[English] 日本語
 Yorodumi
Yorodumi- PDB-6d9h: Cryo-EM structure of the human adenosine A1 receptor-Gi2-protein ... -
+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 6d9h | ||||||
|---|---|---|---|---|---|---|---|
| Title | Cryo-EM structure of the human adenosine A1 receptor-Gi2-protein complex bound to its endogenous agonist | ||||||
 Components Components |
| ||||||
 Keywords Keywords | SIGNALING PROTEIN / membrane protein / active-state G protein-coupled receptor / adenosine A1 receptor | ||||||
| Function / homology |  Function and homology information Function and homology informationpositive regulation of nucleoside transport / negative regulation of neurotrophin production / regulation of glomerular filtration / negative regulation of circadian sleep/wake cycle, non-REM sleep / G protein-coupled purinergic nucleotide receptor signaling pathway / negative regulation of mucus secretion / purine nucleoside binding / positive regulation of peptide secretion / negative regulation of glutamate secretion / negative regulation of synaptic transmission, GABAergic ...positive regulation of nucleoside transport / negative regulation of neurotrophin production / regulation of glomerular filtration / negative regulation of circadian sleep/wake cycle, non-REM sleep / G protein-coupled purinergic nucleotide receptor signaling pathway / negative regulation of mucus secretion / purine nucleoside binding / positive regulation of peptide secretion / negative regulation of glutamate secretion / negative regulation of synaptic transmission, GABAergic / negative regulation of long-term synaptic depression / positive regulation of dephosphorylation / positive regulation of lipid catabolic process / negative regulation of hormone secretion / mucus secretion / negative regulation of adenylate cyclase-activating adrenergic receptor signaling pathway / negative regulation of leukocyte migration / Muscarinic acetylcholine receptors / G protein-coupled acetylcholine receptor activity / Adenosine P1 receptors / regulation of sensory perception of pain / G protein-coupled adenosine receptor activity / regulation of respiratory gaseous exchange by nervous system process / heterotrimeric G-protein binding / response to purine-containing compound / regulation of presynaptic cytosolic calcium ion concentration / negative regulation of calcium ion-dependent exocytosis / G protein-coupled adenosine receptor signaling pathway / negative regulation of adenylate cyclase activity / positive regulation of potassium ion transport / adenylate cyclase-inhibiting G protein-coupled acetylcholine receptor signaling pathway / positive regulation of neural precursor cell proliferation / negative regulation of systemic arterial blood pressure / negative regulation of synaptic transmission / regulation of cardiac muscle cell contraction / presynaptic active zone / positive regulation of urine volume / negative regulation of synaptic transmission, glutamatergic / long-term synaptic depression / protein targeting to membrane / leukocyte migration / triglyceride homeostasis / gamma-aminobutyric acid signaling pathway / regulation of calcium ion transport / regulation of locomotion / temperature homeostasis / negative regulation of acute inflammatory response / detection of temperature stimulus involved in sensory perception of pain / positive regulation of systemic arterial blood pressure / negative regulation of long-term synaptic potentiation / asymmetric synapse / negative regulation of apoptotic signaling pathway / axolemma / fatty acid homeostasis / phagocytosis / G protein-coupled receptor signaling pathway, coupled to cyclic nucleotide second messenger / negative regulation of lipid catabolic process / neuronal dense core vesicle / lipid catabolic process / positive regulation of vascular associated smooth muscle cell proliferation / positive regulation of superoxide anion generation / heat shock protein binding / Adenylate cyclase inhibitory pathway / response to nutrient / calyx of Held / hippocampal mossy fiber to CA3 synapse / apoptotic signaling pathway / Regulation of insulin secretion / excitatory postsynaptic potential / G protein-coupled receptor binding / vasodilation / cognition / G-protein beta/gamma-subunit complex binding / adenylate cyclase-inhibiting G protein-coupled receptor signaling pathway / Olfactory Signaling Pathway / Activation of the phototransduction cascade / G protein-coupled acetylcholine receptor signaling pathway / terminal bouton / G beta:gamma signalling through PLC beta / Presynaptic function of Kainate receptors / Thromboxane signalling through TP receptor / Activation of G protein gated Potassium channels / Inhibition of voltage gated Ca2+ channels via Gbeta/gamma subunits / G-protein activation / Glucagon signaling in metabolic regulation / G beta:gamma signalling through CDC42 / Prostacyclin signalling through prostacyclin receptor / G beta:gamma signalling through BTK / Synthesis, secretion, and inactivation of Glucagon-like Peptide-1 (GLP-1) / photoreceptor disc membrane / ADP signalling through P2Y purinoceptor 12 / Sensory perception of sweet, bitter, and umami (glutamate) taste / Glucagon-type ligand receptors / Adrenaline,noradrenaline inhibits insulin secretion / Vasopressin regulates renal water homeostasis via Aquaporins / Glucagon-like Peptide-1 (GLP1) regulates insulin secretion / G alpha (z) signalling events / cellular response to catecholamine stimulus / cell-cell signaling / ADP signalling through P2Y purinoceptor 1 Similarity search - Function | ||||||
| Biological species |  Homo sapiens (human) Homo sapiens (human) | ||||||
| Method | ELECTRON MICROSCOPY / single particle reconstruction / cryo EM / Resolution: 3.6 Å | ||||||
 Authors Authors | Draper-Joyce, C.J. / Khoshouei, M. / Thal, D.M. / Liang, Y.-L. / Nguyen, A.T.N. / Furness, S.G.B. / Venugopal, H. / Baltos, J. / Plitzko, J.M. / Danev, R. ...Draper-Joyce, C.J. / Khoshouei, M. / Thal, D.M. / Liang, Y.-L. / Nguyen, A.T.N. / Furness, S.G.B. / Venugopal, H. / Baltos, J. / Plitzko, J.M. / Danev, R. / Baumeister, W. / May, L.T. / Wootten, D. / Sexton, P. / Glukhova, A. / Christopoulos, A. | ||||||
 Citation Citation |  Journal: Nature / Year: 2018 Journal: Nature / Year: 2018Title: Structure of the adenosine-bound human adenosine A receptor-G complex. Authors: Christopher J Draper-Joyce / Maryam Khoshouei / David M Thal / Yi-Lynn Liang / Anh T N Nguyen / Sebastian G B Furness / Hariprasad Venugopal / Jo-Anne Baltos / Jürgen M Plitzko / Radostin ...Authors: Christopher J Draper-Joyce / Maryam Khoshouei / David M Thal / Yi-Lynn Liang / Anh T N Nguyen / Sebastian G B Furness / Hariprasad Venugopal / Jo-Anne Baltos / Jürgen M Plitzko / Radostin Danev / Wolfgang Baumeister / Lauren T May / Denise Wootten / Patrick M Sexton / Alisa Glukhova / Arthur Christopoulos /     Abstract: The class A adenosine A receptor (AR) is a G-protein-coupled receptor that preferentially couples to inhibitory G heterotrimeric G proteins, has been implicated in numerous diseases, yet remains ...The class A adenosine A receptor (AR) is a G-protein-coupled receptor that preferentially couples to inhibitory G heterotrimeric G proteins, has been implicated in numerous diseases, yet remains poorly targeted. Here we report the 3.6 Å structure of the human AR in complex with adenosine and heterotrimeric G protein determined by Volta phase plate cryo-electron microscopy. Compared to inactive AR, there is contraction at the extracellular surface in the orthosteric binding site mediated via movement of transmembrane domains 1 and 2. At the intracellular surface, the G protein engages the AR primarily via amino acids in the C terminus of the Gα α5-helix, concomitant with a 10.5 Å outward movement of the AR transmembrane domain 6. Comparison with the agonist-bound β adrenergic receptor-G-protein complex reveals distinct orientations for each G-protein subtype upon engagement with its receptor. This active AR structure provides molecular insights into receptor and G-protein selectivity. | ||||||
| History |
|
- Structure visualization
Structure visualization
| Movie |
 Movie viewer Movie viewer |
|---|---|
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  6d9h.cif.gz 6d9h.cif.gz | 176.3 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb6d9h.ent.gz pdb6d9h.ent.gz | 132.1 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  6d9h.json.gz 6d9h.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/d9/6d9h https://data.pdbj.org/pub/pdb/validation_reports/d9/6d9h ftp://data.pdbj.org/pub/pdb/validation_reports/d9/6d9h ftp://data.pdbj.org/pub/pdb/validation_reports/d9/6d9h | HTTPS FTP |
|---|
-Related structure data
| Related structure data |  7835MC M: map data used to model this data C: citing same article ( |
|---|---|
| Similar structure data |
- Links
Links
- Assembly
Assembly
| Deposited unit | 
|
|---|---|
| 1 |
|
- Components
Components
| #1: Protein | Mass: 40502.863 Da / Num. of mol.: 1 Source method: isolated from a genetically manipulated source Source: (gene. exp.)  Homo sapiens (human) / Gene: GNAI2, GNAI2B / Production host: Homo sapiens (human) / Gene: GNAI2, GNAI2B / Production host:  Trichoplusia ni (cabbage looper) / References: UniProt: P04899 Trichoplusia ni (cabbage looper) / References: UniProt: P04899 |
|---|---|
| #2: Protein | Mass: 38534.062 Da / Num. of mol.: 1 Source method: isolated from a genetically manipulated source Source: (gene. exp.)  Homo sapiens (human) / Gene: GNB1 / Production host: Homo sapiens (human) / Gene: GNB1 / Production host:  Trichoplusia ni (cabbage looper) / References: UniProt: P62873 Trichoplusia ni (cabbage looper) / References: UniProt: P62873 |
| #3: Protein | Mass: 7861.143 Da / Num. of mol.: 1 Source method: isolated from a genetically manipulated source Source: (gene. exp.)  Homo sapiens (human) / Gene: GNG2 / Production host: Homo sapiens (human) / Gene: GNG2 / Production host:  Trichoplusia ni (cabbage looper) / References: UniProt: P59768 Trichoplusia ni (cabbage looper) / References: UniProt: P59768 |
| #4: Protein | Mass: 43537.309 Da / Num. of mol.: 1 Source method: isolated from a genetically manipulated source Source: (gene. exp.)  Homo sapiens (human) / Gene: CHRM4, ADORA1 / Production host: Homo sapiens (human) / Gene: CHRM4, ADORA1 / Production host:  Trichoplusia ni (cabbage looper) / References: UniProt: P08173, UniProt: P30542 Trichoplusia ni (cabbage looper) / References: UniProt: P08173, UniProt: P30542 |
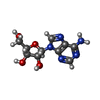
| #5: Chemical | ChemComp-ADN / |
| Has protein modification | Y |
-Experimental details
-Experiment
| Experiment | Method: ELECTRON MICROSCOPY |
|---|---|
| EM experiment | Aggregation state: PARTICLE / 3D reconstruction method: single particle reconstruction |
- Sample preparation
Sample preparation
| Component | Name: Human adenosine A1 receptor-Gi2-protein complex bound to its endogenous agonist adenosine Type: COMPLEX / Entity ID: #1-#4 / Source: RECOMBINANT |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Source (recombinant) | Organism:  Trichoplusia ni (cabbage looper) Trichoplusia ni (cabbage looper) |
| Buffer solution | pH: 7.5 |
| Specimen | Embedding applied: NO / Shadowing applied: NO / Staining applied: NO / Vitrification applied: YES |
| Specimen support | Grid material: COPPER / Grid mesh size: 200 divisions/in. / Grid type: Quantifoil R1.2/1.3 |
| Vitrification | Instrument: FEI VITROBOT MARK IV / Cryogen name: ETHANE-PROPANE / Humidity: 100 % / Chamber temperature: 277 K |
- Electron microscopy imaging
Electron microscopy imaging
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
|---|---|
| Microscopy | Model: FEI TITAN KRIOS |
| Electron gun | Electron source:  FIELD EMISSION GUN / Accelerating voltage: 300 kV / Illumination mode: OTHER FIELD EMISSION GUN / Accelerating voltage: 300 kV / Illumination mode: OTHER |
| Electron lens | Mode: BRIGHT FIELD / Calibrated magnification: 47170 X / C2 aperture diameter: 50 µm |
| Specimen holder | Cryogen: NITROGEN / Specimen holder model: FEI TITAN KRIOS AUTOGRID HOLDER |
| Image recording | Average exposure time: 8 sec. / Electron dose: 50 e/Å2 / Detector mode: COUNTING / Film or detector model: GATAN K2 SUMMIT (4k x 4k) |
| Image scans | Width: 3838 / Height: 3710 / Movie frames/image: 50 |
- Processing
Processing
| Software | Name: PHENIX / Version: 1.12_2829: / Classification: refinement | ||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| CTF correction | Details: Phase plate CTF correction / Type: NONE | ||||||||||||||||||||||||
| Symmetry | Point symmetry: C1 (asymmetric) | ||||||||||||||||||||||||
| 3D reconstruction | Resolution: 3.6 Å / Resolution method: FSC 0.143 CUT-OFF / Num. of particles: 263321 / Symmetry type: POINT | ||||||||||||||||||||||||
| Refine LS restraints |
|
 Movie
Movie Controller
Controller













 PDBj
PDBj