[English] 日本語
 Yorodumi
Yorodumi- PDB-6z44: Crystal structure of the cAMP-dependent protein kinase A in compl... -
+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 6z44 | ||||||
|---|---|---|---|---|---|---|---|
| Title | Crystal structure of the cAMP-dependent protein kinase A in complex with phenol | ||||||
 Components Components | cAMP-dependent protein kinase catalytic subunit alpha | ||||||
 Keywords Keywords | TRANSFERASE / phosphotransferase / signalling pathways / glycogen metabolism / serine/threonine kinase | ||||||
| Function / homology |  Function and homology information Function and homology informationhistone H1-4S35 kinase activity / cAMP-dependent protein kinase / regulation of protein processing / cAMP-dependent protein kinase activity / cellular response to parathyroid hormone stimulus / protein localization to lipid droplet / regulation of bicellular tight junction assembly / cAMP-dependent protein kinase complex / dorsal/ventral neural tube patterning / interleukin-2-mediated signaling pathway ...histone H1-4S35 kinase activity / cAMP-dependent protein kinase / regulation of protein processing / cAMP-dependent protein kinase activity / cellular response to parathyroid hormone stimulus / protein localization to lipid droplet / regulation of bicellular tight junction assembly / cAMP-dependent protein kinase complex / dorsal/ventral neural tube patterning / interleukin-2-mediated signaling pathway / regulation of osteoblast differentiation / sperm capacitation / cellular response to cold / ciliary base / protein kinase A regulatory subunit binding / negative regulation of glycolytic process through fructose-6-phosphate / intracellular potassium ion homeostasis / mesoderm formation / TORC1 signaling / plasma membrane raft / axoneme / sperm flagellum / postsynaptic modulation of chemical synaptic transmission / negative regulation of TORC1 signaling / regulation of proteasomal protein catabolic process / protein serine/threonine/tyrosine kinase activity / protein export from nucleus / cellular response to glucagon stimulus / cellular response to nutrient levels / positive regulation of phagocytosis / positive regulation of gluconeogenesis / acrosomal vesicle / positive regulation of protein export from nucleus / negative regulation of smoothened signaling pathway / neuromuscular junction / neural tube closure / cellular response to glucose stimulus / positive regulation of cholesterol biosynthetic process / mRNA processing / adenylate cyclase-inhibiting G protein-coupled receptor signaling pathway / positive regulation of insulin secretion / manganese ion binding / cellular response to heat / adenylate cyclase-activating G protein-coupled receptor signaling pathway / regulation of cell cycle / nuclear speck / postsynapse / protein domain specific binding / protein serine kinase activity / protein serine/threonine kinase activity / ubiquitin protein ligase binding / centrosome / protein kinase binding / perinuclear region of cytoplasm / glutamatergic synapse / magnesium ion binding / mitochondrion / ATP binding / nucleus / cytoplasm / cytosol Similarity search - Function | ||||||
| Biological species |  | ||||||
| Method |  X-RAY DIFFRACTION / X-RAY DIFFRACTION /  SYNCHROTRON / SYNCHROTRON /  MOLECULAR REPLACEMENT / Resolution: 1.38 Å MOLECULAR REPLACEMENT / Resolution: 1.38 Å | ||||||
 Authors Authors | Oebbeke, M. / Heine, A. / Klebe, G. | ||||||
 Citation Citation |  Journal: Angew.Chem.Int.Ed.Engl. / Year: 2021 Journal: Angew.Chem.Int.Ed.Engl. / Year: 2021Title: Fragment Binding to Kinase Hinge: If Charge Distribution and Local pK a Shifts Mislead Popular Bioisosterism Concepts. Authors: Oebbeke, M. / Siefker, C. / Wagner, B. / Heine, A. / Klebe, G. | ||||||
| History |
|
- Structure visualization
Structure visualization
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  6z44.cif.gz 6z44.cif.gz | 261 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb6z44.ent.gz pdb6z44.ent.gz | 175 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  6z44.json.gz 6z44.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/z4/6z44 https://data.pdbj.org/pub/pdb/validation_reports/z4/6z44 ftp://data.pdbj.org/pub/pdb/validation_reports/z4/6z44 ftp://data.pdbj.org/pub/pdb/validation_reports/z4/6z44 | HTTPS FTP |
|---|
-Related structure data
| Related structure data |  5n33C  5n3cC  5n3dC  5n3eC  5n3hC  5n3jC  5n3qC  5n3sC  6snnC  6snxC  6soxC  6spmC  6spsC  6spuC  6spyC  6ypsC  6z08C  6zn0C  6f14S S: Starting model for refinement C: citing same article ( |
|---|---|
| Similar structure data |
- Links
Links
- Assembly
Assembly
| Deposited unit | 
| ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 |
| ||||||||||||
| Unit cell |
|
- Components
Components
| #1: Protein | Mass: 41113.766 Da / Num. of mol.: 1 Source method: isolated from a genetically manipulated source Source: (gene. exp.)   | ||||||||
|---|---|---|---|---|---|---|---|---|---|
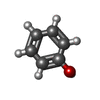
| #2: Chemical | ChemComp-IPH / | ||||||||
| #3: Chemical | | #4: Chemical | ChemComp-MRD / ( | #5: Water | ChemComp-HOH / | Has ligand of interest | Y | Has protein modification | Y | |
-Experimental details
-Experiment
| Experiment | Method:  X-RAY DIFFRACTION / Number of used crystals: 1 X-RAY DIFFRACTION / Number of used crystals: 1 |
|---|
- Sample preparation
Sample preparation
| Crystal | Density Matthews: 2.19 Å3/Da / Density % sol: 43.81 % |
|---|---|
| Crystal grow | Temperature: 277 K / Method: vapor diffusion, sitting drop / pH: 6.9 Details: 0,2 mM PKA in 100 mM MES-BIS-Tris-Buffer, 1 mM dithiothreitol, 0.1 mM sodium EDTA, 75 mM LiCl, 0.2 mM Mega 8 and 23 % methanol (v/v) 0.003 mL drop volume, 0.5 mL reservoir volume Soaking: ...Details: 0,2 mM PKA in 100 mM MES-BIS-Tris-Buffer, 1 mM dithiothreitol, 0.1 mM sodium EDTA, 75 mM LiCl, 0.2 mM Mega 8 and 23 % methanol (v/v) 0.003 mL drop volume, 0.5 mL reservoir volume Soaking: 100mM phenol in buffer described above and 30% MPD |
-Data collection
| Diffraction | Mean temperature: 100 K / Serial crystal experiment: N |
|---|---|
| Diffraction source | Source:  SYNCHROTRON / Site: SYNCHROTRON / Site:  BESSY BESSY  / Beamline: 14.2 / Wavelength: 0.9184 Å / Beamline: 14.2 / Wavelength: 0.9184 Å |
| Detector | Type: DECTRIS PILATUS3 2M / Detector: PIXEL / Date: Nov 8, 2019 |
| Radiation | Protocol: SINGLE WAVELENGTH / Monochromatic (M) / Laue (L): M / Scattering type: x-ray |
| Radiation wavelength | Wavelength: 0.9184 Å / Relative weight: 1 |
| Reflection | Resolution: 1.379→48.705 Å / Num. obs: 74984 / % possible obs: 99.8 % / Redundancy: 6.5 % / Biso Wilson estimate: 15.85 Å2 / Rsym value: 0.042 / Net I/σ(I): 21.7 |
| Reflection shell | Resolution: 1.38→1.46 Å / Mean I/σ(I) obs: 3.07 / Num. unique obs: 11843 / Rsym value: 0.497 |
- Processing
Processing
| Software |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Refinement | Method to determine structure:  MOLECULAR REPLACEMENT MOLECULAR REPLACEMENTStarting model: 6F14 Resolution: 1.38→20.12 Å / SU ML: 0.0995 / Cross valid method: FREE R-VALUE / σ(F): 1.37 / Phase error: 15.2211 Stereochemistry target values: GeoStd + Monomer Library + CDL v1.2
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Solvent computation | Shrinkage radii: 0.9 Å / VDW probe radii: 1.11 Å / Solvent model: FLAT BULK SOLVENT MODEL | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Displacement parameters | Biso mean: 20.12 Å2 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refinement step | Cycle: LAST / Resolution: 1.38→20.12 Å
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refine LS restraints |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| LS refinement shell |
|
 Movie
Movie Controller
Controller












 PDBj
PDBj