+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 6x0p | ||||||
|---|---|---|---|---|---|---|---|
| Title | Ash1L SET domain Q2265A mutant in complex with AS-5 | ||||||
 Components Components | Histone-lysine N-methyltransferase ASH1L | ||||||
 Keywords Keywords | PROTEIN BINDING / TRANSFERASE/INHIBITOR / Methyltransferase / complex / TRANSFERASE-INHIBITOR complex | ||||||
| Function / homology |  Function and homology information Function and homology informationuterine gland development / tarsal gland development / histone H3K9 monomethyltransferase activity / [histone H3]-lysine36 N-trimethyltransferase / histone H3K36 trimethyltransferase activity / uterus morphogenesis / [histone H3]-lysine9 N-methyltransferase / flagellated sperm motility / histone H3K9 methyltransferase activity / histone H3K9me2 methyltransferase activity ...uterine gland development / tarsal gland development / histone H3K9 monomethyltransferase activity / [histone H3]-lysine36 N-trimethyltransferase / histone H3K36 trimethyltransferase activity / uterus morphogenesis / [histone H3]-lysine9 N-methyltransferase / flagellated sperm motility / histone H3K9 methyltransferase activity / histone H3K9me2 methyltransferase activity / histone H3K36 methyltransferase activity / histone H3K4 methyltransferase activity / histone H3 methyltransferase activity / negative regulation of acute inflammatory response / decidualization / bicellular tight junction / single fertilization / negative regulation of MAPK cascade / post-embryonic development / skeletal system development / PKMTs methylate histone lysines / MAPK cascade / chromosome / methylation / transcription by RNA polymerase II / inflammatory response / chromatin binding / regulation of DNA-templated transcription / Golgi apparatus / positive regulation of transcription by RNA polymerase II / DNA binding / nucleoplasm / nucleus / metal ion binding Similarity search - Function | ||||||
| Biological species |  Homo sapiens (human) Homo sapiens (human) | ||||||
| Method |  X-RAY DIFFRACTION / X-RAY DIFFRACTION /  SYNCHROTRON / SYNCHROTRON /  MOLECULAR REPLACEMENT / Resolution: 1.69 Å MOLECULAR REPLACEMENT / Resolution: 1.69 Å | ||||||
 Authors Authors | Rogawski, D.S. / Li, H. / Borkin, D. / Cierpicki, T. / Grembecka, J. | ||||||
| Funding support |  United States, 1items United States, 1items
| ||||||
 Citation Citation |  Journal: Nat Commun / Year: 2021 Journal: Nat Commun / Year: 2021Title: Discovery of first-in-class inhibitors of ASH1L histone methyltransferase with anti-leukemic activity. Authors: Rogawski, D.S. / Deng, J. / Li, H. / Miao, H. / Borkin, D. / Purohit, T. / Song, J. / Chase, J. / Li, S. / Ndoj, J. / Klossowski, S. / Kim, E. / Mao, F. / Zhou, B. / Ropa, J. / Krotoska, M.Z. ...Authors: Rogawski, D.S. / Deng, J. / Li, H. / Miao, H. / Borkin, D. / Purohit, T. / Song, J. / Chase, J. / Li, S. / Ndoj, J. / Klossowski, S. / Kim, E. / Mao, F. / Zhou, B. / Ropa, J. / Krotoska, M.Z. / Jin, Z. / Ernst, P. / Feng, X. / Huang, G. / Nishioka, K. / Kelly, S. / He, M. / Wen, B. / Sun, D. / Muntean, A. / Dou, Y. / Maillard, I. / Cierpicki, T. / Grembecka, J. | ||||||
| History |
|
- Structure visualization
Structure visualization
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  6x0p.cif.gz 6x0p.cif.gz | 270.5 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb6x0p.ent.gz pdb6x0p.ent.gz | 171.8 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  6x0p.json.gz 6x0p.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Summary document |  6x0p_validation.pdf.gz 6x0p_validation.pdf.gz | 2.8 MB | Display |  wwPDB validaton report wwPDB validaton report |
|---|---|---|---|---|
| Full document |  6x0p_full_validation.pdf.gz 6x0p_full_validation.pdf.gz | 2.8 MB | Display | |
| Data in XML |  6x0p_validation.xml.gz 6x0p_validation.xml.gz | 46.1 KB | Display | |
| Data in CIF |  6x0p_validation.cif.gz 6x0p_validation.cif.gz | 68 KB | Display | |
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/x0/6x0p https://data.pdbj.org/pub/pdb/validation_reports/x0/6x0p ftp://data.pdbj.org/pub/pdb/validation_reports/x0/6x0p ftp://data.pdbj.org/pub/pdb/validation_reports/x0/6x0p | HTTPS FTP |
-Related structure data
| Related structure data |  6wzwC  4ynmS S: Starting model for refinement C: citing same article ( |
|---|---|
| Similar structure data |
- Links
Links
- Assembly
Assembly
| Deposited unit | 
| ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | 
| ||||||||||||
| 2 | 
| ||||||||||||
| 3 | 
| ||||||||||||
| 4 | 
| ||||||||||||
| Unit cell |
|
- Components
Components
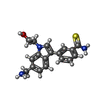
| #1: Protein | Mass: 25959.465 Da / Num. of mol.: 4 / Mutation: Q2265A Source method: isolated from a genetically manipulated source Source: (gene. exp.)  Homo sapiens (human) / Gene: ASH1L, KIAA1420, KMT2H / Production host: Homo sapiens (human) / Gene: ASH1L, KIAA1420, KMT2H / Production host:  References: UniProt: Q9NR48, Transferases; Transferring one-carbon groups; Methyltransferases, [histone H3]-lysine36 N-trimethyltransferase #2: Chemical | ChemComp-UK7 / #3: Chemical | ChemComp-ZN / #4: Chemical | ChemComp-SAM / #5: Water | ChemComp-HOH / | Has ligand of interest | Y | |
|---|
-Experimental details
-Experiment
| Experiment | Method:  X-RAY DIFFRACTION / Number of used crystals: 1 X-RAY DIFFRACTION / Number of used crystals: 1 |
|---|
- Sample preparation
Sample preparation
| Crystal | Density Matthews: 2.39 Å3/Da / Density % sol: 48.48 % |
|---|---|
| Crystal grow | Temperature: 290 K / Method: vapor diffusion, sitting drop / pH: 8.5 / Details: 0.1 M Tris pH 8.5, 0.2 M MgCl2, 30% PEG4000 |
-Data collection
| Diffraction | Mean temperature: 100 K / Serial crystal experiment: N |
|---|---|
| Diffraction source | Source:  SYNCHROTRON / Site: SYNCHROTRON / Site:  APS APS  / Beamline: 21-ID-F / Wavelength: 0.9787 Å / Beamline: 21-ID-F / Wavelength: 0.9787 Å |
| Detector | Type: MARMOSAIC 300 mm CCD / Detector: CCD / Date: Apr 21, 2015 |
| Radiation | Monochromator: M / Protocol: SINGLE WAVELENGTH / Monochromatic (M) / Laue (L): M / Scattering type: x-ray |
| Radiation wavelength | Wavelength: 0.9787 Å / Relative weight: 1 |
| Reflection | Resolution: 1.69→40.86 Å / Num. obs: 101290 / % possible obs: 95.8 % / Redundancy: 3.9 % / Biso Wilson estimate: 12.51 Å2 / Rsym value: 0.096 / Net I/σ(I): 14.4 |
| Reflection shell | Resolution: 1.7→1.73 Å / Redundancy: 3.7 % / Mean I/σ(I) obs: 2.1 / Num. unique obs: 9261 / Rsym value: 0.591 |
- Processing
Processing
| Software |
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Refinement | Method to determine structure:  MOLECULAR REPLACEMENT MOLECULAR REPLACEMENTStarting model: 4YNM Resolution: 1.69→40.86 Å / SU ML: 0.1651 / Cross valid method: FREE R-VALUE / σ(F): 1.98 / Phase error: 20.5298 Stereochemistry target values: GeoStd + Monomer Library + CDL v1.2
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Solvent computation | Shrinkage radii: 0.9 Å / VDW probe radii: 1.11 Å / Solvent model: FLAT BULK SOLVENT MODEL | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Displacement parameters | Biso mean: 18.93 Å2 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refinement step | Cycle: LAST / Resolution: 1.69→40.86 Å
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refine LS restraints |
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| LS refinement shell |
|
 Movie
Movie Controller
Controller













 PDBj
PDBj