[English] 日本語
 Yorodumi
Yorodumi- PDB-5i02: tRNA guanine transglycosylase (TGT) in co-crystallized complex wi... -
+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 5i02 | ||||||
|---|---|---|---|---|---|---|---|
| Title | tRNA guanine transglycosylase (TGT) in co-crystallized complex with 6-amino-4-{2-[(cyclohexylmethyl)amino]ethyl}-2-(methylamino)-1,7-dihydro-8H-imidazo[4,5-g]quinazolin-8-one | ||||||
 Components Components | Queuine tRNA-ribosyltransferase | ||||||
 Keywords Keywords |  TRANSFERASE / co-crystallization / TRANSFERASE / co-crystallization /  shigellosis / TRANSFERASE INHIBITOR shigellosis / TRANSFERASE INHIBITOR | ||||||
| Function / homology |  Function and homology information Function and homology informationtRNA-guanosine34 preQ1 transglycosylase / tRNA-guanosine(34) queuine transglycosylase activity / tRNA-guanine transglycosylation / queuosine biosynthetic process /  metal ion binding metal ion bindingSimilarity search - Function | ||||||
| Biological species |   Zymomonas mobilis subsp. mobilis (bacteria) Zymomonas mobilis subsp. mobilis (bacteria) | ||||||
| Method |  X-RAY DIFFRACTION / X-RAY DIFFRACTION /  SYNCHROTRON / SYNCHROTRON /  MOLECULAR REPLACEMENT / Resolution: 1.25 Å MOLECULAR REPLACEMENT / Resolution: 1.25 Å | ||||||
 Authors Authors | Ehrmann, F.R. / Nguyen, D. / Heine, A. / Klebe, G. | ||||||
 Citation Citation |  Journal: To be Published Journal: To be PublishedTitle: Co-crystallization, Isothermal titration calorimetry and nanoESI-MS reveal dimer disturbing inhibitors Authors: Ehrmann, F.R. / Stojko, J. / Heine, A. / Diederich, F. / Reuter, K. / Sanglier-Cianferani, S. / Klebe, G. | ||||||
| History |
|
- Structure visualization
Structure visualization
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  5i02.cif.gz 5i02.cif.gz | 172.8 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb5i02.ent.gz pdb5i02.ent.gz | 135.2 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  5i02.json.gz 5i02.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/i0/5i02 https://data.pdbj.org/pub/pdb/validation_reports/i0/5i02 ftp://data.pdbj.org/pub/pdb/validation_reports/i0/5i02 ftp://data.pdbj.org/pub/pdb/validation_reports/i0/5i02 | HTTPS FTP |
|---|
-Related structure data
| Related structure data |  5i00C  5i03C  5i06C  5i07C  5i09C  1p0dS S: Starting model for refinement C: citing same article ( |
|---|---|
| Similar structure data |
- Links
Links
- Assembly
Assembly
| Deposited unit | 
| ||||||||
|---|---|---|---|---|---|---|---|---|---|
| 1 | 
| ||||||||
| Unit cell |
|
- Components
Components
-Protein , 1 types, 1 molecules A
| #1: Protein |  / Guanine insertion enzyme / tRNA-guanine transglycosylase / Guanine insertion enzyme / tRNA-guanine transglycosylaseMass: 42838.625 Da / Num. of mol.: 1 Source method: isolated from a genetically manipulated source Source: (gene. exp.)   Zymomonas mobilis subsp. mobilis (strain ATCC 31821 / ZM4 / CP4) (bacteria) Zymomonas mobilis subsp. mobilis (strain ATCC 31821 / ZM4 / CP4) (bacteria)Gene: tgt, ZMO0363 / Production host:   Escherichia coli (E. coli) Escherichia coli (E. coli)References: UniProt: P28720, tRNA-guanosine34 preQ1 transglycosylase |
|---|
-Non-polymers , 6 types, 325 molecules 
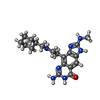









| #2: Chemical | ChemComp-ZN / | ||||||
|---|---|---|---|---|---|---|---|
| #3: Chemical | ChemComp-PK2 / | ||||||
| #4: Chemical |  Ethylene glycol Ethylene glycol#5: Chemical |  Glycerol Glycerol#6: Chemical | ChemComp-CL / |  Chloride Chloride#7: Water | ChemComp-HOH / |  Water Water |
-Experimental details
-Experiment
| Experiment | Method:  X-RAY DIFFRACTION / Number of used crystals: 1 X-RAY DIFFRACTION / Number of used crystals: 1 |
|---|
- Sample preparation
Sample preparation
| Crystal | Density Matthews: 2.26 Å3/Da / Density % sol: 45.69 % |
|---|---|
Crystal grow | Temperature: 288 K / Method: vapor diffusion, hanging drop / pH: 5.5 / Details: 13% PEG 8000,100mM MES, 1mM DTT, 10% DMSO |
-Data collection
| Diffraction | Mean temperature: 100 K |
|---|---|
| Diffraction source | Source:  SYNCHROTRON / Site: SYNCHROTRON / Site:  ELETTRA ELETTRA  / Beamline: 5.2R / Wavelength: 1 Å / Beamline: 5.2R / Wavelength: 1 Å |
| Detector | Type: DECTRIS PILATUS 2M / Detector: PIXEL / Date: Aug 3, 2015 |
| Radiation | Protocol: SINGLE WAVELENGTH / Monochromatic (M) / Laue (L): M / Scattering type: x-ray |
| Radiation wavelength | Wavelength : 1 Å / Relative weight: 1 : 1 Å / Relative weight: 1 |
| Reflection | Resolution: 1.25→42.53 Å / Num. obs: 105188 / % possible obs: 99.1 % / Redundancy: 3.3 % / Rsym value: 0.055 / Net I/σ(I): 11.3 |
| Reflection shell | Resolution: 1.25→1.33 Å / Redundancy: 3.3 % / Rmerge(I) obs: 0.491 / Mean I/σ(I) obs: 2.1 / % possible all: 97.9 |
- Processing
Processing
| Software |
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Refinement | Method to determine structure : :  MOLECULAR REPLACEMENT MOLECULAR REPLACEMENTStarting model: 1P0D Resolution: 1.25→42.152 Å / SU ML: 0.1 / Cross valid method: FREE R-VALUE / σ(F): 1.35 / Phase error: 14.71
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Solvent computation | Shrinkage radii: 0.9 Å / VDW probe radii: 1.11 Å | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refinement step | Cycle: LAST / Resolution: 1.25→42.152 Å
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refine LS restraints |
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| LS refinement shell |
|
 Movie
Movie Controller
Controller

































 PDBj
PDBj




