[English] 日本語
 Yorodumi
Yorodumi- PDB-4ysz: Crystal structure of Mitochondrial rhodoquinol-fumarate reductase... -
+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 4ysz | ||||||
|---|---|---|---|---|---|---|---|
| Title | Crystal structure of Mitochondrial rhodoquinol-fumarate reductase from Ascaris suum with 2-iodo-N-[3-(1-methylethoxy)phenyl]benzamide | ||||||
 Components Components |
| ||||||
 Keywords Keywords | OXIDOREDUCTASE/OXIDOREDUCTASE INHIBITOR / rhodoquinol-fumarate reductase / Complex II / OXIDOREDUCTASE-OXIDOREDUCTASE INHIBITOR complex | ||||||
| Function / homology |  Function and homology information Function and homology informationrespiratory chain complex II (succinate dehydrogenase) / mitochondrial electron transport, succinate to ubiquinone / succinate dehydrogenase (quinone) activity / succinate dehydrogenase / 3 iron, 4 sulfur cluster binding / ubiquinone binding / tricarboxylic acid cycle / mitochondrial membrane / 2 iron, 2 sulfur cluster binding / 4 iron, 4 sulfur cluster binding ...respiratory chain complex II (succinate dehydrogenase) / mitochondrial electron transport, succinate to ubiquinone / succinate dehydrogenase (quinone) activity / succinate dehydrogenase / 3 iron, 4 sulfur cluster binding / ubiquinone binding / tricarboxylic acid cycle / mitochondrial membrane / 2 iron, 2 sulfur cluster binding / 4 iron, 4 sulfur cluster binding / electron transfer activity / mitochondrial inner membrane / heme binding / metal ion binding Similarity search - Function | ||||||
| Biological species |  Ascaris suum (pig roundworm) Ascaris suum (pig roundworm) | ||||||
| Method |  X-RAY DIFFRACTION / X-RAY DIFFRACTION /  SYNCHROTRON / Resolution: 3.3 Å SYNCHROTRON / Resolution: 3.3 Å | ||||||
 Authors Authors | Harada, S. / Shiba, T. / Sato, D. / Yamamoto, A. / Nagahama, M. / Yone, A. / Inaoka, D.K. / Sakamoto, K. / Inoue, M. / Honma, T. / Kita, K. | ||||||
 Citation Citation |  Journal: Int J Mol Sci / Year: 2015 Journal: Int J Mol Sci / Year: 2015Title: Structural Insights into the Molecular Design of Flutolanil Derivatives Targeted for Fumarate Respiration of Parasite Mitochondria Authors: Inaoka, D.K. / Shiba, T. / Sato, D. / Balogun, E.O. / Sasaki, T. / Nagahama, M. / Oda, M. / Matsuoka, S. / Ohmori, J. / Honma, T. / Inoue, M. / Kita, K. / Harada, S. | ||||||
| History |
|
- Structure visualization
Structure visualization
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  4ysz.cif.gz 4ysz.cif.gz | 471.4 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb4ysz.ent.gz pdb4ysz.ent.gz | 376.7 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  4ysz.json.gz 4ysz.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Summary document |  4ysz_validation.pdf.gz 4ysz_validation.pdf.gz | 2.4 MB | Display |  wwPDB validaton report wwPDB validaton report |
|---|---|---|---|---|
| Full document |  4ysz_full_validation.pdf.gz 4ysz_full_validation.pdf.gz | 2.4 MB | Display | |
| Data in XML |  4ysz_validation.xml.gz 4ysz_validation.xml.gz | 93.6 KB | Display | |
| Data in CIF |  4ysz_validation.cif.gz 4ysz_validation.cif.gz | 118.2 KB | Display | |
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/ys/4ysz https://data.pdbj.org/pub/pdb/validation_reports/ys/4ysz ftp://data.pdbj.org/pub/pdb/validation_reports/ys/4ysz ftp://data.pdbj.org/pub/pdb/validation_reports/ys/4ysz | HTTPS FTP |
-Related structure data
| Related structure data |  3abvC  3ae7C  3ae9C  3aeaC  4ysxC  4ysyC  4yt0C  4ytmC  4ytpC  4yxdC  5c2tC C: citing same article ( |
|---|---|
| Similar structure data |
- Links
Links
- Assembly
Assembly
| Deposited unit | 
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | 
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 2 | 
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Unit cell |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Noncrystallographic symmetry (NCS) | NCS domain:
NCS domain segments:
NCS ensembles :
NCS oper:
|
- Components
Components
-Protein , 2 types, 4 molecules AECG
| #1: Protein | Mass: 71392.203 Da / Num. of mol.: 2 / Source method: isolated from a natural source / Source: (natural)  Ascaris suum (pig roundworm) / References: UniProt: U1LRQ3 Ascaris suum (pig roundworm) / References: UniProt: U1LRQ3#3: Protein | Mass: 21168.160 Da / Num. of mol.: 2 / Source method: isolated from a natural source / Source: (natural)  Ascaris suum (pig roundworm) / References: UniProt: P92506, UniProt: F1LC27*PLUS Ascaris suum (pig roundworm) / References: UniProt: P92506, UniProt: F1LC27*PLUS |
|---|
-Succinate dehydrogenase [ubiquinone] ... , 2 types, 4 molecules BFDH
| #2: Protein | Mass: 31673.951 Da / Num. of mol.: 2 / Source method: isolated from a natural source / Source: (natural)  Ascaris suum (pig roundworm) / References: UniProt: O44074, succinate dehydrogenase Ascaris suum (pig roundworm) / References: UniProt: O44074, succinate dehydrogenase#4: Protein | Mass: 17014.932 Da / Num. of mol.: 2 / Source method: isolated from a natural source / Source: (natural)  Ascaris suum (pig roundworm) / References: UniProt: P92507 Ascaris suum (pig roundworm) / References: UniProt: P92507 |
|---|
-Non-polymers , 9 types, 31 molecules 





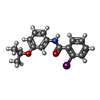









| #5: Chemical | | #6: Chemical | #7: Chemical | #8: Chemical | #9: Chemical | #10: Chemical | #11: Chemical | #12: Chemical | #13: Water | ChemComp-HOH / | |
|---|
-Details
| Has protein modification | Y |
|---|
-Experimental details
-Experiment
| Experiment | Method:  X-RAY DIFFRACTION / Number of used crystals: 1 X-RAY DIFFRACTION / Number of used crystals: 1 |
|---|
- Sample preparation
Sample preparation
| Crystal | Density Matthews: 3.05 Å3/Da / Density % sol: 59.72 % |
|---|---|
| Crystal grow | Temperature: 293 K / Method: microdialysis / pH: 8.4 Details: 15% (W/V) PEG 3350, 100MM TRIS-HCL, 200MM NACL, 1MM SODIUM MALONATE, 0.06% (W/V) C12E8, 0.04% (W/V) C12M |
-Data collection
| Diffraction | Mean temperature: 100 K |
|---|---|
| Diffraction source | Source:  SYNCHROTRON / Site: SYNCHROTRON / Site:  Photon Factory Photon Factory  / Beamline: AR-NW12A / Wavelength: 1 Å / Beamline: AR-NW12A / Wavelength: 1 Å |
| Detector | Type: ADSC QUANTUM 210r / Detector: CCD / Date: Dec 8, 2013 |
| Radiation | Monochromator: double-crystal Si(111) / Protocol: SINGLE WAVELENGTH / Monochromatic (M) / Laue (L): M / Scattering type: x-ray |
| Radiation wavelength | Wavelength: 1 Å / Relative weight: 1 |
| Reflection | Resolution: 3.3→50 Å / Num. obs: 52914 / % possible obs: 98.2 % / Redundancy: 5.5 % / Rmerge(I) obs: 0.137 / Net I/σ(I): 5.9 |
| Reflection shell | Resolution: 3.3→3.36 Å / Redundancy: 4.7 % / Rmerge(I) obs: 0.449 / Mean I/σ(I) obs: 3.3 / % possible all: 93.7 |
- Processing
Processing
| Software |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Refinement | Resolution: 3.3→29.82 Å / Cor.coef. Fo:Fc: 0.927 / Cor.coef. Fo:Fc free: 0.855 / SU B: 22.287 / SU ML: 0.363 / Cross valid method: THROUGHOUT / ESU R Free: 0.519 / Stereochemistry target values: MAXIMUM LIKELIHOOD / Details: HYDROGENS HAVE BEEN ADDED IN THE RIDING POSITIONS
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Solvent computation | Ion probe radii: 0.8 Å / Shrinkage radii: 0.8 Å / VDW probe radii: 1.2 Å / Solvent model: MASK | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Displacement parameters | Biso mean: 62.04 Å2
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refinement step | Cycle: LAST / Resolution: 3.3→29.82 Å
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refine LS restraints |
|
 Movie
Movie Controller
Controller






















 PDBj
PDBj



















