+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 7pq8 | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
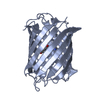
| Title | Crystal structure of Campylobacter jejuni DsbA1 | |||||||||
 Components Components | Thiol:disulfide interchange protein DsbA | |||||||||
 Keywords Keywords | OXIDOREDUCTASE / disulfide bond / thioredoxin fold / periplasmic / posttranslational modification | |||||||||
| Function / homology | Thiol:disulphide interchange protein DsbA/DsbL / : / DSBA-like thioredoxin domain / DSBA-like thioredoxin domain / isomerase activity / Thioredoxin-like superfamily / oxidoreductase activity / Thiol:disulfide interchange protein DsbA Function and homology information Function and homology information | |||||||||
| Biological species |  | |||||||||
| Method |  X-RAY DIFFRACTION / X-RAY DIFFRACTION /  SYNCHROTRON / SYNCHROTRON /  MOLECULAR REPLACEMENT / Resolution: 1.329 Å MOLECULAR REPLACEMENT / Resolution: 1.329 Å | |||||||||
 Authors Authors | Orlikowska, M. / Bocian-Ostrzycka, K.M. / Banas, A.M. / Jagusztyn-Krynicka, E.K. | |||||||||
| Funding support |  Poland, 2items Poland, 2items
| |||||||||
 Citation Citation |  Journal: Int J Mol Sci / Year: 2021 Journal: Int J Mol Sci / Year: 2021Title: Interplay between DsbA1, DsbA2 and C8J_1298 Periplasmic Oxidoreductases of Campylobacter jejuni and Their Impact on Bacterial Physiology and Pathogenesis. Authors: Banas, A.M. / Bocian-Ostrzycka, K.M. / Dunin-Horkawicz, S. / Ludwiczak, J. / Wilk, P. / Orlikowska, M. / Wyszynska, A. / Dabrowska, M. / Plichta, M. / Spodzieja, M. / Polanska, M.A. / ...Authors: Banas, A.M. / Bocian-Ostrzycka, K.M. / Dunin-Horkawicz, S. / Ludwiczak, J. / Wilk, P. / Orlikowska, M. / Wyszynska, A. / Dabrowska, M. / Plichta, M. / Spodzieja, M. / Polanska, M.A. / Malinowska, A. / Jagusztyn-Krynicka, E.K. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  7pq8.cif.gz 7pq8.cif.gz | 98.7 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb7pq8.ent.gz pdb7pq8.ent.gz | 71.5 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  7pq8.json.gz 7pq8.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/pq/7pq8 https://data.pdbj.org/pub/pdb/validation_reports/pq/7pq8 ftp://data.pdbj.org/pub/pdb/validation_reports/pq/7pq8 ftp://data.pdbj.org/pub/pdb/validation_reports/pq/7pq8 | HTTPS FTP |
|---|
-Related structure data
| Related structure data |  7pq7SC  7pqfC S: Starting model for refinement C: citing same article ( |
|---|---|
| Similar structure data |
- Links
Links
- Assembly
Assembly
| Deposited unit | 
| ||||||||
|---|---|---|---|---|---|---|---|---|---|
| 1 |
| ||||||||
| Unit cell |
|
- Components
Components
| #1: Protein | Mass: 23396.359 Da / Num. of mol.: 1 Source method: isolated from a genetically manipulated source Source: (gene. exp.)  Gene: B5Y32_04750, B7Q70_04295, BCB47_05875, D4Q41_05880, DPG08_09245, DUY05_07510, DYE84_06245, EAX31_06480, F0166_06130, F0J04_04905, F6982_05945, F7521_05270, F7J47_05180, F8803_06690, F9778_ ...Gene: B5Y32_04750, B7Q70_04295, BCB47_05875, D4Q41_05880, DPG08_09245, DUY05_07510, DYE84_06245, EAX31_06480, F0166_06130, F0J04_04905, F6982_05945, F7521_05270, F7J47_05180, F8803_06690, F9778_05085, FVY60_01390, FW488_04080, FZ445_05615, FZ732_06740, G3M94_001567, GFF90_03350, GMG47_05310, GNO00_06010, GXA88_04885, GXS76_000973, GZ489_001237, GZ502_000952 Production host:  |
|---|---|
| #2: Chemical | ChemComp-PG4 / |
| #3: Water | ChemComp-HOH / |
| Has ligand of interest | Y |
-Experimental details
-Experiment
| Experiment | Method:  X-RAY DIFFRACTION / Number of used crystals: 1 X-RAY DIFFRACTION / Number of used crystals: 1 |
|---|
- Sample preparation
Sample preparation
| Crystal | Density Matthews: 2.12 Å3/Da / Density % sol: 41.99 % |
|---|---|
| Crystal grow | Temperature: 291 K / Method: vapor diffusion, hanging drop / pH: 8.5 Details: 0.12 M Ethylene glycols, 0.1M Tris/BICINE pH 8.5, 50 % v/v Precipitant Mix 1 |
-Data collection
| Diffraction | Mean temperature: 100 K / Serial crystal experiment: N |
|---|---|
| Diffraction source | Source:  SYNCHROTRON / Site: SYNCHROTRON / Site:  BESSY BESSY  / Beamline: 14.1 / Wavelength: 0.9184 Å / Beamline: 14.1 / Wavelength: 0.9184 Å |
| Detector | Type: DECTRIS PILATUS3 6M / Detector: PIXEL / Date: Jun 24, 2018 |
| Radiation | Protocol: SINGLE WAVELENGTH / Monochromatic (M) / Laue (L): M / Scattering type: x-ray |
| Radiation wavelength | Wavelength: 0.9184 Å / Relative weight: 1 |
| Reflection | Resolution: 1.329→48.86 Å / Num. obs: 51341 / % possible obs: 97.8 % / Redundancy: 3.78 % / CC1/2: 0.99 / Net I/σ(I): 8.94 |
| Reflection shell | Resolution: 1.33→1.41 Å / Num. unique obs: 12965 / CC1/2: 0.65 |
- Processing
Processing
| Software |
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Refinement | Method to determine structure:  MOLECULAR REPLACEMENT MOLECULAR REPLACEMENTStarting model: 7PQ7 Resolution: 1.329→46.889 Å / Cor.coef. Fo:Fc: 0.963 / Cor.coef. Fo:Fc free: 0.947 / SU B: 2.402 / SU ML: 0.045 / Cross valid method: FREE R-VALUE / ESU R: 0.056 / ESU R Free: 0.063 Details: Hydrogens have been added in their riding positions
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Solvent computation | Ion probe radii: 0.8 Å / Shrinkage radii: 0.8 Å / VDW probe radii: 1.2 Å / Solvent model: MASK BULK SOLVENT | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Displacement parameters | Biso mean: 14.025 Å2
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refinement step | Cycle: LAST / Resolution: 1.329→46.889 Å
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refine LS restraints |
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| LS refinement shell | Refine-ID: X-RAY DIFFRACTION / Total num. of bins used: 20
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refinement TLS params. | Method: refined / Refine-ID: X-RAY DIFFRACTION
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refinement TLS group |
|
 Movie
Movie Controller
Controller












 PDBj
PDBj


