[English] 日本語
 Yorodumi
Yorodumi- PDB-8avi: Crystal structure of IsdG from Bacillus cereus in complex with heme -
+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 8avi | ||||||
|---|---|---|---|---|---|---|---|
| Title | Crystal structure of IsdG from Bacillus cereus in complex with heme | ||||||
 Components Components | Heme-degrading monooxygenase | ||||||
 Keywords Keywords | OXIDOREDUCTASE / HEME DEGRADATION / MONOOXYGENASE | ||||||
| Function / homology |  Function and homology information Function and homology informationheme oxygenase (biliverdin-producing) / iron import into cell / heme oxygenase (decyclizing) activity / heme catabolic process / iron ion binding / heme binding / cytoplasm Similarity search - Function | ||||||
| Biological species |  | ||||||
| Method |  X-RAY DIFFRACTION / X-RAY DIFFRACTION /  SYNCHROTRON / SYNCHROTRON /  MOLECULAR REPLACEMENT / Resolution: 2 Å MOLECULAR REPLACEMENT / Resolution: 2 Å | ||||||
 Authors Authors | Andersen, H.K. / Hammerstad, M. / Hersleth, H.-P. | ||||||
| Funding support |  Norway, 1items Norway, 1items
| ||||||
 Citation Citation |  Journal: Antioxidants / Year: 2023 Journal: Antioxidants / Year: 2023Title: Functional Diversity of Homologous Oxidoreductases-Tuning of Substrate Specificity by a FAD-Stacking Residue for Iron Acquisition and Flavodoxin Reduction. Authors: Hammerstad, M. / Rugtveit, A.K. / Dahlen, S. / Andersen, H.K. / Hersleth, H.P. | ||||||
| History |
|
- Structure visualization
Structure visualization
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  8avi.cif.gz 8avi.cif.gz | 72.9 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb8avi.ent.gz pdb8avi.ent.gz | 43.6 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  8avi.json.gz 8avi.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/av/8avi https://data.pdbj.org/pub/pdb/validation_reports/av/8avi ftp://data.pdbj.org/pub/pdb/validation_reports/av/8avi ftp://data.pdbj.org/pub/pdb/validation_reports/av/8avi | HTTPS FTP |
|---|
-Related structure data
| Related structure data |  8avhSC  8c16C  8c3mC S: Starting model for refinement C: citing same article ( |
|---|---|
| Similar structure data | Similarity search - Function & homology  F&H Search F&H Search |
- Links
Links
- Assembly
Assembly
| Deposited unit | 
| ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 |
| ||||||||||||
| Unit cell |
|
- Components
Components
-Protein , 1 types, 2 molecules AB
| #1: Protein | Mass: 12049.820 Da / Num. of mol.: 2 Source method: isolated from a genetically manipulated source Source: (gene. exp.)  Strain: ATCC 14579 / DSM 31 / CCUG 7414 / JCM 2152 / NBRC 15305 / NCIMB 9373 / NCTC 2599 / NRRL B-3711 Gene: isdG, BC_4542 / Plasmid: pET22b(+) / Production host:  References: UniProt: Q812Q3, heme oxygenase (staphylobilin-producing) |
|---|
-Non-polymers , 5 types, 47 molecules 
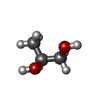
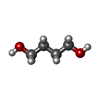
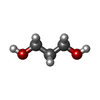





| #2: Chemical | | #3: Chemical | ChemComp-PGO / | #4: Chemical | ChemComp-BU1 / | #5: Chemical | ChemComp-PDO / | #6: Water | ChemComp-HOH / | |
|---|
-Details
| Has ligand of interest | Y |
|---|
-Experimental details
-Experiment
| Experiment | Method:  X-RAY DIFFRACTION / Number of used crystals: 1 X-RAY DIFFRACTION / Number of used crystals: 1 |
|---|
- Sample preparation
Sample preparation
| Crystal | Density Matthews: 2.42 Å3/Da / Density % sol: 49.15 % |
|---|---|
| Crystal grow | Temperature: 298 K / Method: vapor diffusion, sitting drop / pH: 7.5 Details: 18 mg/mL IsdaG (1:1) 12.5% w/v PEG 1000, 12.5% w/v PEG 3350, 12.5% v/v MPD, 0.02 M 1,6-hexanediol, 0.02 M 1-butanol, 0.02 M (RS)-1,2-propanediol, 0.02 M 2-propanol, 0.02 M 1,4-butanediol, 0. ...Details: 18 mg/mL IsdaG (1:1) 12.5% w/v PEG 1000, 12.5% w/v PEG 3350, 12.5% v/v MPD, 0.02 M 1,6-hexanediol, 0.02 M 1-butanol, 0.02 M (RS)-1,2-propanediol, 0.02 M 2-propanol, 0.02 M 1,4-butanediol, 0.02 M 1,3-propanediol, 0.1 M MOPS/HEPES-Na pH 7.5 |
-Data collection
| Diffraction | Mean temperature: 100 K / Serial crystal experiment: N |
|---|---|
| Diffraction source | Source:  SYNCHROTRON / Site: SYNCHROTRON / Site:  MAX IV MAX IV  / Beamline: BioMAX / Wavelength: 0.969998 Å / Beamline: BioMAX / Wavelength: 0.969998 Å |
| Detector | Type: DECTRIS EIGER X 16M / Detector: PIXEL / Date: Mar 31, 2022 |
| Radiation | Protocol: SINGLE WAVELENGTH / Monochromatic (M) / Laue (L): M / Scattering type: x-ray |
| Radiation wavelength | Wavelength: 0.969998 Å / Relative weight: 1 |
| Reflection | Resolution: 2→44.04 Å / Num. obs: 16227 / % possible obs: 99.7 % / Redundancy: 6.5 % / Biso Wilson estimate: 47.69 Å2 / CC1/2: 0.997 / Rmerge(I) obs: 0.065 / Rpim(I) all: 0.028 / Rrim(I) all: 0.071 / Net I/σ(I): 12.2 |
| Reflection shell | Resolution: 2→2.05 Å / Redundancy: 6.7 % / Rmerge(I) obs: 1.213 / Mean I/σ(I) obs: 1.6 / Num. unique obs: 1200 / CC1/2: 0.579 / Rpim(I) all: 0.498 / Rrim(I) all: 1.313 / % possible all: 100 |
- Processing
Processing
| Software |
| |||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Refinement | Method to determine structure:  MOLECULAR REPLACEMENT MOLECULAR REPLACEMENTStarting model: 8AVH Resolution: 2→44.04 Å / SU ML: 0.2365 / Cross valid method: FREE R-VALUE / σ(F): 1.35 / Phase error: 26.5781 Stereochemistry target values: GeoStd + Monomer Library + CDL v1.2
| |||||||||||||||||||||||||||||||||||||||||||||||||
| Solvent computation | Shrinkage radii: 0.9 Å / VDW probe radii: 1.11 Å / Solvent model: FLAT BULK SOLVENT MODEL | |||||||||||||||||||||||||||||||||||||||||||||||||
| Displacement parameters | Biso mean: 61.14 Å2 | |||||||||||||||||||||||||||||||||||||||||||||||||
| Refinement step | Cycle: LAST / Resolution: 2→44.04 Å
| |||||||||||||||||||||||||||||||||||||||||||||||||
| Refine LS restraints |
| |||||||||||||||||||||||||||||||||||||||||||||||||
| LS refinement shell |
|
 Movie
Movie Controller
Controller


 PDBj
PDBj






