+ データを開く
データを開く
- 基本情報
基本情報
| 登録情報 | データベース: PDB / ID: 7bgw | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| タイトル | 14-3-3 sigma with Pin1 binding site pS72 and covalently bound LvD1011 | |||||||||
 要素 要素 |
| |||||||||
 キーワード キーワード | PEPTIDE BINDING PROTEIN / 1433 / Peptidyl-prolyl cis-trans isomerase NIMA-interacting 1 | |||||||||
| 機能・相同性 |  機能・相同性情報 機能・相同性情報cis-trans isomerase activity / phosphothreonine residue binding / negative regulation of cell motility / ubiquitin ligase activator activity / regulation of protein localization to nucleus / GTPase activating protein binding / mitogen-activated protein kinase kinase binding / protein targeting to mitochondrion / protein peptidyl-prolyl isomerization / regulation of mitotic nuclear division ...cis-trans isomerase activity / phosphothreonine residue binding / negative regulation of cell motility / ubiquitin ligase activator activity / regulation of protein localization to nucleus / GTPase activating protein binding / mitogen-activated protein kinase kinase binding / protein targeting to mitochondrion / protein peptidyl-prolyl isomerization / regulation of mitotic nuclear division / regulation of epidermal cell division / protein kinase C inhibitor activity / positive regulation of epidermal cell differentiation / keratinocyte development / keratinization / negative regulation of SMAD protein signal transduction / PI5P Regulates TP53 Acetylation / regulation of cell-cell adhesion / negative regulation of amyloid-beta formation / cytoskeletal motor activity / cAMP/PKA signal transduction / Regulation of localization of FOXO transcription factors / keratinocyte proliferation / RHO GTPases Activate NADPH Oxidases / phosphoserine residue binding / Activation of BAD and translocation to mitochondria / negative regulation of keratinocyte proliferation / establishment of skin barrier / negative regulation of protein localization to plasma membrane / postsynaptic cytosol / Chk1/Chk2(Cds1) mediated inactivation of Cyclin B:Cdk1 complex / SARS-CoV-2 targets host intracellular signalling and regulatory pathways / negative regulation of protein kinase activity / negative regulation of stem cell proliferation / negative regulation of protein binding / Rho protein signal transduction / SARS-CoV-1 targets host intracellular signalling and regulatory pathways / RHO GTPases activate PKNs / positive regulation of protein localization / positive regulation of cell adhesion / protein sequestering activity / protein export from nucleus / negative regulation of innate immune response / TP53 Regulates Transcription of Genes Involved in G2 Cell Cycle Arrest / release of cytochrome c from mitochondria / positive regulation of protein export from nucleus / stem cell proliferation / regulation of cytokinesis / TP53 Regulates Metabolic Genes / Translocation of SLC2A4 (GLUT4) to the plasma membrane / peptidylprolyl isomerase / Negative regulators of DDX58/IFIH1 signaling / peptidyl-prolyl cis-trans isomerase activity / phosphoprotein binding / negative regulation of transforming growth factor beta receptor signaling pathway / synapse organization / beta-catenin binding / negative regulation of protein catabolic process / regulation of protein stability / negative regulation of ERK1 and ERK2 cascade / ISG15 antiviral mechanism / tau protein binding / positive regulation of protein phosphorylation / neuron differentiation / intrinsic apoptotic signaling pathway in response to DNA damage / intracellular protein localization / positive regulation of canonical Wnt signaling pathway / regulation of protein localization / regulation of gene expression / positive regulation of cell growth / midbody / cellular response to hypoxia / Regulation of TP53 Activity through Phosphorylation / response to hypoxia / regulation of cell cycle / protein stabilization / nuclear speck / ciliary basal body / cadherin binding / protein kinase binding / glutamatergic synapse / negative regulation of transcription by RNA polymerase II / signal transduction / positive regulation of transcription by RNA polymerase II / extracellular space / extracellular exosome / nucleoplasm / identical protein binding / nucleus / cytoplasm / cytosol 類似検索 - 分子機能 | |||||||||
| 生物種 |  Homo sapiens (ヒト) Homo sapiens (ヒト) | |||||||||
| 手法 |  X線回折 / X線回折 /  分子置換 / 解像度: 1.9 Å 分子置換 / 解像度: 1.9 Å | |||||||||
 データ登録者 データ登録者 | Wolter, M. / Dijck, L.v. / Cossar, P.J. / Ottmann, C. | |||||||||
| 資金援助 | 2件
| |||||||||
 引用 引用 |  ジャーナル: J.Am.Chem.Soc. / 年: 2021 ジャーナル: J.Am.Chem.Soc. / 年: 2021タイトル: Reversible Covalent Imine-Tethering for Selective Stabilization of 14-3-3 Hub Protein Interactions. 著者: Cossar, P.J. / Wolter, M. / van Dijck, L. / Valenti, D. / Levy, L.M. / Ottmann, C. / Brunsveld, L. | |||||||||
| 履歴 |
|
- 構造の表示
構造の表示
| 構造ビューア | 分子:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- ダウンロードとリンク
ダウンロードとリンク
- ダウンロード
ダウンロード
| PDBx/mmCIF形式 |  7bgw.cif.gz 7bgw.cif.gz | 74 KB | 表示 |  PDBx/mmCIF形式 PDBx/mmCIF形式 |
|---|---|---|---|---|
| PDB形式 |  pdb7bgw.ent.gz pdb7bgw.ent.gz | 51.3 KB | 表示 |  PDB形式 PDB形式 |
| PDBx/mmJSON形式 |  7bgw.json.gz 7bgw.json.gz | ツリー表示 |  PDBx/mmJSON形式 PDBx/mmJSON形式 | |
| その他 |  その他のダウンロード その他のダウンロード |
-検証レポート
| 文書・要旨 |  7bgw_validation.pdf.gz 7bgw_validation.pdf.gz | 633 KB | 表示 |  wwPDB検証レポート wwPDB検証レポート |
|---|---|---|---|---|
| 文書・詳細版 |  7bgw_full_validation.pdf.gz 7bgw_full_validation.pdf.gz | 633 KB | 表示 | |
| XML形式データ |  7bgw_validation.xml.gz 7bgw_validation.xml.gz | 14.4 KB | 表示 | |
| CIF形式データ |  7bgw_validation.cif.gz 7bgw_validation.cif.gz | 22.2 KB | 表示 | |
| アーカイブディレクトリ |  https://data.pdbj.org/pub/pdb/validation_reports/bg/7bgw https://data.pdbj.org/pub/pdb/validation_reports/bg/7bgw ftp://data.pdbj.org/pub/pdb/validation_reports/bg/7bgw ftp://data.pdbj.org/pub/pdb/validation_reports/bg/7bgw | HTTPS FTP |
-関連構造データ
| 関連構造データ |  7aogSC  7axnC  7ayfC  7az1C  7az2C  7bdpC  7bdtC  7bdyC  7bfwC  7bg3C  7bgqC  7bgrC  7bgvC  7nifC  7nigC  7nixC  7nj6C  7nj8C  7njaC  7nqpC  7nrkC  7nrlC  7nsvC S: 精密化の開始モデル C: 同じ文献を引用 ( |
|---|---|
| 類似構造データ |
- リンク
リンク
- 集合体
集合体
| 登録構造単位 | 
| ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | 
| ||||||||||||
| 単位格子 |
| ||||||||||||
| Components on special symmetry positions |
|
- 要素
要素
-タンパク質 / タンパク質・ペプチド , 2種, 2分子 AP
| #1: タンパク質 | 分子量: 28226.518 Da / 分子数: 1 / 由来タイプ: 組換発現 / 由来: (組換発現)  Homo sapiens (ヒト) / 遺伝子: SFN, HME1 / 発現宿主: Homo sapiens (ヒト) / 遺伝子: SFN, HME1 / 発現宿主:  |
|---|---|
| #2: タンパク質・ペプチド | 分子量: 2195.378 Da / 分子数: 1 / 由来タイプ: 合成 / 由来: (合成)  Homo sapiens (ヒト) / 参照: UniProt: Q13526, peptidylprolyl isomerase Homo sapiens (ヒト) / 参照: UniProt: Q13526, peptidylprolyl isomerase |
-非ポリマー , 5種, 352分子 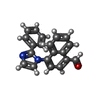








| #3: 化合物 | ChemComp-TLQ / | ||||||
|---|---|---|---|---|---|---|---|
| #4: 化合物 | | #5: 化合物 | ChemComp-CL / | #6: 化合物 | ChemComp-NA / | #7: 水 | ChemComp-HOH / | |
-詳細
| 研究の焦点であるリガンドがあるか | Y |
|---|---|
| Has protein modification | Y |
-実験情報
-実験
| 実験 | 手法:  X線回折 / 使用した結晶の数: 1 X線回折 / 使用した結晶の数: 1 |
|---|
- 試料調製
試料調製
| 結晶 | マシュー密度: 2.37 Å3/Da / 溶媒含有率: 48.09 % |
|---|---|
| 結晶化 | 温度: 277 K / 手法: 蒸気拡散法, ハンギングドロップ法 詳細: 0.095 M HEPES Na pH 7.1, 27% PEG400, 0.19M Calcium chloride, 5% Glycerol |
-データ収集
| 回折 | 平均測定温度: 100 K / Serial crystal experiment: N | ||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 放射光源 | 由来: SEALED TUBE / タイプ: RIGAKU MICROMAX-003 / 波長: 1.54187 Å | ||||||||||||||||||||||||||||||
| 検出器 | タイプ: DECTRIS PILATUS 200K / 検出器: PIXEL / 日付: 2020年1月29日 | ||||||||||||||||||||||||||||||
| 放射 | プロトコル: SINGLE WAVELENGTH / 単色(M)・ラウエ(L): M / 散乱光タイプ: x-ray | ||||||||||||||||||||||||||||||
| 放射波長 | 波長: 1.54187 Å / 相対比: 1 | ||||||||||||||||||||||||||||||
| 反射 | 解像度: 1.9→34.424 Å / Num. obs: 22305 / % possible obs: 96.4 % / 冗長度: 6.5 % / Biso Wilson estimate: 10.79 Å2 / CC1/2: 0.997 / Rmerge(I) obs: 0.094 / Rpim(I) all: 0.039 / Rrim(I) all: 0.102 / Net I/σ(I): 13.3 | ||||||||||||||||||||||||||||||
| 反射 シェル | Diffraction-ID: 1
|
- 解析
解析
| ソフトウェア |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 精密化 | 構造決定の手法:  分子置換 分子置換開始モデル: 7AOG 解像度: 1.9→34.424 Å / SU ML: 0.16 / 交差検証法: FREE R-VALUE / σ(F): 1.34 / 位相誤差: 20.1 / 立体化学のターゲット値: ML
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 溶媒の処理 | 減衰半径: 0.9 Å / VDWプローブ半径: 1.11 Å / 溶媒モデル: FLAT BULK SOLVENT MODEL | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 原子変位パラメータ | Biso max: 70.45 Å2 / Biso mean: 14.3277 Å2 / Biso min: 2.45 Å2 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 精密化ステップ | サイクル: final / 解像度: 1.9→34.424 Å
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 拘束条件 |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| LS精密化 シェル | Refine-ID: X-RAY DIFFRACTION / Rfactor Rfree error: 0
|
 ムービー
ムービー コントローラー
コントローラー























 PDBj
PDBj















