID: |
|---|---|
| Final reconstruction | Resolution.type: BY AUTHOR / Resolution: 3.9 Å / Resolution method: FSC 0.143 CUT-OFF / Number images used: 182534 |
| Initial angle assignment | Type: MAXIMUM LIKELIHOOD |
| Final angle assignment | Type: MAXIMUM LIKELIHOOD |
 Movie
Movie Controller
Controller





















 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)