Entry Database : PDB / ID : 4xfsTitle Structure of IL-18 SER Mutant I Interleukin-18 Keywords / / / / Function / homology Function Domain/homology Component
/ / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / Biological species Homo sapiens (human)Method / / / Resolution : 1.91 Å Authors Krumm, B.E. / Meng, X. / Xiang, Y. / Deng, J. Funding support Organization Grant number Country National Institutes of Health/National Institute Of Allergy and Infectious Diseases (NIH/NIAID) AI113539 National Institutes of Health/National Institute Of Allergy and Infectious Diseases (NIH/NIAID) AI079217 Oklahoma State University OKL02848
Journal : Acta Crystallogr.,Sect.F / Year : 2015Title : Crystallization of interleukin-18 for structure-based inhibitor design.Authors : Krumm, B. / Meng, X. / Xiang, Y. / Deng, J. History Deposition Dec 28, 2014 Deposition site / Processing site Revision 1.0 Jun 10, 2015 Provider / Type Revision 1.1 Jul 1, 2015 Group Revision 1.2 Sep 20, 2017 Group Advisory / Author supporting evidence ... Advisory / Author supporting evidence / Derived calculations / Source and taxonomy Category entity_src_gen / pdbx_audit_support ... entity_src_gen / pdbx_audit_support / pdbx_struct_assembly / pdbx_struct_assembly_gen / pdbx_struct_assembly_prop / pdbx_struct_oper_list / pdbx_unobs_or_zero_occ_atoms Item _entity_src_gen.pdbx_alt_source_flag / _pdbx_audit_support.funding_organization ... _entity_src_gen.pdbx_alt_source_flag / _pdbx_audit_support.funding_organization / _pdbx_struct_assembly.oligomeric_details / _pdbx_struct_assembly_gen.asym_id_list / _pdbx_struct_assembly_prop.type / _pdbx_struct_assembly_prop.value / _pdbx_struct_oper_list.symmetry_operation Revision 1.3 Dec 11, 2019 Group / Category / Item Revision 1.4 Sep 27, 2023 Group Advisory / Data collection ... Advisory / Data collection / Database references / Refinement description Category chem_comp_atom / chem_comp_bond ... chem_comp_atom / chem_comp_bond / database_2 / pdbx_initial_refinement_model / pdbx_unobs_or_zero_occ_atoms / struct_ncs_dom_lim Item _database_2.pdbx_DOI / _database_2.pdbx_database_accession ... _database_2.pdbx_DOI / _database_2.pdbx_database_accession / _struct_ncs_dom_lim.beg_auth_comp_id / _struct_ncs_dom_lim.beg_label_asym_id / _struct_ncs_dom_lim.beg_label_comp_id / _struct_ncs_dom_lim.beg_label_seq_id / _struct_ncs_dom_lim.end_auth_comp_id / _struct_ncs_dom_lim.end_label_asym_id / _struct_ncs_dom_lim.end_label_comp_id / _struct_ncs_dom_lim.end_label_seq_id
Show all Show less
 Open data
Open data Basic information
Basic information Components
Components Keywords
Keywords Function and homology information
Function and homology information Homo sapiens (human)
Homo sapiens (human) X-RAY DIFFRACTION /
X-RAY DIFFRACTION /  SYNCHROTRON /
SYNCHROTRON /  MOLECULAR REPLACEMENT / Resolution: 1.91 Å
MOLECULAR REPLACEMENT / Resolution: 1.91 Å  Authors
Authors United States, 3items
United States, 3items  Citation
Citation Journal: Acta Crystallogr.,Sect.F / Year: 2015
Journal: Acta Crystallogr.,Sect.F / Year: 2015 Structure visualization
Structure visualization Molmil
Molmil Jmol/JSmol
Jmol/JSmol Downloads & links
Downloads & links Download
Download 4xfs.cif.gz
4xfs.cif.gz PDBx/mmCIF format
PDBx/mmCIF format pdb4xfs.ent.gz
pdb4xfs.ent.gz PDB format
PDB format 4xfs.json.gz
4xfs.json.gz PDBx/mmJSON format
PDBx/mmJSON format Other downloads
Other downloads 4xfs_validation.pdf.gz
4xfs_validation.pdf.gz wwPDB validaton report
wwPDB validaton report 4xfs_full_validation.pdf.gz
4xfs_full_validation.pdf.gz 4xfs_validation.xml.gz
4xfs_validation.xml.gz 4xfs_validation.cif.gz
4xfs_validation.cif.gz https://data.pdbj.org/pub/pdb/validation_reports/xf/4xfs
https://data.pdbj.org/pub/pdb/validation_reports/xf/4xfs ftp://data.pdbj.org/pub/pdb/validation_reports/xf/4xfs
ftp://data.pdbj.org/pub/pdb/validation_reports/xf/4xfs


 Links
Links Assembly
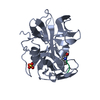
Assembly


 Components
Components Homo sapiens (human) / Gene: IL18, IGIF, IL1F4 / Plasmid: pET / Production host:
Homo sapiens (human) / Gene: IL18, IGIF, IL1F4 / Plasmid: pET / Production host: 
 X-RAY DIFFRACTION
X-RAY DIFFRACTION Sample preparation
Sample preparation SYNCHROTRON / Site:
SYNCHROTRON / Site:  APS
APS  / Beamline: 19-ID / Wavelength: 0.97926 Å
/ Beamline: 19-ID / Wavelength: 0.97926 Å Processing
Processing MOLECULAR REPLACEMENT
MOLECULAR REPLACEMENT Movie
Movie Controller
Controller












 PDBj
PDBj

















