[English] 日本語
 Yorodumi
Yorodumi- PDB-5oc7: Crystal structure of the pleckstrin-homology domain of Bcr-Abl in... -
+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 5oc7 | ||||||
|---|---|---|---|---|---|---|---|
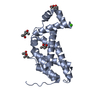
| Title | Crystal structure of the pleckstrin-homology domain of Bcr-Abl in complex with monobody Mb(Bcr-PH_4). | ||||||
 Components Components |
| ||||||
 Keywords Keywords | SIGNALING PROTEIN / pleckstrin-homology / monobody / Bcr-Abl / phosphoinositide-binding | ||||||
| Function / homology |  Function and homology information Function and homology informationnegative regulation of respiratory burst / negative regulation of cellular extravasation / negative regulation of macrophage migration / neutrophil degranulation / negative regulation of blood vessel remodeling / negative regulation of neutrophil degranulation / macrophage migration / intracellular protein transmembrane transport / renal system process / regulation of Rho protein signal transduction ...negative regulation of respiratory burst / negative regulation of cellular extravasation / negative regulation of macrophage migration / neutrophil degranulation / negative regulation of blood vessel remodeling / negative regulation of neutrophil degranulation / macrophage migration / intracellular protein transmembrane transport / renal system process / regulation of Rho protein signal transduction / regulation of vascular permeability / activation of GTPase activity / focal adhesion assembly / Signaling by cytosolic FGFR1 fusion mutants / definitive hemopoiesis / regulation of small GTPase mediated signal transduction / inner ear morphogenesis / neuromuscular process controlling balance / RHOB GTPase cycle / small GTPase-mediated signal transduction / RHOC GTPase cycle / negative regulation of reactive oxygen species metabolic process / CDC42 GTPase cycle / homeostasis of number of cells / RHOA GTPase cycle / RAC2 GTPase cycle / RAC3 GTPase cycle / phagocytosis / keratinocyte differentiation / RAC1 GTPase cycle / Signaling by FGFR1 in disease / GTPase activator activity / positive regulation of phagocytosis / guanyl-nucleotide exchange factor activity / modulation of chemical synaptic transmission / brain development / negative regulation of inflammatory response / Schaffer collateral - CA1 synapse / cellular response to lipopolysaccharide / actin cytoskeleton organization / protein tyrosine kinase activity / dendritic spine / protein phosphorylation / non-specific serine/threonine protein kinase / regulation of cell cycle / postsynaptic density / axon / protein serine kinase activity / protein serine/threonine kinase activity / glutamatergic synapse / signal transduction / protein-containing complex / extracellular exosome / ATP binding / membrane / plasma membrane / cytosol Similarity search - Function | ||||||
| Biological species |  Homo sapiens (human) Homo sapiens (human)synthetic construct (others) | ||||||
| Method |  X-RAY DIFFRACTION / X-RAY DIFFRACTION /  SYNCHROTRON / SYNCHROTRON /  MOLECULAR REPLACEMENT / Resolution: 1.652 Å MOLECULAR REPLACEMENT / Resolution: 1.652 Å | ||||||
 Authors Authors | Reckel, S. / Reynaud, A. / Pojer, F. / Hantschel, O. | ||||||
| Funding support |  Switzerland, 1items Switzerland, 1items
| ||||||
 Citation Citation |  Journal: Nat Commun / Year: 2017 Journal: Nat Commun / Year: 2017Title: Structural and functional dissection of the DH and PH domains of oncogenic Bcr-Abl tyrosine kinase. Authors: Sina Reckel / Charlotte Gehin / Delphine Tardivon / Sandrine Georgeon / Tim Kükenshöner / Frank Löhr / Akiko Koide / Lena Buchner / Alejandro Panjkovich / Aline Reynaud / Sara Pinho / ...Authors: Sina Reckel / Charlotte Gehin / Delphine Tardivon / Sandrine Georgeon / Tim Kükenshöner / Frank Löhr / Akiko Koide / Lena Buchner / Alejandro Panjkovich / Aline Reynaud / Sara Pinho / Barbara Gerig / Dmitri Svergun / Florence Pojer / Peter Güntert / Volker Dötsch / Shohei Koide / Anne-Claude Gavin / Oliver Hantschel /     Abstract: The two isoforms of the Bcr-Abl tyrosine kinase, p210 and p190, are associated with different leukemias and have a dramatically different signaling network, despite similar kinase activity. To ...The two isoforms of the Bcr-Abl tyrosine kinase, p210 and p190, are associated with different leukemias and have a dramatically different signaling network, despite similar kinase activity. To provide a molecular rationale for these observations, we study the Dbl-homology (DH) and Pleckstrin-homology (PH) domains of Bcr-Abl p210, which constitute the only structural differences to p190. Here we report high-resolution structures of the DH and PH domains and characterize conformations of the DH-PH unit in solution. Our structural and functional analyses show no evidence that the DH domain acts as a guanine nucleotide exchange factor, whereas the PH domain binds to various phosphatidylinositol-phosphates. PH-domain mutants alter subcellular localization and result in decreased interactions with p210-selective interaction partners. Hence, the PH domain, but not the DH domain, plays an important role in the formation of the differential p210 and p190 Bcr-Abl signaling networks. | ||||||
| History |
|
- Structure visualization
Structure visualization
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  5oc7.cif.gz 5oc7.cif.gz | 175.7 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb5oc7.ent.gz pdb5oc7.ent.gz | 142.2 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  5oc7.json.gz 5oc7.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Summary document |  5oc7_validation.pdf.gz 5oc7_validation.pdf.gz | 876.8 KB | Display |  wwPDB validaton report wwPDB validaton report |
|---|---|---|---|---|
| Full document |  5oc7_full_validation.pdf.gz 5oc7_full_validation.pdf.gz | 881 KB | Display | |
| Data in XML |  5oc7_validation.xml.gz 5oc7_validation.xml.gz | 20.5 KB | Display | |
| Data in CIF |  5oc7_validation.cif.gz 5oc7_validation.cif.gz | 28.9 KB | Display | |
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/oc/5oc7 https://data.pdbj.org/pub/pdb/validation_reports/oc/5oc7 ftp://data.pdbj.org/pub/pdb/validation_reports/oc/5oc7 ftp://data.pdbj.org/pub/pdb/validation_reports/oc/5oc7 | HTTPS FTP |
-Related structure data
| Related structure data |  5n6rC  5n7eC  2dfkS S: Starting model for refinement C: citing same article ( |
|---|---|
| Similar structure data |
- Links
Links
- Assembly
Assembly
| Deposited unit | 
| ||||||||
|---|---|---|---|---|---|---|---|---|---|
| 1 | 
| ||||||||
| 2 | 
| ||||||||
| 3 |
| ||||||||
| 4 | 
| ||||||||
| Unit cell |
|
- Components
Components
| #1: Protein | Mass: 15643.062 Da / Num. of mol.: 2 / Mutation: delta 770-829,delta 770-829 Source method: isolated from a genetically manipulated source Source: (gene. exp.)  Homo sapiens (human) / Gene: BCR, BCR1, D22S11 / Production host: Homo sapiens (human) / Gene: BCR, BCR1, D22S11 / Production host:  References: UniProt: P11274, non-specific serine/threonine protein kinase #2: Protein | Mass: 9632.773 Da / Num. of mol.: 2 Source method: isolated from a genetically manipulated source Source: (gene. exp.) synthetic construct (others) / Production host:  #3: Chemical | ChemComp-GOL / #4: Chemical | ChemComp-IP2 / | #5: Water | ChemComp-HOH / | |
|---|
-Experimental details
-Experiment
| Experiment | Method:  X-RAY DIFFRACTION / Number of used crystals: 1 X-RAY DIFFRACTION / Number of used crystals: 1 |
|---|
- Sample preparation
Sample preparation
| Crystal | Density Matthews: 2.47 Å3/Da / Density % sol: 50 % |
|---|---|
| Crystal grow | Temperature: 291 K / Method: vapor diffusion, sitting drop Details: 0.1 M Potassium thiocyanate, 30% (w/v) PEG MME 2000 |
-Data collection
| Diffraction | Mean temperature: 100 K |
|---|---|
| Diffraction source | Source:  SYNCHROTRON / Site: SYNCHROTRON / Site:  SLS SLS  / Beamline: X06DA / Wavelength: 0.99987 Å / Beamline: X06DA / Wavelength: 0.99987 Å |
| Detector | Type: DECTRIS PILATUS 2M / Detector: PIXEL / Date: May 19, 2017 |
| Radiation | Protocol: SINGLE WAVELENGTH / Monochromatic (M) / Laue (L): M / Scattering type: x-ray |
| Radiation wavelength | Wavelength: 0.99987 Å / Relative weight: 1 |
| Reflection | Resolution: 1.652→33.68 Å / Num. obs: 49951 / % possible obs: 97.13 % / Redundancy: 4.899 % / Rmerge(I) obs: 0.03096 / Rpim(I) all: 0.01554 / Rrim(I) all: 0.03473 / Net I/σ(I): 23.01 |
| Reflection shell | Resolution: 1.652→1.711 Å / Rmerge(I) obs: 0.6334 / Num. unique obs: 4899 / Rpim(I) all: 0.3281 / Rrim(I) all: 0.7159 / % possible all: 96.1 |
- Processing
Processing
| Software |
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Refinement | Method to determine structure:  MOLECULAR REPLACEMENT MOLECULAR REPLACEMENTStarting model: 2DFK Resolution: 1.652→33.68 Å / SU ML: 0.2 / Cross valid method: NONE / σ(F): 1.97 / Phase error: 21.66
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Solvent computation | Shrinkage radii: 0.9 Å / VDW probe radii: 1.11 Å | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refinement step | Cycle: LAST / Resolution: 1.652→33.68 Å
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refine LS restraints |
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| LS refinement shell |
|
 Movie
Movie Controller
Controller














 PDBj
PDBj