+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 9hj3 | |||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Title | Bacteroides thetaiotaomicron BAM complex | |||||||||||||||
 Components Components |
| |||||||||||||||
 Keywords Keywords | MEMBRANE PROTEIN / Outer membrane protein biogenesis / Beta-barrel assembly machinery / BAM | |||||||||||||||
| Function / homology |  Function and homology information Function and homology informationmembrane assembly / peptidylprolyl isomerase / peptidyl-prolyl cis-trans isomerase activity / cell outer membrane Similarity search - Function | |||||||||||||||
| Biological species |  Bacteroides thetaiotaomicron VPI-5482 (bacteria) Bacteroides thetaiotaomicron VPI-5482 (bacteria) | |||||||||||||||
| Method | ELECTRON MICROSCOPY / single particle reconstruction / cryo EM / Resolution: 3.46 Å | |||||||||||||||
 Authors Authors | Silale, A. / van den Berg, B. | |||||||||||||||
| Funding support |  United Kingdom, 1items United Kingdom, 1items
| |||||||||||||||
 Citation Citation |  Journal: To Be Published Journal: To Be PublishedTitle: To be updated Authors: Silale, A. / van den Berg, B. | |||||||||||||||
| History |
|
- Structure visualization
Structure visualization
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  9hj3.cif.gz 9hj3.cif.gz | 454.2 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb9hj3.ent.gz pdb9hj3.ent.gz | Display |  PDB format PDB format | |
| PDBx/mmJSON format |  9hj3.json.gz 9hj3.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Summary document |  9hj3_validation.pdf.gz 9hj3_validation.pdf.gz | 973 KB | Display |  wwPDB validaton report wwPDB validaton report |
|---|---|---|---|---|
| Full document |  9hj3_full_validation.pdf.gz 9hj3_full_validation.pdf.gz | 1010 KB | Display | |
| Data in XML |  9hj3_validation.xml.gz 9hj3_validation.xml.gz | 68.9 KB | Display | |
| Data in CIF |  9hj3_validation.cif.gz 9hj3_validation.cif.gz | 107.6 KB | Display | |
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/hj/9hj3 https://data.pdbj.org/pub/pdb/validation_reports/hj/9hj3 ftp://data.pdbj.org/pub/pdb/validation_reports/hj/9hj3 ftp://data.pdbj.org/pub/pdb/validation_reports/hj/9hj3 | HTTPS FTP |
-Related structure data
| Related structure data |  52209MC  8eqvC M: map data used to model this data C: citing same article ( |
|---|---|
| Similar structure data | Similarity search - Function & homology  F&H Search F&H Search |
- Links
Links
- Assembly
Assembly
| Deposited unit | 
|
|---|---|
| 1 |
|
- Components
Components
-Outer membrane ... , 2 types, 2 molecules AF
| #1: Protein | Mass: 101527.664 Da / Num. of mol.: 1 Source method: isolated from a genetically manipulated source Source: (gene. exp.)  Bacteroides thetaiotaomicron VPI-5482 (bacteria) Bacteroides thetaiotaomicron VPI-5482 (bacteria)Gene: BT_3725 Production host:  Bacteroides thetaiotaomicron VPI-5482 (bacteria) Bacteroides thetaiotaomicron VPI-5482 (bacteria)References: UniProt: Q8A1E1 |
|---|---|
| #3: Protein | Mass: 47860.691 Da / Num. of mol.: 1 / Source method: isolated from a natural source Source: (natural)  Bacteroides thetaiotaomicron VPI-5482 (bacteria) Bacteroides thetaiotaomicron VPI-5482 (bacteria)References: UniProt: Q89ZL0 |
-Protein , 5 types, 5 molecules DHIJG
| #2: Protein | Mass: 31415.555 Da / Num. of mol.: 1 / Source method: isolated from a natural source Source: (natural)  Bacteroides thetaiotaomicron VPI-5482 (bacteria) Bacteroides thetaiotaomicron VPI-5482 (bacteria)References: UniProt: Q8AA92 |
|---|---|
| #4: Protein | Mass: 55108.051 Da / Num. of mol.: 1 / Source method: isolated from a natural source Source: (natural)  Bacteroides thetaiotaomicron VPI-5482 (bacteria) Bacteroides thetaiotaomicron VPI-5482 (bacteria)References: UniProt: Q8A1D9 |
| #5: Protein | Mass: 21884.557 Da / Num. of mol.: 1 / Source method: isolated from a natural source Source: (natural)  Bacteroides thetaiotaomicron VPI-5482 (bacteria) Bacteroides thetaiotaomicron VPI-5482 (bacteria)References: UniProt: Q8A1P7, peptidylprolyl isomerase |
| #6: Protein | Mass: 24712.709 Da / Num. of mol.: 1 / Source method: isolated from a natural source Source: (natural)  Bacteroides thetaiotaomicron VPI-5482 (bacteria) Bacteroides thetaiotaomicron VPI-5482 (bacteria)References: UniProt: Q8A0S2 |
| #7: Protein | Mass: 58264.816 Da / Num. of mol.: 1 / Source method: isolated from a natural source Source: (natural)  Bacteroides thetaiotaomicron VPI-5482 (bacteria) Bacteroides thetaiotaomicron VPI-5482 (bacteria)References: UniProt: Q89ZS0 |
-Non-polymers , 2 types, 2 molecules 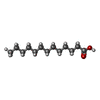


| #8: Chemical | ChemComp-TDA / |
|---|---|
| #9: Chemical | ChemComp-Z41 / ( |
-Details
| Has ligand of interest | N |
|---|---|
| Has protein modification | Y |
-Experimental details
-Experiment
| Experiment | Method: ELECTRON MICROSCOPY |
|---|---|
| EM experiment | Aggregation state: PARTICLE / 3D reconstruction method: single particle reconstruction |
- Sample preparation
Sample preparation
| Component | Name: Bacteroides thetaiotaomicron BAM complex / Type: COMPLEX / Entity ID: #1-#7 / Source: NATURAL | ||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Molecular weight | Value: 0.325 MDa / Experimental value: NO | ||||||||||||||||||||
| Source (natural) | Organism:  Bacteroides thetaiotaomicron VPI-5482 (bacteria) Bacteroides thetaiotaomicron VPI-5482 (bacteria) | ||||||||||||||||||||
| Buffer solution | pH: 7.5 | ||||||||||||||||||||
| Buffer component |
| ||||||||||||||||||||
| Specimen | Conc.: 8 mg/ml / Embedding applied: NO / Shadowing applied: NO / Staining applied: NO / Vitrification applied: YES | ||||||||||||||||||||
| Specimen support | Grid material: COPPER / Grid mesh size: 200 divisions/in. / Grid type: Quantifoil R1.2/1.3 | ||||||||||||||||||||
| Vitrification | Instrument: FEI VITROBOT MARK IV / Cryogen name: ETHANE / Humidity: 100 % / Chamber temperature: 277 K |
- Electron microscopy imaging
Electron microscopy imaging
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
|---|---|
| Microscopy | Model: TFS KRIOS |
| Electron gun | Electron source:  FIELD EMISSION GUN / Accelerating voltage: 300 kV / Illumination mode: FLOOD BEAM FIELD EMISSION GUN / Accelerating voltage: 300 kV / Illumination mode: FLOOD BEAM |
| Electron lens | Mode: BRIGHT FIELD / Nominal magnification: 165000 X / Nominal defocus max: 2000 nm / Nominal defocus min: 800 nm |
| Specimen holder | Cryogen: NITROGEN / Specimen holder model: FEI TITAN KRIOS AUTOGRID HOLDER |
| Image recording | Electron dose: 35 e/Å2 / Film or detector model: FEI FALCON IV (4k x 4k) / Num. of grids imaged: 1 / Num. of real images: 13558 |
| EM imaging optics | Energyfilter name: TFS Selectris / Energyfilter slit width: 10 eV |
- Processing
Processing
| EM software | Name: PHENIX / Category: model refinement | ||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| CTF correction | Type: PHASE FLIPPING AND AMPLITUDE CORRECTION | ||||||||||||||||||||||||
| Particle selection | Num. of particles selected: 2266553 | ||||||||||||||||||||||||
| Symmetry | Point symmetry: C1 (asymmetric) | ||||||||||||||||||||||||
| 3D reconstruction | Resolution: 3.46 Å / Resolution method: OTHER / Num. of particles: 105155 / Algorithm: FOURIER SPACE Details: The best parts of two maps at 3.28 A and 3.46 A were combined using phenix.combine_focused_maps Num. of class averages: 2 / Symmetry type: POINT | ||||||||||||||||||||||||
| Atomic model building | B value: 64.9 / Protocol: RIGID BODY FIT / Space: REAL Details: Experimental models were fit into the combined focused map. | ||||||||||||||||||||||||
| Atomic model building |
| ||||||||||||||||||||||||
| Refine LS restraints |
|
 Movie
Movie Controller
Controller






 PDBj
PDBj













