+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 7vpx | ||||||
|---|---|---|---|---|---|---|---|
| Title | The cryo-EM structure of the human pre-A complex | ||||||
 Components Components |
| ||||||
 Keywords Keywords | SPLICING / human spliceosome / pre-A complex / PRP5 / A complex / U1 snRNP / U2 snRNP | ||||||
| Function / homology |  Function and homology information Function and homology informationnegative regulation of protein refolding / U2AF complex / U11/U12 snRNP / U2 snRNP binding / U7 snRNA binding / histone pre-mRNA DCP binding / U7 snRNP / histone pre-mRNA 3'end processing complex / SLBP independent Processing of Histone Pre-mRNAs / SLBP Dependent Processing of Replication-Dependent Histone Pre-mRNAs ...negative regulation of protein refolding / U2AF complex / U11/U12 snRNP / U2 snRNP binding / U7 snRNA binding / histone pre-mRNA DCP binding / U7 snRNP / histone pre-mRNA 3'end processing complex / SLBP independent Processing of Histone Pre-mRNAs / SLBP Dependent Processing of Replication-Dependent Histone Pre-mRNAs / B-WICH complex / protein methylation / 7-methylguanosine cap hypermethylation / U12-type spliceosomal complex / U1 snRNP binding / methylosome / pICln-Sm protein complex / positive regulation of mRNA splicing, via spliceosome / RNA splicing, via transesterification reactions / small nuclear ribonucleoprotein complex / SMN-Sm protein complex / spliceosomal tri-snRNP complex / splicing factor binding / P granule / snRNP binding / commitment complex / U2-type precatalytic spliceosome / mRNA cis splicing, via spliceosome / telomerase holoenzyme complex / telomerase RNA binding / U2-type catalytic step 2 spliceosome / U2-type prespliceosome assembly / U2-type spliceosomal complex / U1 snRNP / SAGA complex / RNA Polymerase II Transcription Termination / U2 snRNP / U4 snRNP / pre-mRNA 5'-splice site binding / U2-type prespliceosome / negative regulation of chaperone-mediated autophagy / positive regulation of transcription by RNA polymerase III / precatalytic spliceosome / mRNA 5'-splice site recognition / mRNA 3'-splice site recognition / regulation of RNA splicing / mRNA Splicing - Minor Pathway / positive regulation of transcription by RNA polymerase I / spliceosomal complex assembly / U5 snRNP / spliceosomal snRNP assembly / U2 snRNA binding / cellular response to transforming growth factor beta stimulus / regulation of DNA repair / Cajal body / U1 snRNA binding / U4/U6 x U5 tri-snRNP complex / intercellular bridge / cellular response to retinoic acid / catalytic step 2 spliceosome / mRNA Splicing - Major Pathway / lysosomal lumen / Gene and protein expression by JAK-STAT signaling after Interleukin-12 stimulation / Hsp70 protein binding / RNA splicing / stem cell differentiation / spliceosomal complex / negative regulation of smooth muscle cell proliferation / mRNA splicing, via spliceosome / positive regulation of neuron projection development / negative regulation of protein catabolic process / B-WICH complex positively regulates rRNA expression / cellular response to tumor necrosis factor / fibrillar center / mRNA processing / nuclear matrix / cytoplasmic ribonucleoprotein granule / transcription corepressor activity / snRNP Assembly / spermatogenesis / SARS-CoV-2 modulates host translation machinery / RNA helicase activity / single-stranded RNA binding / nuclear speck / nuclear body / RNA helicase / ribosome / chromatin remodeling / mRNA binding / positive regulation of DNA-templated transcription / protein-containing complex binding / nucleolus / enzyme binding / protein homodimerization activity / ATP hydrolysis activity / positive regulation of transcription by RNA polymerase II / DNA binding / RNA binding / extracellular exosome / zinc ion binding Similarity search - Function | ||||||
| Biological species |  Homo sapiens (human) Homo sapiens (human) unidentified adenovirus unidentified adenovirus | ||||||
| Method | ELECTRON MICROSCOPY / single particle reconstruction / cryo EM / Resolution: 3 Å | ||||||
 Authors Authors | Zhang, X. / Zhan, X. / Shi, Y. | ||||||
| Funding support |  China, 1items China, 1items
| ||||||
 Citation Citation |  Journal: Nat Struct Mol Biol / Year: 2024 Journal: Nat Struct Mol Biol / Year: 2024Title: Structural insights into branch site proofreading by human spliceosome. Authors: Xiaofeng Zhang / Xiechao Zhan / Tong Bian / Fenghua Yang / Pan Li / Yichen Lu / Zhihan Xing / Rongyan Fan / Qiangfeng Cliff Zhang / Yigong Shi /  Abstract: Selection of the pre-mRNA branch site (BS) by the U2 small nuclear ribonucleoprotein (snRNP) is crucial to prespliceosome (A complex) assembly. The RNA helicase PRP5 proofreads BS selection but the ...Selection of the pre-mRNA branch site (BS) by the U2 small nuclear ribonucleoprotein (snRNP) is crucial to prespliceosome (A complex) assembly. The RNA helicase PRP5 proofreads BS selection but the underlying mechanism remains unclear. Here we report the atomic structures of two sequential complexes leading to prespliceosome assembly: human 17S U2 snRNP and a cross-exon pre-A complex. PRP5 is anchored on 17S U2 snRNP mainly through occupation of the RNA path of SF3B1 by an acidic loop of PRP5; the helicase domain of PRP5 associates with U2 snRNA; the BS-interacting stem-loop (BSL) of U2 snRNA is shielded by TAT-SF1, unable to engage the BS. In the pre-A complex, an initial U2-BS duplex is formed; the translocated helicase domain of PRP5 stays with U2 snRNA and the acidic loop still occupies the RNA path. The pre-A conformation is specifically stabilized by the splicing factors SF1, DNAJC8 and SF3A2. Cancer-derived mutations in SF3B1 damage its association with PRP5, compromising BS proofreading. Together, these findings reveal key insights into prespliceosome assembly and BS selection or proofreading by PRP5. | ||||||
| History |
|
- Structure visualization
Structure visualization
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  7vpx.cif.gz 7vpx.cif.gz | 1.2 MB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb7vpx.ent.gz pdb7vpx.ent.gz | 848.3 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  7vpx.json.gz 7vpx.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/vp/7vpx https://data.pdbj.org/pub/pdb/validation_reports/vp/7vpx ftp://data.pdbj.org/pub/pdb/validation_reports/vp/7vpx ftp://data.pdbj.org/pub/pdb/validation_reports/vp/7vpx | HTTPS FTP |
|---|
-Related structure data
| Related structure data |  32074MC  7evoC M: map data used to model this data C: citing same article ( |
|---|---|
| Similar structure data | Similarity search - Function & homology  F&H Search F&H Search |
- Links
Links
- Assembly
Assembly
| Deposited unit | 
|
|---|---|
| 1 |
|
- Components
Components
-Splicing factor 3A subunit ... , 3 types, 3 molecules BAC
| #1: Protein | Mass: 49327.355 Da / Num. of mol.: 1 / Source method: isolated from a natural source / Source: (natural)  Homo sapiens (human) / References: UniProt: Q15428 Homo sapiens (human) / References: UniProt: Q15428 |
|---|---|
| #10: Protein | Mass: 88991.094 Da / Num. of mol.: 1 / Source method: isolated from a natural source / Source: (natural)  Homo sapiens (human) / References: UniProt: Q15459 Homo sapiens (human) / References: UniProt: Q15459 |
| #11: Protein | Mass: 58934.844 Da / Num. of mol.: 1 / Source method: isolated from a natural source / Source: (natural)  Homo sapiens (human) / References: UniProt: Q12874 Homo sapiens (human) / References: UniProt: Q12874 |
-Protein , 5 types, 6 molecules DJEfk6
| #2: Protein | Mass: 68401.742 Da / Num. of mol.: 1 / Source method: isolated from a natural source / Source: (natural)  Homo sapiens (human) / References: UniProt: Q15637 Homo sapiens (human) / References: UniProt: Q15637 | ||
|---|---|---|---|
| #3: Protein | Mass: 29900.764 Da / Num. of mol.: 1 / Source method: isolated from a natural source / Source: (natural)  Homo sapiens (human) / References: UniProt: O75937 Homo sapiens (human) / References: UniProt: O75937 | ||
| #12: Protein | Mass: 117576.484 Da / Num. of mol.: 1 / Source method: isolated from a natural source / Source: (natural)  Homo sapiens (human) / References: UniProt: Q7L014, RNA helicase Homo sapiens (human) / References: UniProt: Q7L014, RNA helicase | ||
| #20: Protein | Mass: 24642.131 Da / Num. of mol.: 2 / Source method: isolated from a natural source / Source: (natural)  Homo sapiens (human) / References: UniProt: P14678 Homo sapiens (human) / References: UniProt: P14678#28: Protein | | Mass: 12427.524 Da / Num. of mol.: 1 / Source method: isolated from a natural source / Source: (natural)  Homo sapiens (human) / References: UniProt: Q7RTV0 Homo sapiens (human) / References: UniProt: Q7RTV0 |
-RNA chain , 4 types, 4 molecules HIKL
| #4: RNA chain | Mass: 60186.445 Da / Num. of mol.: 1 / Source method: isolated from a natural source / Source: (natural)  Homo sapiens (human) / References: GenBank: 340097 Homo sapiens (human) / References: GenBank: 340097 |
|---|---|
| #22: RNA chain | Mass: 83089.961 Da / Num. of mol.: 1 / Source method: obtained synthetically / Source: (synth.)  unidentified adenovirus unidentified adenovirus |
| #23: RNA chain | Mass: 3256.996 Da / Num. of mol.: 1 / Source method: obtained synthetically / Source: (synth.)  unidentified adenovirus unidentified adenovirus |
| #24: RNA chain | Mass: 52697.070 Da / Num. of mol.: 1 / Source method: isolated from a natural source / Source: (natural)  Homo sapiens (human) / References: GenBank: 1781322691 Homo sapiens (human) / References: GenBank: 1781322691 |
-Splicing factor 3B subunit ... , 5 types, 5 molecules 12345
| #5: Protein | Mass: 146024.938 Da / Num. of mol.: 1 / Source method: isolated from a natural source / Source: (natural)  Homo sapiens (human) / References: UniProt: O75533 Homo sapiens (human) / References: UniProt: O75533 |
|---|---|
| #6: Protein | Mass: 100377.812 Da / Num. of mol.: 1 / Source method: isolated from a natural source / Source: (natural)  Homo sapiens (human) / References: UniProt: Q13435 Homo sapiens (human) / References: UniProt: Q13435 |
| #7: Protein | Mass: 135718.844 Da / Num. of mol.: 1 / Source method: isolated from a natural source / Source: (natural)  Homo sapiens (human) / References: UniProt: Q15393 Homo sapiens (human) / References: UniProt: Q15393 |
| #8: Protein | Mass: 44436.570 Da / Num. of mol.: 1 / Source method: isolated from a natural source / Source: (natural)  Homo sapiens (human) / References: UniProt: Q15427 Homo sapiens (human) / References: UniProt: Q15427 |
| #9: Protein | Mass: 10149.369 Da / Num. of mol.: 1 / Source method: isolated from a natural source / Source: (natural)  Homo sapiens (human) / References: UniProt: Q9BWJ5 Homo sapiens (human) / References: UniProt: Q9BWJ5 |
-U2 small nuclear ribonucleoprotein ... , 2 types, 2 molecules FG
| #13: Protein | Mass: 28456.584 Da / Num. of mol.: 1 / Source method: isolated from a natural source / Source: (natural)  Homo sapiens (human) / References: UniProt: P09661 Homo sapiens (human) / References: UniProt: P09661 |
|---|---|
| #14: Protein | Mass: 25524.367 Da / Num. of mol.: 1 / Source method: isolated from a natural source / Source: (natural)  Homo sapiens (human) / References: UniProt: P08579 Homo sapiens (human) / References: UniProt: P08579 |
-Small nuclear ribonucleoprotein ... , 6 types, 12 molecules aibmcldnejgh
| #15: Protein | Mass: 13551.928 Da / Num. of mol.: 2 / Source method: isolated from a natural source / Source: (natural)  Homo sapiens (human) / References: UniProt: P62316 Homo sapiens (human) / References: UniProt: P62316#16: Protein | Mass: 9734.171 Da / Num. of mol.: 2 / Source method: isolated from a natural source / Source: (natural)  Homo sapiens (human) / References: UniProt: P62306 Homo sapiens (human) / References: UniProt: P62306#17: Protein | Mass: 10817.601 Da / Num. of mol.: 2 / Source method: isolated from a natural source / Source: (natural)  Homo sapiens (human) / References: UniProt: P62304 Homo sapiens (human) / References: UniProt: P62304#18: Protein | Mass: 8508.084 Da / Num. of mol.: 2 / Source method: isolated from a natural source / Source: (natural)  Homo sapiens (human) / References: UniProt: P62308 Homo sapiens (human) / References: UniProt: P62308#19: Protein | Mass: 13940.308 Da / Num. of mol.: 2 / Source method: isolated from a natural source / Source: (natural)  Homo sapiens (human) / References: UniProt: P62318 Homo sapiens (human) / References: UniProt: P62318#21: Protein | Mass: 13310.653 Da / Num. of mol.: 2 / Source method: isolated from a natural source / Source: (natural)  Homo sapiens (human) / References: UniProt: P62314 Homo sapiens (human) / References: UniProt: P62314 |
|---|
-U1 small nuclear ribonucleoprotein ... , 3 types, 3 molecules OMN
| #25: Protein | Mass: 51683.184 Da / Num. of mol.: 1 / Source method: isolated from a natural source / Source: (natural)  Homo sapiens (human) / References: UniProt: P08621 Homo sapiens (human) / References: UniProt: P08621 |
|---|---|
| #26: Protein | Mass: 31319.533 Da / Num. of mol.: 1 / Source method: isolated from a natural source / Source: (natural)  Homo sapiens (human) / References: UniProt: P09012 Homo sapiens (human) / References: UniProt: P09012 |
| #27: Protein | Mass: 17415.727 Da / Num. of mol.: 1 / Source method: isolated from a natural source / Source: (natural)  Homo sapiens (human) / References: UniProt: P09234 Homo sapiens (human) / References: UniProt: P09234 |
-Non-polymers , 2 types, 7 molecules 
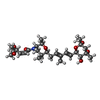

| #29: Chemical | ChemComp-ZN / #30: Chemical | ChemComp-SJT / | |
|---|
-Details
| Has ligand of interest | Y |
|---|
-Experimental details
-Experiment
| Experiment | Method: ELECTRON MICROSCOPY |
|---|---|
| EM experiment | Aggregation state: PARTICLE / 3D reconstruction method: single particle reconstruction |
- Sample preparation
Sample preparation
| Component |
| ||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Source (natural) |
| ||||||||||||||||||||||||
| Buffer solution | pH: 7.9 | ||||||||||||||||||||||||
| Specimen | Embedding applied: NO / Shadowing applied: NO / Staining applied: NO / Vitrification applied: YES | ||||||||||||||||||||||||
| Vitrification | Cryogen name: ETHANE |
- Electron microscopy imaging
Electron microscopy imaging
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
|---|---|
| Microscopy | Model: FEI TITAN KRIOS |
| Electron gun | Electron source:  FIELD EMISSION GUN / Accelerating voltage: 300 kV / Illumination mode: FLOOD BEAM FIELD EMISSION GUN / Accelerating voltage: 300 kV / Illumination mode: FLOOD BEAM |
| Electron lens | Mode: BRIGHT FIELD / Nominal defocus max: 2000 nm / Nominal defocus min: 1500 nm |
| Image recording | Electron dose: 50 e/Å2 / Film or detector model: GATAN K3 (6k x 4k) |
- Processing
Processing
| CTF correction | Type: NONE |
|---|---|
| 3D reconstruction | Resolution: 3 Å / Resolution method: FSC 0.143 CUT-OFF / Num. of particles: 419522 / Symmetry type: POINT |
| Refinement | Highest resolution: 3 Å |
 Movie
Movie Controller
Controller






 PDBj
PDBj













































