[English] 日本語
 Yorodumi
Yorodumi- PDB-6opw: HIV-1 Protease NL4-3 I13V, G16E, V32I, L33F, K45I, M46I, A71V, V8... -
+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 6opw | ||||||
|---|---|---|---|---|---|---|---|
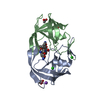
| Title | HIV-1 Protease NL4-3 I13V, G16E, V32I, L33F, K45I, M46I, A71V, V82F, I84V Mutant in complex with darunavir | ||||||
 Components Components | Protease NL4-3 | ||||||
 Keywords Keywords | HYDROLASE/HYDROLASE INHIBITOR / HIV / NL4-3 PROTEASE / DRUG RESISTANCE / PROTEASE INHIBITOR / HYDROLASE INHIBITOR COMPLEX / HYDROLASE / HYDROLASE-HYDROLASE INHIBITOR complex | ||||||
| Function / homology |  Function and homology information Function and homology informationHIV-1 retropepsin / symbiont-mediated activation of host apoptosis / retroviral ribonuclease H / exoribonuclease H / exoribonuclease H activity / host multivesicular body / DNA integration / viral genome integration into host DNA / RNA-directed DNA polymerase / establishment of integrated proviral latency ...HIV-1 retropepsin / symbiont-mediated activation of host apoptosis / retroviral ribonuclease H / exoribonuclease H / exoribonuclease H activity / host multivesicular body / DNA integration / viral genome integration into host DNA / RNA-directed DNA polymerase / establishment of integrated proviral latency / viral penetration into host nucleus / RNA stem-loop binding / symbiont-mediated suppression of host gene expression / RNA-directed DNA polymerase activity / host cell / RNA-DNA hybrid ribonuclease activity / Transferases; Transferring phosphorus-containing groups; Nucleotidyltransferases / viral nucleocapsid / aspartic-type endopeptidase activity / DNA recombination / DNA-directed DNA polymerase / Hydrolases; Acting on ester bonds / DNA-directed DNA polymerase activity / symbiont entry into host cell / lipid binding / host cell nucleus / host cell plasma membrane / structural molecule activity / virion membrane / proteolysis / DNA binding / zinc ion binding / membrane Similarity search - Function | ||||||
| Biological species |   Human immunodeficiency virus 1 Human immunodeficiency virus 1 | ||||||
| Method |  X-RAY DIFFRACTION / X-RAY DIFFRACTION /  MOLECULAR REPLACEMENT / Resolution: 2.1 Å MOLECULAR REPLACEMENT / Resolution: 2.1 Å | ||||||
 Authors Authors | Lockbaum, G.J. / Henes, M. / Kosovrasti, K. / Leidner, F. / Nachum, G.S. / Nalivaika, E.A. / Bolon, D.N.A. / KurtYilmaz, N. / Schiffer, C.A. | ||||||
| Funding support |  United States, 1items United States, 1items
| ||||||
 Citation Citation |  Journal: Acs Chem.Biol. / Year: 2019 Journal: Acs Chem.Biol. / Year: 2019Title: Picomolar to Micromolar: Elucidating the Role of Distal Mutations in HIV-1 Protease in Conferring Drug Resistance. Authors: Henes, M. / Lockbaum, G.J. / Kosovrasti, K. / Leidner, F. / Nachum, G.S. / Nalivaika, E.A. / Lee, S.K. / Spielvogel, E. / Zhou, S. / Swanstrom, R. / Bolon, D.N.A. / Kurt Yilmaz, N. / Schiffer, C.A. | ||||||
| History |
|
- Structure visualization
Structure visualization
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  6opw.cif.gz 6opw.cif.gz | 89.5 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb6opw.ent.gz pdb6opw.ent.gz | 68.5 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  6opw.json.gz 6opw.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Summary document |  6opw_validation.pdf.gz 6opw_validation.pdf.gz | 793.1 KB | Display |  wwPDB validaton report wwPDB validaton report |
|---|---|---|---|---|
| Full document |  6opw_full_validation.pdf.gz 6opw_full_validation.pdf.gz | 796.9 KB | Display | |
| Data in XML |  6opw_validation.xml.gz 6opw_validation.xml.gz | 12.1 KB | Display | |
| Data in CIF |  6opw_validation.cif.gz 6opw_validation.cif.gz | 16.1 KB | Display | |
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/op/6opw https://data.pdbj.org/pub/pdb/validation_reports/op/6opw ftp://data.pdbj.org/pub/pdb/validation_reports/op/6opw ftp://data.pdbj.org/pub/pdb/validation_reports/op/6opw | HTTPS FTP |
-Related structure data
| Related structure data |  6opsC  6optC  6opuC  6opvC  6opxC  6opyC  6opzC C: citing same article ( |
|---|---|
| Similar structure data |
- Links
Links
- Assembly
Assembly
| Deposited unit | 
| ||||||||
|---|---|---|---|---|---|---|---|---|---|
| 1 |
| ||||||||
| Unit cell |
|
- Components
Components
| #1: Protein | Mass: 10965.921 Da / Num. of mol.: 2 Mutation: I13V, G16E, V32I, L33F, K45I, M46I, A71V, V82F, I84V Source method: isolated from a genetically manipulated source Source: (gene. exp.)   Human immunodeficiency virus 1 / Gene: pol / Plasmid: pXC35 / Production host: Human immunodeficiency virus 1 / Gene: pol / Plasmid: pXC35 / Production host:  #2: Chemical | ChemComp-SO4 / | #3: Chemical | ChemComp-017 / ( | #4: Water | ChemComp-HOH / | |
|---|
-Experimental details
-Experiment
| Experiment | Method:  X-RAY DIFFRACTION / Number of used crystals: 1 X-RAY DIFFRACTION / Number of used crystals: 1 |
|---|
- Sample preparation
Sample preparation
| Crystal | Density Matthews: 2.09 Å3/Da / Density % sol: 41.05 % |
|---|---|
| Crystal grow | Temperature: 293 K / Method: vapor diffusion, hanging drop Details: 19-26% (w/v) Ammonium Sulfate, 0.1M Bis-Tris-Methane-HCl Buffer pH 5.5 |
-Data collection
| Diffraction | Mean temperature: 100 K / Serial crystal experiment: N |
|---|---|
| Diffraction source | Source:  ROTATING ANODE / Type: RIGAKU MICROMAX-007 HF / Wavelength: 1.54178 Å ROTATING ANODE / Type: RIGAKU MICROMAX-007 HF / Wavelength: 1.54178 Å |
| Detector | Type: RIGAKU SATURN 944 / Detector: CCD / Date: Aug 2, 2018 |
| Radiation | Protocol: SINGLE WAVELENGTH / Monochromatic (M) / Laue (L): M / Scattering type: x-ray |
| Radiation wavelength | Wavelength: 1.54178 Å / Relative weight: 1 |
| Reflection | Resolution: 2.1→50 Å / Num. obs: 10571 / % possible obs: 99.9 % / Redundancy: 10.5 % / Net I/σ(I): 37.1 |
| Reflection shell | Resolution: 2.102→2.3138 Å / Num. unique obs: 2614 |
- Processing
Processing
| Software |
| |||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Refinement | Method to determine structure:  MOLECULAR REPLACEMENT / Resolution: 2.1→24.778 Å / Cross valid method: FREE R-VALUE / σ(F): 5.25 / Phase error: 36.9 MOLECULAR REPLACEMENT / Resolution: 2.1→24.778 Å / Cross valid method: FREE R-VALUE / σ(F): 5.25 / Phase error: 36.9
| |||||||||||||||||||||||||||||||||||
| Solvent computation | Shrinkage radii: 0.9 Å / VDW probe radii: 1.11 Å | |||||||||||||||||||||||||||||||||||
| Refinement step | Cycle: LAST / Resolution: 2.1→24.778 Å
| |||||||||||||||||||||||||||||||||||
| Refine LS restraints |
| |||||||||||||||||||||||||||||||||||
| LS refinement shell |
|
 Movie
Movie Controller
Controller





















 PDBj
PDBj