+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 9b70 | ||||||
|---|---|---|---|---|---|---|---|
| Title | Cryo-EM structure of MraY in complex with analogue 2 | ||||||
 Components Components |
| ||||||
 Keywords Keywords | TRANSFERASE / inhibitor / antibiotics | ||||||
| Function / homology |  Function and homology information Function and homology informationphospho-N-acetylmuramoyl-pentapeptide-transferase / UDP-N-acetylmuramoyl-L-alanyl-D-glutamyl-meso-2,6-diaminopimelyl-D-alanyl-D-alanine:undecaprenyl-phosphate transferase activity / phospho-N-acetylmuramoyl-pentapeptide-transferase activity / cell wall macromolecule biosynthetic process / phosphotransferase activity, for other substituted phosphate groups / peptidoglycan biosynthetic process / cell wall organization / regulation of cell shape / cell division / identical protein binding ...phospho-N-acetylmuramoyl-pentapeptide-transferase / UDP-N-acetylmuramoyl-L-alanyl-D-glutamyl-meso-2,6-diaminopimelyl-D-alanyl-D-alanine:undecaprenyl-phosphate transferase activity / phospho-N-acetylmuramoyl-pentapeptide-transferase activity / cell wall macromolecule biosynthetic process / phosphotransferase activity, for other substituted phosphate groups / peptidoglycan biosynthetic process / cell wall organization / regulation of cell shape / cell division / identical protein binding / metal ion binding / plasma membrane Similarity search - Function | ||||||
| Biological species |   Aquifex aeolicus VF5 (bacteria) Aquifex aeolicus VF5 (bacteria) | ||||||
| Method | ELECTRON MICROSCOPY / single particle reconstruction / cryo EM / Resolution: 2.88 Å | ||||||
 Authors Authors | Hao, A. / Lee, S.-Y. | ||||||
| Funding support |  United States, 1items United States, 1items
| ||||||
 Citation Citation |  Journal: Nat Commun / Year: 2024 Journal: Nat Commun / Year: 2024Title: Development of a natural product optimization strategy for inhibitors against MraY, a promising antibacterial target. Authors: Kazuki Yamamoto / Toyotaka Sato / Aili Hao / Kenta Asao / Rintaro Kaguchi / Shintaro Kusaka / Radhakrishnam Raju Ruddarraju / Daichi Kazamori / Kiki Seo / Satoshi Takahashi / Motohiro ...Authors: Kazuki Yamamoto / Toyotaka Sato / Aili Hao / Kenta Asao / Rintaro Kaguchi / Shintaro Kusaka / Radhakrishnam Raju Ruddarraju / Daichi Kazamori / Kiki Seo / Satoshi Takahashi / Motohiro Horiuchi / Shin-Ichi Yokota / Seok-Yong Lee / Satoshi Ichikawa /   Abstract: MraY (phospho-N-acetylmuramoyl-pentapeptide-transferase) inhibitory natural products are attractive molecules as candidates for a new class of antibacterial agents to combat antimicrobial-resistant ...MraY (phospho-N-acetylmuramoyl-pentapeptide-transferase) inhibitory natural products are attractive molecules as candidates for a new class of antibacterial agents to combat antimicrobial-resistant bacteria. Structural optimization of these natural products is required to improve their drug-like properties for therapeutic use. However, chemical modifications of these natural products are painstaking tasks due to complex synthetic processes, which is a bottleneck in advancing natural products to the clinic. Here, we develop a strategy for a comprehensive in situ evaluation of the build-up library, which enables us to streamline the preparation of the analogue library and directly assess its biological activities. We apply this approach to a series of MraY inhibitory natural products. Through construction and evaluation of the 686-compound library, we identify promising analogues that exhibit potent and broad-spectrum antibacterial activity against highly drug-resistant strains in vitro as well as in vivo in an acute thigh infection model. Structures of the MraY-analogue complexes reveal distinct interaction patterns, suggesting that these analogues represent MraY inhibitors with unique binding modes. We further demonstrate the generality of our strategy by applying it to tubulin-binding natural products to modulate their tubulin polymerization activities. | ||||||
| History |
|
- Structure visualization
Structure visualization
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  9b70.cif.gz 9b70.cif.gz | 346.9 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb9b70.ent.gz pdb9b70.ent.gz | Display |  PDB format PDB format | |
| PDBx/mmJSON format |  9b70.json.gz 9b70.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Summary document |  9b70_validation.pdf.gz 9b70_validation.pdf.gz | 1.2 MB | Display |  wwPDB validaton report wwPDB validaton report |
|---|---|---|---|---|
| Full document |  9b70_full_validation.pdf.gz 9b70_full_validation.pdf.gz | 1.2 MB | Display | |
| Data in XML |  9b70_validation.xml.gz 9b70_validation.xml.gz | 35.7 KB | Display | |
| Data in CIF |  9b70_validation.cif.gz 9b70_validation.cif.gz | 51.9 KB | Display | |
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/b7/9b70 https://data.pdbj.org/pub/pdb/validation_reports/b7/9b70 ftp://data.pdbj.org/pub/pdb/validation_reports/b7/9b70 ftp://data.pdbj.org/pub/pdb/validation_reports/b7/9b70 | HTTPS FTP |
-Related structure data
| Related structure data | 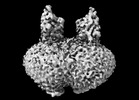 44293MC  9b71C M: map data used to model this data C: citing same article ( |
|---|---|
| Similar structure data | Similarity search - Function & homology  F&H Search F&H Search |
- Links
Links
- Assembly
Assembly
| Deposited unit | 
|
|---|---|
| 1 |
|
- Components
Components
| #1: Protein | Mass: 40954.684 Da / Num. of mol.: 2 Source method: isolated from a genetically manipulated source Source: (gene. exp.)   Aquifex aeolicus VF5 (bacteria) / Gene: mraY, aq_053 / Production host: Aquifex aeolicus VF5 (bacteria) / Gene: mraY, aq_053 / Production host:  References: UniProt: O66465, phospho-N-acetylmuramoyl-pentapeptide-transferase #2: Antibody | Mass: 15057.651 Da / Num. of mol.: 2 Source method: isolated from a genetically manipulated source Source: (gene. exp.)   #3: Chemical | Mass: 934.127 Da / Num. of mol.: 2 / Source method: obtained synthetically / Formula: C46H75N7O13 / Feature type: SUBJECT OF INVESTIGATION #4: Water | ChemComp-HOH / | Has ligand of interest | Y | |
|---|
-Experimental details
-Experiment
| Experiment | Method: ELECTRON MICROSCOPY |
|---|---|
| EM experiment | Aggregation state: PARTICLE / 3D reconstruction method: single particle reconstruction |
- Sample preparation
Sample preparation
| Component | Name: Complex of MraY AA dimer with nanobody bound / Type: COMPLEX / Entity ID: #1-#2 / Source: MULTIPLE SOURCES | |||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Molecular weight | Value: 0.2 MDa / Experimental value: NO | |||||||||||||||||||||||||
| Source (natural) | Organism:   Aquifex aeolicus VF5 (bacteria) Aquifex aeolicus VF5 (bacteria) | |||||||||||||||||||||||||
| Source (recombinant) | Organism:  | |||||||||||||||||||||||||
| Buffer solution | pH: 8 / Details: 20mM Tris-HCl, 150mM NaCl, 2mM DTT, 5mM DM | |||||||||||||||||||||||||
| Buffer component |
| |||||||||||||||||||||||||
| Specimen | Conc.: 7.7 mg/ml / Embedding applied: NO / Shadowing applied: NO / Staining applied: NO / Vitrification applied: YES | |||||||||||||||||||||||||
| Specimen support | Grid material: GOLD / Grid mesh size: 300 divisions/in. / Grid type: UltrAuFoil R1.2/1.3 | |||||||||||||||||||||||||
| Vitrification | Instrument: LEICA EM GP / Cryogen name: ETHANE / Humidity: 90 % / Chamber temperature: 4 K |
- Electron microscopy imaging
Electron microscopy imaging
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
|---|---|
| Microscopy | Model: FEI TITAN KRIOS |
| Electron gun | Electron source:  FIELD EMISSION GUN / Accelerating voltage: 300 kV / Illumination mode: FLOOD BEAM FIELD EMISSION GUN / Accelerating voltage: 300 kV / Illumination mode: FLOOD BEAM |
| Electron lens | Mode: BRIGHT FIELD / Nominal magnification: 81000 X / Nominal defocus max: 2000 nm / Nominal defocus min: 800 nm / Alignment procedure: COMA FREE |
| Specimen holder | Cryogen: NITROGEN / Specimen holder model: FEI TITAN KRIOS AUTOGRID HOLDER |
| Image recording | Average exposure time: 4.6 sec. / Electron dose: 60 e/Å2 / Film or detector model: GATAN K3 BIOQUANTUM (6k x 4k) / Num. of grids imaged: 1 / Num. of real images: 2299 |
| EM imaging optics | Energyfilter name: GIF Bioquantum / Energyfilter slit width: 20 eV |
| Image scans | Width: 5760 / Height: 4092 |
- Processing
Processing
| EM software |
| ||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| CTF correction | Type: PHASE FLIPPING AND AMPLITUDE CORRECTION | ||||||||||||||||||||||||
| 3D reconstruction | Resolution: 2.88 Å / Resolution method: FSC 0.143 CUT-OFF / Num. of particles: 277701 / Num. of class averages: 1 / Symmetry type: POINT | ||||||||||||||||||||||||
| Atomic model building | B value: 30 | ||||||||||||||||||||||||
| Atomic model building | PDB-ID: 5CKR Accession code: 5CKR / Source name: PDB / Type: experimental model | ||||||||||||||||||||||||
| Refine LS restraints |
|
 Movie
Movie Controller
Controller



 PDBj
PDBj



