+ データを開く
データを開く
- 基本情報
基本情報
| 登録情報 | データベース: EMDB / ID: EMD-20055 | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
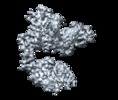
| タイトル | Cryo-EM structure of Her2 extracellular domain-Trastuzumab Fab-Pertuzumab Fab complex | |||||||||
 マップデータ マップデータ | Sharpened map with phenix.auto_sharpen | |||||||||
 試料 試料 |
| |||||||||
| 機能・相同性 |  機能・相同性情報 機能・相同性情報negative regulation of immature T cell proliferation in thymus / ERBB3:ERBB2 complex / PLCG1 events in ERBB2 signaling / ERBB2-ERBB4 signaling pathway / IgD immunoglobulin complex / GRB7 events in ERBB2 signaling / immature T cell proliferation in thymus / RNA polymerase I core binding / ERBB2-EGFR signaling pathway / IgM immunoglobulin complex ...negative regulation of immature T cell proliferation in thymus / ERBB3:ERBB2 complex / PLCG1 events in ERBB2 signaling / ERBB2-ERBB4 signaling pathway / IgD immunoglobulin complex / GRB7 events in ERBB2 signaling / immature T cell proliferation in thymus / RNA polymerase I core binding / ERBB2-EGFR signaling pathway / IgM immunoglobulin complex / IgA immunoglobulin complex / IgE immunoglobulin complex / ERBB2 Activates PTK6 Signaling / semaphorin receptor complex / regulation of microtubule-based process / CD22 mediated BCR regulation / ErbB-3 class receptor binding / ERBB2 Regulates Cell Motility / Sema4D induced cell migration and growth-cone collapse / motor neuron axon guidance / IgG immunoglobulin complex / Fc epsilon receptor (FCERI) signaling / PI3K events in ERBB2 signaling / neurotransmitter receptor localization to postsynaptic specialization membrane / Classical antibody-mediated complement activation / positive regulation of Rho protein signal transduction / neuromuscular junction development / Initial triggering of complement / Drug-mediated inhibition of ERBB2 signaling / Resistance of ERBB2 KD mutants to trastuzumab / Resistance of ERBB2 KD mutants to sapitinib / Resistance of ERBB2 KD mutants to tesevatinib / Resistance of ERBB2 KD mutants to neratinib / Resistance of ERBB2 KD mutants to osimertinib / Resistance of ERBB2 KD mutants to afatinib / Resistance of ERBB2 KD mutants to AEE788 / Resistance of ERBB2 KD mutants to lapatinib / Drug resistance in ERBB2 TMD/JMD mutants / enzyme-linked receptor protein signaling pathway / ERBB2-ERBB3 signaling pathway / positive regulation of transcription by RNA polymerase I / oligodendrocyte differentiation / semaphorin-plexin signaling pathway / cellular response to epidermal growth factor stimulus / Signaling by ERBB2 / positive regulation of cell adhesion / FCGR activation / immunoglobulin mediated immune response / positive regulation of protein targeting to membrane / TFAP2 (AP-2) family regulates transcription of growth factors and their receptors / GRB2 events in ERBB2 signaling / Role of phospholipids in phagocytosis / transmembrane receptor protein tyrosine kinase activity / Role of LAT2/NTAL/LAB on calcium mobilization / regulation of angiogenesis / neurogenesis / SHC1 events in ERBB2 signaling / Scavenging of heme from plasma / immunoglobulin complex, circulating / coreceptor activity / immunoglobulin receptor binding / regulation of ERK1 and ERK2 cascade / Schwann cell development / basal plasma membrane / positive regulation of epithelial cell proliferation / cell surface receptor protein tyrosine kinase signaling pathway / myelination / phosphatidylinositol 3-kinase/protein kinase B signal transduction / Downregulation of ERBB2:ERBB3 signaling / Constitutive Signaling by Overexpressed ERBB2 / FCERI mediated Ca+2 mobilization / FCGR3A-mediated IL10 synthesis / complement activation, classical pathway / Signaling by ERBB2 TMD/JMD mutants / positive regulation of MAP kinase activity / Antigen activates B Cell Receptor (BCR) leading to generation of second messengers / Signaling by ERBB2 ECD mutants / Regulation of Complement cascade / antigen binding / Signaling by ERBB2 KD Mutants / positive regulation of translation / receptor protein-tyrosine kinase / Cell surface interactions at the vascular wall / FCERI mediated MAPK activation / FCGR3A-mediated phagocytosis / Downregulation of ERBB2 signaling / B cell receptor signaling pathway / ruffle membrane / wound healing / neuromuscular junction / peptidyl-tyrosine phosphorylation / neuron differentiation / transmembrane signaling receptor activity / Regulation of actin dynamics for phagocytic cup formation / receptor tyrosine kinase binding / cellular response to growth factor stimulus / Constitutive Signaling by Aberrant PI3K in Cancer / FCERI mediated NF-kB activation / Immunoregulatory interactions between a Lymphoid and a non-Lymphoid cell / PIP3 activates AKT signaling 類似検索 - 分子機能 | |||||||||
| 生物種 |  Homo sapiens (ヒト) Homo sapiens (ヒト) | |||||||||
| 手法 | 単粒子再構成法 / クライオ電子顕微鏡法 / 解像度: 4.36 Å | |||||||||
 データ登録者 データ登録者 | Hao Y / Yu X / Bai Y / Huang X | |||||||||
 引用 引用 |  ジャーナル: PLoS One / 年: 2019 ジャーナル: PLoS One / 年: 2019タイトル: Cryo-EM Structure of HER2-trastuzumab-pertuzumab complex. 著者: Yue Hao / Xinchao Yu / Yonghong Bai / Helen J McBride / Xin Huang /  要旨: Trastuzumab and pertuzumab are monoclonal antibodies that bind to distinct subdomains of the extracellular domain of human epidermal growth factor receptor 2 (HER2). Adding these monoclonal ...Trastuzumab and pertuzumab are monoclonal antibodies that bind to distinct subdomains of the extracellular domain of human epidermal growth factor receptor 2 (HER2). Adding these monoclonal antibodies to the treatment regimen of HER2-positive breast cancer has changed the paradigm for treatment in that form of cancer. Synergistic activity has been observed with the combination of these two antibodies leading to hypotheses regarding the mechanism(s) and to the development of bispecific antibodies to maximize the clinical effect further. Although the individual crystal structures of HER2-trastuzumab and HER2-pertuzumab revealed the distinct binding sites and provided the structural basis for their anti-tumor activities, detailed structural information on the HER2-trastuzumab-pertuzumab complex has been elusive. Here we present the cryo-EM structure of HER2-trastuzumab-pertuzumab at 4.36 Å resolution. Comparison with the binary complexes reveals no cooperative interaction between trastuzumab and pertuzumab, and provides key insights into the design of novel, high-avidity bispecific molecules with potentially greater clinical efficacy. | |||||||||
| 履歴 |
|
- 構造の表示
構造の表示
| ムービー |
 ムービービューア ムービービューア |
|---|---|
| 構造ビューア | EMマップ:  SurfView SurfView Molmil Molmil Jmol/JSmol Jmol/JSmol |
| 添付画像 |
- ダウンロードとリンク
ダウンロードとリンク
-EMDBアーカイブ
| マップデータ |  emd_20055.map.gz emd_20055.map.gz | 31.9 MB |  EMDBマップデータ形式 EMDBマップデータ形式 | |
|---|---|---|---|---|
| ヘッダ (付随情報) |  emd-20055-v30.xml emd-20055-v30.xml emd-20055.xml emd-20055.xml | 19 KB 19 KB | 表示 表示 |  EMDBヘッダ EMDBヘッダ |
| 画像 |  emd_20055.png emd_20055.png | 145.4 KB | ||
| アーカイブディレクトリ |  http://ftp.pdbj.org/pub/emdb/structures/EMD-20055 http://ftp.pdbj.org/pub/emdb/structures/EMD-20055 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-20055 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-20055 | HTTPS FTP |
-検証レポート
| 文書・要旨 |  emd_20055_validation.pdf.gz emd_20055_validation.pdf.gz | 475.5 KB | 表示 |  EMDB検証レポート EMDB検証レポート |
|---|---|---|---|---|
| 文書・詳細版 |  emd_20055_full_validation.pdf.gz emd_20055_full_validation.pdf.gz | 475.1 KB | 表示 | |
| XML形式データ |  emd_20055_validation.xml.gz emd_20055_validation.xml.gz | 6 KB | 表示 | |
| CIF形式データ |  emd_20055_validation.cif.gz emd_20055_validation.cif.gz | 6.8 KB | 表示 | |
| アーカイブディレクトリ |  https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-20055 https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-20055 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-20055 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-20055 | HTTPS FTP |
-関連構造データ
- リンク
リンク
| EMDBのページ |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|---|
| 「今月の分子」の関連する項目 |
- マップ
マップ
| ファイル |  ダウンロード / ファイル: emd_20055.map.gz / 形式: CCP4 / 大きさ: 34.3 MB / タイプ: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) ダウンロード / ファイル: emd_20055.map.gz / 形式: CCP4 / 大きさ: 34.3 MB / タイプ: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 注釈 | Sharpened map with phenix.auto_sharpen | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| ボクセルのサイズ | X=Y=Z: 1.059 Å | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 密度 |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 対称性 | 空間群: 1 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 詳細 | EMDB XML:
CCP4マップ ヘッダ情報:
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
-添付データ
- 試料の構成要素
試料の構成要素
+全体 : Her2 extracellular domain-Trastuzumab Fab-Pertuzumab Fab complex
+超分子 #1: Her2 extracellular domain-Trastuzumab Fab-Pertuzumab Fab complex
+超分子 #2: Human HER2 extracellular domain
+超分子 #3: Pertuzumab Fab
+超分子 #4: Trastuzumab Fab
+分子 #1: Receptor tyrosine-protein kinase erbB-2
+分子 #2: Pertuzumab FAB LIGHT CHAIN
+分子 #3: Pertuzumab FAB HEAVY CHAIN
+分子 #4: Trastuzumab FAB LIGHT CHAIN
+分子 #5: Trastuzumab FAB HEAVY CHAIN
+分子 #7: 2-acetamido-2-deoxy-beta-D-glucopyranose
-実験情報
-構造解析
| 手法 | クライオ電子顕微鏡法 |
|---|---|
 解析 解析 | 単粒子再構成法 |
| 試料の集合状態 | particle |
- 試料調製
試料調製
| 濃度 | 2.4 mg/mL | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| 緩衝液 | pH: 7.5 構成要素:
| |||||||||
| グリッド | モデル: Quantifoil R1.2/1.3 / メッシュ: 300 | |||||||||
| 凍結 | 凍結剤: ETHANE / チャンバー内湿度: 100 % / 装置: FEI VITROBOT MARK IV |
- 電子顕微鏡法
電子顕微鏡法
| 顕微鏡 | FEI TITAN KRIOS |
|---|---|
| 撮影 | フィルム・検出器のモデル: GATAN K2 SUMMIT (4k x 4k) 検出モード: SUPER-RESOLUTION / 平均露光時間: 6.0 sec. / 平均電子線量: 45.0 e/Å2 |
| 電子線 | 加速電圧: 300 kV / 電子線源:  FIELD EMISSION GUN FIELD EMISSION GUN |
| 電子光学系 | 照射モード: FLOOD BEAM / 撮影モード: BRIGHT FIELD / Cs: 2.7 mm / 最大 デフォーカス(公称値): -1.5 µm / 最小 デフォーカス(公称値): -3.5 µm / 倍率(公称値): 130000 |
| 実験機器 |  モデル: Titan Krios / 画像提供: FEI Company |
+ 画像解析
画像解析
-原子モデル構築 1
| 精密化 | 空間: REAL / プロトコル: FLEXIBLE FIT |
|---|---|
| 得られたモデル |  PDB-6oge: |
 ムービー
ムービー コントローラー
コントローラー