+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 1uti | ||||||
|---|---|---|---|---|---|---|---|
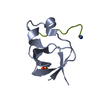
| Title | Mona/Gads SH3C in complex with HPK derived peptide | ||||||
 Components Components |
| ||||||
 Keywords Keywords |  SIGNALING PROTEIN REGULATOR / SH3 DOMAIN-COMPLEX / SIGNALING PROTEIN REGULATOR / SH3 DOMAIN-COMPLEX /  ADAPTOR PROTEIN (MONA) / PROTEIN SERINE/THREONINE KINASE (HPK1) / ANTIGEN RECEPTOR SIGNALLING MEDIATOR (BOTH) / ADAPTOR PROTEIN (MONA) / PROTEIN SERINE/THREONINE KINASE (HPK1) / ANTIGEN RECEPTOR SIGNALLING MEDIATOR (BOTH) /  SH3 DOMAIN / PPII HELIX SH3 DOMAIN / PPII HELIX | ||||||
| Function / homology |  Function and homology information Function and homology information : / FLT3 Signaling / CD28 co-stimulation / FCERI mediated MAPK activation / Signaling by SCF-KIT / : / FLT3 Signaling / CD28 co-stimulation / FCERI mediated MAPK activation / Signaling by SCF-KIT /  MAP kinase kinase kinase kinase activity / Generation of second messenger molecules / FCERI mediated Ca+2 mobilization / cellular response to phorbol 13-acetate 12-myristate / DAP12 signaling ... MAP kinase kinase kinase kinase activity / Generation of second messenger molecules / FCERI mediated Ca+2 mobilization / cellular response to phorbol 13-acetate 12-myristate / DAP12 signaling ... : / FLT3 Signaling / CD28 co-stimulation / FCERI mediated MAPK activation / Signaling by SCF-KIT / : / FLT3 Signaling / CD28 co-stimulation / FCERI mediated MAPK activation / Signaling by SCF-KIT /  MAP kinase kinase kinase kinase activity / Generation of second messenger molecules / FCERI mediated Ca+2 mobilization / cellular response to phorbol 13-acetate 12-myristate / DAP12 signaling / MAP kinase kinase kinase kinase activity / Generation of second messenger molecules / FCERI mediated Ca+2 mobilization / cellular response to phorbol 13-acetate 12-myristate / DAP12 signaling /  regulation of MAPK cascade / phosphotyrosine residue binding / peptidyl-serine phosphorylation / cell population proliferation / protein autophosphorylation / regulation of MAPK cascade / phosphotyrosine residue binding / peptidyl-serine phosphorylation / cell population proliferation / protein autophosphorylation /  non-specific serine/threonine protein kinase / non-specific serine/threonine protein kinase /  endosome / intracellular signal transduction / endosome / intracellular signal transduction /  protein phosphorylation / protein serine/threonine kinase activity / protein phosphorylation / protein serine/threonine kinase activity /  signal transduction / signal transduction /  nucleoplasm / nucleoplasm /  ATP binding / ATP binding /  nucleus / nucleus /  plasma membrane / plasma membrane /  cytosol / cytosol /  cytoplasm cytoplasmSimilarity search - Function | ||||||
| Biological species |   MUS MUSCULUS (house mouse) MUS MUSCULUS (house mouse) | ||||||
| Method |  X-RAY DIFFRACTION / X-RAY DIFFRACTION /  SYNCHROTRON / SYNCHROTRON /  MOLECULAR REPLACEMENT / Resolution: 1.5 Å MOLECULAR REPLACEMENT / Resolution: 1.5 Å | ||||||
 Authors Authors | Lewitzky, M. / Harkiolaki, M. / Domart, M.C. / Feller, S.M. | ||||||
 Citation Citation |  Journal: J.Biol.Chem. / Year: 2004 Journal: J.Biol.Chem. / Year: 2004Title: Mona/Gads Sh3C Binding to Hematopoietic Progenitor Kinase 1 (Hpk1) Combines an Atypical SH3 Binding Motif, R/Kxxk, with a Classical Pxxp Motif Embedded in a Polyproline Type II (Ppii) Helix Authors: Lewitzky, M. / Harkiolaki, M. / Domart, M.C. / Jones, E. / Feller, S.M. | ||||||
| History |
|
- Structure visualization
Structure visualization
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  1uti.cif.gz 1uti.cif.gz | 28.5 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb1uti.ent.gz pdb1uti.ent.gz | 18.6 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  1uti.json.gz 1uti.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/ut/1uti https://data.pdbj.org/pub/pdb/validation_reports/ut/1uti ftp://data.pdbj.org/pub/pdb/validation_reports/ut/1uti ftp://data.pdbj.org/pub/pdb/validation_reports/ut/1uti | HTTPS FTP |
|---|
-Related structure data
| Related structure data |  1oebS S: Starting model for refinement |
|---|---|
| Similar structure data |
- Links
Links
- Assembly
Assembly
| Deposited unit | 
| ||||||||
|---|---|---|---|---|---|---|---|---|---|
| 1 |
| ||||||||
| Unit cell |
| ||||||||
| Details | MONA/GADS SH3C IS A MONOMER IN SOLUTION, PRESENT IN THISCRYSTAL IN COMPLEX WITH AN HPK1 DERIVED PEPTIDE AND HENCEIN A HETERODIMERIC STATE. |
- Components
Components
| #1: Protein | Mass: 6734.568 Da / Num. of mol.: 1 / Fragment: SH3C DOMAIN, RESIDUES 265-322 Source method: isolated from a genetically manipulated source Source: (gene. exp.)   MUS MUSCULUS (house mouse) / Plasmid: PGEX-6P-1 / Production host: MUS MUSCULUS (house mouse) / Plasmid: PGEX-6P-1 / Production host:   ESCHERICHIA COLI (E. coli) / Strain (production host): BL21(DE3) / References: UniProt: O89100 ESCHERICHIA COLI (E. coli) / Strain (production host): BL21(DE3) / References: UniProt: O89100 | ||
|---|---|---|---|
| #2: Protein/peptide | Mass: 1823.232 Da / Num. of mol.: 1 / Fragment: SH3 BINDING PEPTIDE, RESIDUES 465-480 / Source method: obtained synthetically / Source: (synth.)   MUS MUSCULUS (house mouse) / References: UniProt: P70218, EC: 2.7.1.37 MUS MUSCULUS (house mouse) / References: UniProt: P70218, EC: 2.7.1.37 | ||
| #3: Water | ChemComp-HOH /  Water Water | ||
| Compound details | GADS/MONA:INTERACTS WITH SLP-76 TO REGULATE NF-AT ACTIVATION| Sequence details | ONLY THE C-TERMINAL DOMAIN HAS BEEN CRYSTALLIS | |
-Experimental details
-Experiment
| Experiment | Method:  X-RAY DIFFRACTION / Number of used crystals: 1 X-RAY DIFFRACTION / Number of used crystals: 1 |
|---|
- Sample preparation
Sample preparation
| Crystal | Density Matthews: 1.8 Å3/Da / Density % sol: 32.7 % |
|---|---|
Crystal grow | pH: 7.5 / Details: 2M AMMONIUM SULFATE, 0.05M HEPES PH7.5, pH 7.50 |
-Data collection
| Diffraction | Mean temperature: 100 K |
|---|---|
| Diffraction source | Source:  SYNCHROTRON / Site: SYNCHROTRON / Site:  ESRF ESRF  / Beamline: BM14 / Wavelength: 0.97932 / Beamline: BM14 / Wavelength: 0.97932 |
| Detector | Type: MARRESEARCH / Detector: CCD / Date: Aug 15, 2003 / Details: MIRRORS |
| Radiation | Monochromator: SI / Protocol: SINGLE WAVELENGTH / Monochromatic (M) / Laue (L): M / Scattering type: x-ray |
| Radiation wavelength | Wavelength : 0.97932 Å / Relative weight: 1 : 0.97932 Å / Relative weight: 1 |
| Reflection | Resolution: 1.5→20 Å / Num. obs: 9855 / % possible obs: 97.4 % / Redundancy: 3.4 % / Rmerge(I) obs: 0.03 / Net I/σ(I): 25.2 |
| Reflection shell | Resolution: 1.5→1.55 Å / Redundancy: 3 % / Rmerge(I) obs: 0.065 / Mean I/σ(I) obs: 14.7 / % possible all: 98.2 |
- Processing
Processing
| Software |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Refinement | Method to determine structure : :  MOLECULAR REPLACEMENT MOLECULAR REPLACEMENTStarting model: PDB ENTRY 1OEB Resolution: 1.5→34.71 Å / Cor.coef. Fo:Fc: 0.944 / Cor.coef. Fo:Fc free: 0.94 / SU B: 1.308 / SU ML: 0.051 / Cross valid method: THROUGHOUT / ESU R: 0.098 / ESU R Free: 0.089 / Stereochemistry target values: MAXIMUM LIKELIHOOD / Details: HYDROGENS HAVE BEEN ADDED IN THE RIDING POSITIONS
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Solvent computation | Ion probe radii: 0.8 Å / Shrinkage radii: 0.8 Å / VDW probe radii: 1.4 Å / Solvent model: BABINET MODEL WITH MASK | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Displacement parameters | Biso mean: 14.04 Å2
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refinement step | Cycle: LAST / Resolution: 1.5→34.71 Å
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refine LS restraints |
|
 Movie
Movie Controller
Controller













 PDBj
PDBj



