+ Open data
Open data
- Basic information
Basic information
| Entry |  | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Title | Symmetry-free cryo-EM map of TRiC-actin | |||||||||
 Map data Map data | ||||||||||
 Sample Sample |
| |||||||||
| Biological species |  | |||||||||
| Method | single particle reconstruction / cryo EM / Resolution: 9.1 Å | |||||||||
 Authors Authors | Milicic G / Balchin D / Hayer-Hartl M / Hartl FU | |||||||||
 Citation Citation |  Journal: Cell / Year: 2018 Journal: Cell / Year: 2018Title: Pathway of Actin Folding Directed by the Eukaryotic Chaperonin TRiC. Authors: David Balchin / Goran Miličić / Mike Strauss / Manajit Hayer-Hartl / F Ulrich Hartl /  Abstract: The hetero-oligomeric chaperonin of eukarya, TRiC, is required to fold the cytoskeletal protein actin. The simpler bacterial chaperonin system, GroEL/GroES, is unable to mediate actin folding. Here, ...The hetero-oligomeric chaperonin of eukarya, TRiC, is required to fold the cytoskeletal protein actin. The simpler bacterial chaperonin system, GroEL/GroES, is unable to mediate actin folding. Here, we use spectroscopic and structural techniques to determine how TRiC promotes the conformational progression of actin to the native state. We find that actin fails to fold spontaneously even in the absence of aggregation but populates a kinetically trapped, conformationally dynamic state. Binding of this frustrated intermediate to TRiC specifies an extended topology of actin with native-like secondary structure. In contrast, GroEL stabilizes bound actin in an unfolded state. ATP binding to TRiC effects an asymmetric conformational change in the chaperonin ring. This step induces the partial release of actin, priming it for folding upon complete release into the chaperonin cavity, mediated by ATP hydrolysis. Our results reveal how the unique features of TRiC direct the folding pathway of an obligate eukaryotic substrate. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Structure viewer | EM map:  SurfView SurfView Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|---|
| Supplemental images |
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_0017.map.gz emd_0017.map.gz | 5 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-0017-v30.xml emd-0017-v30.xml emd-0017.xml emd-0017.xml | 7.1 KB 7.1 KB | Display Display |  EMDB header EMDB header |
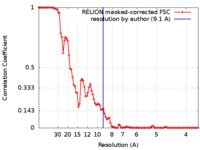
| FSC (resolution estimation) |  emd_0017_fsc.xml emd_0017_fsc.xml | 9.2 KB | Display |  FSC data file FSC data file |
| Images |  emd_0017.png emd_0017.png | 19.4 KB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-0017 http://ftp.pdbj.org/pub/emdb/structures/EMD-0017 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-0017 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-0017 | HTTPS FTP |
-Validation report
| Summary document |  emd_0017_validation.pdf.gz emd_0017_validation.pdf.gz | 235.2 KB | Display |  EMDB validaton report EMDB validaton report |
|---|---|---|---|---|
| Full document |  emd_0017_full_validation.pdf.gz emd_0017_full_validation.pdf.gz | 234.3 KB | Display | |
| Data in XML |  emd_0017_validation.xml.gz emd_0017_validation.xml.gz | 10.7 KB | Display | |
| Arichive directory |  https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-0017 https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-0017 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-0017 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-0017 | HTTPS FTP |
-Related structure data
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|
- Map
Map
| File |  Download / File: emd_0017.map.gz / Format: CCP4 / Size: 75.1 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_0017.map.gz / Format: CCP4 / Size: 75.1 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & slices | Image control
Images are generated by Spider. | ||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 1.85 Å | ||||||||||||||||||||||||||||||||||||
| Density |
| ||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
|
-Supplemental data
- Sample components
Sample components
-Entire : Bovine TRiC in complex with actin
| Entire | Name: Bovine TRiC in complex with actin |
|---|---|
| Components |
|
-Supramolecule #1: Bovine TRiC in complex with actin
| Supramolecule | Name: Bovine TRiC in complex with actin / type: complex / ID: 1 / Parent: 0 |
|---|---|
| Source (natural) | Organism:  |
| Molecular weight | Theoretical: 1.04 MDa |
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Buffer | pH: 7.4 |
|---|---|
| Vitrification | Cryogen name: ETHANE |
- Electron microscopy
Electron microscopy
| Microscope | FEI TITAN |
|---|---|
| Image recording | Film or detector model: FEI FALCON II (4k x 4k) / Average electron dose: 18.0 e/Å2 |
| Electron beam | Acceleration voltage: 300 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD |
 Movie
Movie Controller
Controller
















 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)