+ データを開く
データを開く
- 基本情報
基本情報
| 登録情報 | データベース: PDB / ID: 5n9l | ||||||
|---|---|---|---|---|---|---|---|
| タイトル | Crystal structure of human Protein kinase CK2 catalytic subunit in complex with the ATP-competitive dibenzofuran inhibitor TF (4b) | ||||||
 要素 要素 | Casein kinase II subunit alpha | ||||||
 キーワード キーワード | TRANSFERASE / Protein kinase / CK2 / Casein kinase 2 / Protein phosphorylation / ATP-competitive inhititors / dibenzofuran derivatives / TIGHT-BINDING INHIBITORS | ||||||
| 機能・相同性 |  機能・相同性情報 機能・相同性情報regulation of chromosome separation / positive regulation of aggrephagy / WNT mediated activation of DVL / Condensation of Prometaphase Chromosomes / protein kinase CK2 complex / symbiont-mediated disruption of host cell PML body / Receptor Mediated Mitophagy / Sin3-type complex / Synthesis of PC / RUNX1 interacts with co-factors whose precise effect on RUNX1 targets is not known ...regulation of chromosome separation / positive regulation of aggrephagy / WNT mediated activation of DVL / Condensation of Prometaphase Chromosomes / protein kinase CK2 complex / symbiont-mediated disruption of host cell PML body / Receptor Mediated Mitophagy / Sin3-type complex / Synthesis of PC / RUNX1 interacts with co-factors whose precise effect on RUNX1 targets is not known / Maturation of hRSV A proteins / negative regulation of apoptotic signaling pathway / positive regulation of Wnt signaling pathway / negative regulation of double-strand break repair via homologous recombination / chaperone-mediated protein folding / negative regulation of ubiquitin-dependent protein catabolic process / Signal transduction by L1 / peptidyl-threonine phosphorylation / Hsp90 protein binding / negative regulation of cysteine-type endopeptidase activity involved in apoptotic process / PML body / Wnt signaling pathway / Regulation of PTEN stability and activity / positive regulation of protein catabolic process / Cooperation of PDCL (PhLP1) and TRiC/CCT in G-protein beta folding / KEAP1-NFE2L2 pathway / double-strand break repair / rhythmic process / kinase activity / positive regulation of cell growth / peptidyl-serine phosphorylation / Regulation of TP53 Activity through Phosphorylation / protein stabilization / negative regulation of translation / non-specific serine/threonine protein kinase / regulation of cell cycle / cell cycle / protein phosphorylation / protein serine kinase activity / protein serine/threonine kinase activity / apoptotic process / DNA damage response / positive regulation of cell population proliferation / signal transduction / nucleoplasm / ATP binding / identical protein binding / nucleus / plasma membrane / cytosol 類似検索 - 分子機能 | ||||||
| 生物種 |  Homo sapiens (ヒト) Homo sapiens (ヒト) | ||||||
| 手法 |  X線回折 / X線回折 /  シンクロトロン / シンクロトロン /  分子置換 / 解像度: 1.79 Å 分子置換 / 解像度: 1.79 Å | ||||||
 データ登録者 データ登録者 | Schnitzler, A. / Gratz, A. / Bollacke, A. / Weyrich, M. / Kucklaender, U. / Wuensch, B. / Goetz, C. / Niefind, K. / Jose, J. | ||||||
 引用 引用 |  ジャーナル: Pharmaceuticals / 年: 2018 ジャーナル: Pharmaceuticals / 年: 2018タイトル: A pi-Halogen Bond of Dibenzofuranones with the Gatekeeper Phe113 in Human Protein Kinase CK2 Leads to Potent Tight Binding Inhibitors. 著者: Schnitzler, A. / Gratz, A. / Bollacke, A. / Weyrich, M. / Kucklander, U. / Wunsch, B. / Gotz, C. / Niefind, K. / Jose, J. #1:  ジャーナル: J. Mol. Biol. / 年: 2003 ジャーナル: J. Mol. Biol. / 年: 2003タイトル: Crystal structure of a C-terminal deletion mutant of human protein kinase CK2 catalytic subunit. 著者: Ermakova, I. / Boldyreff, B. / Issinger, O.G. / Niefind, K. #2:  ジャーナル: J. Mol. Biol. / 年: 2005 ジャーナル: J. Mol. Biol. / 年: 2005タイトル: Inclining the purine base binding plane in protein kinase CK2 by exchanging the flanking side-chains generates a preference for ATP as a cosubstrate. 著者: Yde, C.W. / Ermakova, I. / Issinger, O.G. / Niefind, K. #3:  ジャーナル: Pharmaceuticals (Basel) / 年: 2017 ジャーナル: Pharmaceuticals (Basel) / 年: 2017タイトル: Structural Hypervariability of the Two Human Protein Kinase CK2 Catalytic Subunit Paralogs Revealed by Complex Structures with a Flavonol- and a Thieno[2,3-d]pyrimidine-Based Inhibitor. 著者: Niefind, K. / Bischoff, N. / Golub, A.G. / Bdzhola, V.G. / Balanda, A.O. / Prykhod'ko, A.O. / Yarmoluk, S.M. #4: ジャーナル: Biochim. Biophys. Acta / 年: 2012 タイトル: TF--a novel cell-permeable and selective inhibitor of human protein kinase CK2 induces apoptosis in the prostate cancer cell line LNCaP. 著者: Goetz, C. / Gratz, A. / Kucklaender, U. / Jose, J. | ||||||
| 履歴 |
|
- 構造の表示
構造の表示
| 構造ビューア | 分子:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- ダウンロードとリンク
ダウンロードとリンク
- ダウンロード
ダウンロード
| PDBx/mmCIF形式 |  5n9l.cif.gz 5n9l.cif.gz | 160.8 KB | 表示 |  PDBx/mmCIF形式 PDBx/mmCIF形式 |
|---|---|---|---|---|
| PDB形式 |  pdb5n9l.ent.gz pdb5n9l.ent.gz | 126.4 KB | 表示 |  PDB形式 PDB形式 |
| PDBx/mmJSON形式 |  5n9l.json.gz 5n9l.json.gz | ツリー表示 |  PDBx/mmJSON形式 PDBx/mmJSON形式 | |
| その他 |  その他のダウンロード その他のダウンロード |
-検証レポート
| 文書・要旨 |  5n9l_validation.pdf.gz 5n9l_validation.pdf.gz | 806.2 KB | 表示 |  wwPDB検証レポート wwPDB検証レポート |
|---|---|---|---|---|
| 文書・詳細版 |  5n9l_full_validation.pdf.gz 5n9l_full_validation.pdf.gz | 808.5 KB | 表示 | |
| XML形式データ |  5n9l_validation.xml.gz 5n9l_validation.xml.gz | 16.7 KB | 表示 | |
| CIF形式データ |  5n9l_validation.cif.gz 5n9l_validation.cif.gz | 24 KB | 表示 | |
| アーカイブディレクトリ |  https://data.pdbj.org/pub/pdb/validation_reports/n9/5n9l https://data.pdbj.org/pub/pdb/validation_reports/n9/5n9l ftp://data.pdbj.org/pub/pdb/validation_reports/n9/5n9l ftp://data.pdbj.org/pub/pdb/validation_reports/n9/5n9l | HTTPS FTP |
-関連構造データ
- リンク
リンク
- 集合体
集合体
| 登録構造単位 | 
| ||||||||
|---|---|---|---|---|---|---|---|---|---|
| 1 |
| ||||||||
| 単位格子 |
|
- 要素
要素
| #1: タンパク質 | 分子量: 40066.742 Da / 分子数: 1 / 由来タイプ: 組換発現 / 由来: (組換発現)  Homo sapiens (ヒト) / 遺伝子: CSNK2A1, CK2A1 / 発現宿主: Homo sapiens (ヒト) / 遺伝子: CSNK2A1, CK2A1 / 発現宿主:  参照: UniProt: P68400, non-specific serine/threonine protein kinase | ||||
|---|---|---|---|---|---|
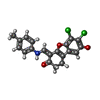
| #2: 化合物 | ChemComp-8QH / ( | ||||
| #3: 化合物 | | #4: 化合物 | ChemComp-GOL / #5: 水 | ChemComp-HOH / | |
-実験情報
-実験
| 実験 | 手法:  X線回折 / 使用した結晶の数: 1 X線回折 / 使用した結晶の数: 1 |
|---|
- 試料調製
試料調製
| 結晶 | マシュー密度: 1.93 Å3/Da / 溶媒含有率: 36.38 % |
|---|---|
| 結晶化 | 温度: 293 K / 手法: 蒸気拡散法, シッティングドロップ法 / pH: 5.6 詳細: Prior to the crystallization TF was solubilized in 100 % DMSO in a concentration of 10 mM. TF was mixed with human CK2alpha (construct 1-335; 8-10 mg/ml in 500 mM sodium chloride, 25 mM ...詳細: Prior to the crystallization TF was solubilized in 100 % DMSO in a concentration of 10 mM. TF was mixed with human CK2alpha (construct 1-335; 8-10 mg/ml in 500 mM sodium chloride, 25 mM Tris/HCl pH 8.5) in a ratio of 1:5. After a short time of incubation this mixture was mixed with reservoir solution [32 % (w/v) PEG4000, 0.2 M ammonium acetate, 0.1 M citrate pH 5.6] in a ratio of 5:2. 3.5 microliter of the resulting mixture was then equilibrated against the reservoir solution. The crystal growth was induced by seeding with 150 nanoliter seed suspension after an equilibration time of two days. |
-データ収集
| 回折 | 平均測定温度: 100 K |
|---|---|
| 放射光源 | 由来:  シンクロトロン / サイト: シンクロトロン / サイト:  SLS SLS  / ビームライン: X06DA / 波長: 0.99987 Å / ビームライン: X06DA / 波長: 0.99987 Å |
| 検出器 | タイプ: DECTRIS PILATUS 2M / 検出器: PIXEL / 日付: 2012年10月12日 |
| 放射 | プロトコル: SINGLE WAVELENGTH / 単色(M)・ラウエ(L): M / 散乱光タイプ: x-ray |
| 放射波長 | 波長: 0.99987 Å / 相対比: 1 |
| 反射 | 解像度: 1.79→36.14 Å / Num. obs: 27738 / % possible obs: 95.1 % / 冗長度: 3.5 % / CC1/2: 0.998 / Rmerge(I) obs: 0.056 / Rsym value: 0.056 / Net I/σ(I): 11.3 |
| 反射 シェル | 解像度: 1.79→1.83 Å / 冗長度: 2.9 % / Rmerge(I) obs: 0.545 / Mean I/σ(I) obs: 1.5 / Num. unique obs: 1127 / CC1/2: 0.76 / Rsym value: 0.545 / % possible all: 65.9 |
- 解析
解析
| ソフトウェア |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 精密化 | 構造決定の手法:  分子置換 分子置換開始モデル: 2PVR 解像度: 1.79→36.135 Å / SU ML: 0.18 / 交差検証法: FREE R-VALUE / σ(F): 1.39 / 位相誤差: 21.34
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 溶媒の処理 | 減衰半径: 0.9 Å / VDWプローブ半径: 1.11 Å | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 精密化ステップ | サイクル: LAST / 解像度: 1.79→36.135 Å
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 拘束条件 |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| LS精密化 シェル |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 精密化 TLS | 手法: refined / Refine-ID: X-RAY DIFFRACTION
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 精密化 TLSグループ |
|
 ムービー
ムービー コントローラー
コントローラー























 PDBj
PDBj