[English] 日本語
 Yorodumi
Yorodumi- PDB-7d7m: Cryo-EM Structure of the Prostaglandin E Receptor EP4 Coupled to ... -
+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 7d7m | ||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Title | Cryo-EM Structure of the Prostaglandin E Receptor EP4 Coupled to G Protein | ||||||||||||||||||
 Components Components |
| ||||||||||||||||||
 Keywords Keywords | MEMBRANE PROTEIN / prostaglandin E receptor / EP4 / GPCR / G protein | ||||||||||||||||||
| Function / homology |  Function and homology information Function and homology informationnegative regulation of eosinophil extravasation / prostaglandin E receptor activity / negative regulation of integrin activation / Prostanoid ligand receptors / T-helper cell differentiation / regulation of stress fiber assembly / negative regulation of cytokine production / adenylate cyclase-activating G protein-coupled bile acid receptor signaling pathway / adenylate cyclase-activating serotonin receptor signaling pathway / regulation of skeletal muscle contraction ...negative regulation of eosinophil extravasation / prostaglandin E receptor activity / negative regulation of integrin activation / Prostanoid ligand receptors / T-helper cell differentiation / regulation of stress fiber assembly / negative regulation of cytokine production / adenylate cyclase-activating G protein-coupled bile acid receptor signaling pathway / adenylate cyclase-activating serotonin receptor signaling pathway / regulation of skeletal muscle contraction / PKA activation in glucagon signalling / hair follicle placode formation / developmental growth / intracellular transport / D1 dopamine receptor binding / response to mechanical stimulus / vascular endothelial cell response to laminar fluid shear stress / renal water homeostasis / activation of adenylate cyclase activity / Hedgehog 'off' state / cellular response to acidic pH / adenylate cyclase-activating adrenergic receptor signaling pathway / JNK cascade / ERK1 and ERK2 cascade / cellular response to glucagon stimulus / intracellular glucose homeostasis / positive regulation of insulin secretion involved in cellular response to glucose stimulus / positive regulation of cytokine production / adenylate cyclase activator activity / trans-Golgi network membrane / negative regulation of inflammatory response to antigenic stimulus / response to prostaglandin E / cellular response to mechanical stimulus / bone development / platelet aggregation / negative regulation of inflammatory response / cognition / G-protein beta/gamma-subunit complex binding / adenylate cyclase-modulating G protein-coupled receptor signaling pathway / adenylate cyclase-inhibiting G protein-coupled receptor signaling pathway / Olfactory Signaling Pathway / Activation of the phototransduction cascade / positive regulation of insulin secretion / G protein-coupled acetylcholine receptor signaling pathway / positive regulation of inflammatory response / G beta:gamma signalling through PLC beta / Presynaptic function of Kainate receptors / Thromboxane signalling through TP receptor / Activation of G protein gated Potassium channels / Inhibition of voltage gated Ca2+ channels via Gbeta/gamma subunits / G-protein activation / Glucagon signaling in metabolic regulation / G beta:gamma signalling through CDC42 / Prostacyclin signalling through prostacyclin receptor / sensory perception of smell / Synthesis, secretion, and inactivation of Glucagon-like Peptide-1 (GLP-1) / G beta:gamma signalling through BTK / photoreceptor disc membrane / ADP signalling through P2Y purinoceptor 12 / Sensory perception of sweet, bitter, and umami (glutamate) taste / Glucagon-type ligand receptors / Adrenaline,noradrenaline inhibits insulin secretion / Vasopressin regulates renal water homeostasis via Aquaporins / Glucagon-like Peptide-1 (GLP1) regulates insulin secretion / G alpha (z) signalling events / cellular response to catecholamine stimulus / ADP signalling through P2Y purinoceptor 1 / ADORA2B mediated anti-inflammatory cytokines production / G beta:gamma signalling through PI3Kgamma / adenylate cyclase-activating dopamine receptor signaling pathway / Cooperation of PDCL (PhLP1) and TRiC/CCT in G-protein beta folding / GPER1 signaling / cellular response to prostaglandin E stimulus / positive regulation of cold-induced thermogenesis / heterotrimeric G-protein complex / G alpha (12/13) signalling events / Inactivation, recovery and regulation of the phototransduction cascade / G-protein beta-subunit binding / extracellular vesicle / sensory perception of taste / Thrombin signalling through proteinase activated receptors (PARs) / signaling receptor complex adaptor activity / adenylate cyclase-activating G protein-coupled receptor signaling pathway / positive regulation of cytosolic calcium ion concentration / retina development in camera-type eye / GTPase binding / G protein activity / fibroblast proliferation / Ca2+ pathway / High laminar flow shear stress activates signaling by PIEZO1 and PECAM1:CDH5:KDR in endothelial cells / G alpha (i) signalling events / G alpha (s) signalling events / phospholipase C-activating G protein-coupled receptor signaling pathway / G alpha (q) signalling events / Hydrolases; Acting on acid anhydrides; Acting on GTP to facilitate cellular and subcellular movement / Ras protein signal transduction / Extra-nuclear estrogen signaling / cell population proliferation / immune response / G protein-coupled receptor signaling pathway Similarity search - Function | ||||||||||||||||||
| Biological species |  Homo sapiens (human) Homo sapiens (human) | ||||||||||||||||||
| Method | ELECTRON MICROSCOPY / single particle reconstruction / cryo EM / Resolution: 3.3 Å | ||||||||||||||||||
 Authors Authors | Nojima, S. / Fujita, Y. / Kimura, T.K. / Nomura, N. / Suno, R. / Morimoto, K. / Yamamoto, M. / Noda, T. / Iwata, S. / Shigematsu, H. / Kobayashi, T. | ||||||||||||||||||
| Funding support |  Japan, 5items Japan, 5items
| ||||||||||||||||||
 Citation Citation |  Journal: Structure / Year: 2021 Journal: Structure / Year: 2021Title: Cryo-EM Structure of the Prostaglandin E Receptor EP4 Coupled to G Protein. Authors: Shingo Nojima / Yoko Fujita / Kanako Terakado Kimura / Norimichi Nomura / Ryoji Suno / Kazushi Morimoto / Masaki Yamamoto / Takeshi Noda / So Iwata / Hideki Shigematsu / Takuya Kobayashi /  Abstract: Prostaglandin E receptor EP4, a class A G protein-coupled receptor (GPCR), is a common drug target in various disorders, such as acute decompensated heart failure and ulcerative colitis. Here, we ...Prostaglandin E receptor EP4, a class A G protein-coupled receptor (GPCR), is a common drug target in various disorders, such as acute decompensated heart failure and ulcerative colitis. Here, we report the cryoelectron microscopy (cryo-EM) structure of the EP4-heterotrimeric G protein (Gs) complex with the endogenous ligand at a global resolution of 3.3 Å. In this structure, compared with that in the inactive EP4 structure, the sixth transmembrane domain is shifted outward on the intracellular side, although the shift is smaller than that in other class A GPCRs bound to Gs. Instead, the C-terminal helix of Gs is inserted toward TM2 of EP4, and the conserved C-terminal hook structure formsthe extended state. These structural features are formed by the conserved residues in prostanoid receptors (Phe54 and Trp327). These findings may be important for the thorough understanding of the G protein-binding mechanism of EP4 and other prostanoid receptors. | ||||||||||||||||||
| History |
|
- Structure visualization
Structure visualization
| Movie |
 Movie viewer Movie viewer |
|---|---|
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  7d7m.cif.gz 7d7m.cif.gz | 205.1 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb7d7m.ent.gz pdb7d7m.ent.gz | 151.4 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  7d7m.json.gz 7d7m.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/d7/7d7m https://data.pdbj.org/pub/pdb/validation_reports/d7/7d7m ftp://data.pdbj.org/pub/pdb/validation_reports/d7/7d7m ftp://data.pdbj.org/pub/pdb/validation_reports/d7/7d7m | HTTPS FTP |
|---|
-Related structure data
| Related structure data |  30608MC M: map data used to model this data C: citing same article ( |
|---|---|
| Similar structure data |
- Links
Links
- Assembly
Assembly
| Deposited unit | 
|
|---|---|
| 1 |
|
- Components
Components
-Guanine nucleotide-binding protein ... , 3 types, 3 molecules BCD
| #2: Protein | Mass: 37728.152 Da / Num. of mol.: 1 Source method: isolated from a genetically manipulated source Source: (gene. exp.)  Homo sapiens (human) / Gene: GNB1 / Plasmid: pFastBac Dual / Production host: Homo sapiens (human) / Gene: GNB1 / Plasmid: pFastBac Dual / Production host:  |
|---|---|
| #3: Protein | Mass: 7861.143 Da / Num. of mol.: 1 Source method: isolated from a genetically manipulated source Source: (gene. exp.)  Homo sapiens (human) / Gene: GNG2 / Plasmid: pFastBac Dual / Production host: Homo sapiens (human) / Gene: GNG2 / Plasmid: pFastBac Dual / Production host:  |
| #4: Protein | Mass: 29014.750 Da / Num. of mol.: 1 Mutation: 65 to 203, 255 to 264 deletion G49D, E50N, L63Y, A249D, S252D, I372A and V375I Source method: isolated from a genetically manipulated source Details: residues from 65 to 203, residues from 255 to 264 were deleted Source: (gene. exp.)  Homo sapiens (human) / Gene: GNAS, GNAS1, GSP / Plasmid: pET21a / Production host: Homo sapiens (human) / Gene: GNAS, GNAS1, GSP / Plasmid: pET21a / Production host:  |
-Protein / Antibody / Non-polymers , 3 types, 3 molecules AE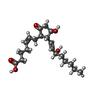

| #1: Protein | Mass: 36953.453 Da / Num. of mol.: 1 / Mutation: N7Q, N177Q, 218-259 deletion Source method: isolated from a genetically manipulated source Details: residues from 218 to 259 were deleted / Source: (gene. exp.)  Homo sapiens (human) / Gene: PTGER4, PTGER2 / Plasmid: pFastBac1 / Production host: Homo sapiens (human) / Gene: PTGER4, PTGER2 / Plasmid: pFastBac1 / Production host:  |
|---|---|
| #5: Antibody | Mass: 14680.293 Da / Num. of mol.: 1 Source method: isolated from a genetically manipulated source Details: The C-terminal "ENLYFQ" of sample sequence is a cleavaged TEV protease recognition sequence. Source: (gene. exp.)   Brevibacillus choshinensis (bacteria) Brevibacillus choshinensis (bacteria) |
| #6: Chemical | ChemComp-P2E / ( |
-Details
| Has ligand of interest | Y |
|---|---|
| Has protein modification | Y |
-Experimental details
-Experiment
| Experiment | Method: ELECTRON MICROSCOPY |
|---|---|
| EM experiment | Aggregation state: 2D ARRAY / 3D reconstruction method: single particle reconstruction |
- Sample preparation
Sample preparation
| Component |
| |||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Molecular weight | Value: 0.12 MDa / Experimental value: YES | |||||||||||||||||||||||||||||||||||
| Source (natural) |
| |||||||||||||||||||||||||||||||||||
| Source (recombinant) |
| |||||||||||||||||||||||||||||||||||
| Buffer solution | pH: 7.5 | |||||||||||||||||||||||||||||||||||
| Buffer component |
| |||||||||||||||||||||||||||||||||||
| Specimen | Conc.: 8.4 mg/ml / Embedding applied: NO / Shadowing applied: NO / Staining applied: NO / Vitrification applied: YES | |||||||||||||||||||||||||||||||||||
| Specimen support | Details: 10 mA / Grid material: COPPER / Grid mesh size: 300 divisions/in. / Grid type: Quantifoil R0.6/1 | |||||||||||||||||||||||||||||||||||
| Vitrification | Instrument: FEI VITROBOT MARK IV / Cryogen name: ETHANE / Humidity: 100 % / Chamber temperature: 281 K / Details: Blot Force 10, Blot Time 3.5 sec, 3 microL apply |
- Electron microscopy imaging
Electron microscopy imaging
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
|---|---|
| Microscopy | Model: FEI TITAN KRIOS |
| Electron gun | Electron source:  FIELD EMISSION GUN / Accelerating voltage: 300 kV / Illumination mode: FLOOD BEAM FIELD EMISSION GUN / Accelerating voltage: 300 kV / Illumination mode: FLOOD BEAM |
| Electron lens | Mode: BRIGHT FIELD / Nominal magnification: 105000 X / Nominal defocus max: 1800 nm / Nominal defocus min: 600 nm / Cs: 2.7 mm / C2 aperture diameter: 50 µm / Alignment procedure: ZEMLIN TABLEAU |
| Specimen holder | Cryogen: NITROGEN / Specimen holder model: FEI TITAN KRIOS AUTOGRID HOLDER / Residual tilt: 0.01 mradians |
| Image recording | Average exposure time: 1.83 sec. / Electron dose: 50 e/Å2 / Film or detector model: GATAN K3 (6k x 4k) / Num. of grids imaged: 1 / Num. of real images: 5743 |
| EM imaging optics | Energyfilter name: GIF Bioquantum / Energyfilter slit width: 25 eV |
| Image scans | Sampling size: 5 µm / Width: 4092 / Height: 5760 |
- Processing
Processing
| EM software |
| |||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| CTF correction | Type: PHASE FLIPPING ONLY | |||||||||||||||||||||||||||||||||||||||||||||
| Particle selection | Num. of particles selected: 2796263 | |||||||||||||||||||||||||||||||||||||||||||||
| Symmetry | Point symmetry: C1 (asymmetric) | |||||||||||||||||||||||||||||||||||||||||||||
| 3D reconstruction | Resolution: 3.3 Å / Resolution method: FSC 0.143 CUT-OFF / Num. of particles: 178217 / Algorithm: FOURIER SPACE / Num. of class averages: 1 / Symmetry type: POINT | |||||||||||||||||||||||||||||||||||||||||||||
| Atomic model building | Protocol: FLEXIBLE FIT / Space: REAL | |||||||||||||||||||||||||||||||||||||||||||||
| Atomic model building | 3D fitting-ID: 1 / Source name: PDB / Type: experimental model
|
 Movie
Movie Controller
Controller












 PDBj
PDBj


























