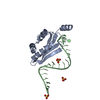
Entry Database : PDB / ID : 6ssrTitle Crystal structure of Human Microsomal Glutathione S-Transferase 2 at 3.8 Angstroms resolution Microsomal glutathione S-transferase 2 Keywords / / / / / Function / homology Function Domain/homology Component
/ / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / Biological species Homo sapiens (human)Method / / / Resolution : 3.8 Å Authors Thulasingam, M. / Nji, E. / Rinaldo-Matthis, A. / Haeggstrom, J.Z. Funding support Organization Grant number Country Swedish Research Council 10350
Journal : Nat Commun / Year : 2021Title : Crystal structures of human MGST2 reveal synchronized conformational changes regulating catalysis.Authors : Thulasingam, M. / Orellana, L. / Nji, E. / Ahmad, S. / Rinaldo-Matthis, A. / Haeggstrom, J.Z. History Deposition Sep 9, 2019 Deposition site / Processing site Revision 1.0 Feb 3, 2021 Provider / Type Revision 1.1 Apr 7, 2021 Group / Database referencesCategory citation / citation_author ... citation / citation_author / diffrn_radiation_wavelength / diffrn_source Item _citation.country / _citation.journal_abbrev ... _citation.country / _citation.journal_abbrev / _citation.journal_id_CSD / _citation.journal_id_ISSN / _citation.journal_volume / _citation.page_first / _citation.page_last / _citation.pdbx_database_id_DOI / _citation.pdbx_database_id_PubMed / _citation.title / _citation.year / _diffrn_radiation_wavelength.wavelength / _diffrn_source.pdbx_wavelength_list Revision 1.2 Jan 24, 2024 Group / Database references / Refinement descriptionCategory chem_comp_atom / chem_comp_bond ... chem_comp_atom / chem_comp_bond / database_2 / pdbx_initial_refinement_model / struct_ncs_dom_lim Item _database_2.pdbx_DOI / _database_2.pdbx_database_accession ... _database_2.pdbx_DOI / _database_2.pdbx_database_accession / _struct_ncs_dom_lim.beg_auth_comp_id / _struct_ncs_dom_lim.beg_label_asym_id / _struct_ncs_dom_lim.beg_label_comp_id / _struct_ncs_dom_lim.beg_label_seq_id / _struct_ncs_dom_lim.end_auth_comp_id / _struct_ncs_dom_lim.end_label_asym_id / _struct_ncs_dom_lim.end_label_comp_id / _struct_ncs_dom_lim.end_label_seq_id
Show all Show less
 Yorodumi
Yorodumi Open data
Open data Basic information
Basic information Components
Components Keywords
Keywords Function and homology information
Function and homology information Homo sapiens (human)
Homo sapiens (human) X-RAY DIFFRACTION /
X-RAY DIFFRACTION /  SYNCHROTRON /
SYNCHROTRON /  MOLECULAR REPLACEMENT / Resolution: 3.8 Å
MOLECULAR REPLACEMENT / Resolution: 3.8 Å  Authors
Authors Sweden, 1items
Sweden, 1items  Citation
Citation Journal: Nat Commun / Year: 2021
Journal: Nat Commun / Year: 2021 Structure visualization
Structure visualization Molmil
Molmil Jmol/JSmol
Jmol/JSmol Downloads & links
Downloads & links Download
Download 6ssr.cif.gz
6ssr.cif.gz PDBx/mmCIF format
PDBx/mmCIF format pdb6ssr.ent.gz
pdb6ssr.ent.gz PDB format
PDB format 6ssr.json.gz
6ssr.json.gz PDBx/mmJSON format
PDBx/mmJSON format Other downloads
Other downloads https://data.pdbj.org/pub/pdb/validation_reports/ss/6ssr
https://data.pdbj.org/pub/pdb/validation_reports/ss/6ssr ftp://data.pdbj.org/pub/pdb/validation_reports/ss/6ssr
ftp://data.pdbj.org/pub/pdb/validation_reports/ss/6ssr



 Links
Links Assembly
Assembly
 Components
Components Homo sapiens (human) / Gene: MGST2, GST2 / Production host:
Homo sapiens (human) / Gene: MGST2, GST2 / Production host:  Komagataella pastoris (fungus) / References: UniProt: Q99735, glutathione transferase
Komagataella pastoris (fungus) / References: UniProt: Q99735, glutathione transferase X-RAY DIFFRACTION / Number of used crystals: 1
X-RAY DIFFRACTION / Number of used crystals: 1  Sample preparation
Sample preparation SYNCHROTRON / Site:
SYNCHROTRON / Site:  Diamond
Diamond  / Beamline: I04 / Wavelength: 0.9795 Å
/ Beamline: I04 / Wavelength: 0.9795 Å Processing
Processing MOLECULAR REPLACEMENT
MOLECULAR REPLACEMENT Movie
Movie Controller
Controller









 PDBj
PDBj

