[English] 日本語
 Yorodumi
Yorodumi- PDB-4dvt: Crystal structure of clade A/E 93TH057 HIV-1 gp120 core in comple... -
+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 4dvt | ||||||
|---|---|---|---|---|---|---|---|
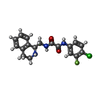
| Title | Crystal structure of clade A/E 93TH057 HIV-1 gp120 core in complex with AS-II-37 | ||||||
 Components Components | clade A/E 93TH057 HIV-1 gp120 core | ||||||
 Keywords Keywords | viral protein/transcription inhibitor / HIV-1 gp120 / small molecule inhibitor / CD4 binding site / AS-II-37 / viral protein-transcription inhibitor complex | ||||||
| Function / homology |  Function and homology information Function and homology informationmembrane fusion involved in viral entry into host cell / viral envelope / symbiont entry into host cell / virion attachment to host cell / virion membrane Similarity search - Function | ||||||
| Biological species |   Human immunodeficiency virus 1 Human immunodeficiency virus 1 | ||||||
| Method |  X-RAY DIFFRACTION / X-RAY DIFFRACTION /  SYNCHROTRON / SYNCHROTRON /  MOLECULAR REPLACEMENT / Resolution: 2.4 Å MOLECULAR REPLACEMENT / Resolution: 2.4 Å | ||||||
 Authors Authors | Kwon, Y.D. / Kwong, P.D. | ||||||
 Citation Citation |  Journal: Plos One / Year: 2014 Journal: Plos One / Year: 2014Title: Crystal Structures of HIV-1 gp120 Envelope Glycoprotein in Complex with NBD Analogues That Target the CD4-Binding Site. Authors: Kwon, Y.D. / Lalonde, J.M. / Yang, Y. / Elban, M.A. / Sugawara, A. / Courter, J.R. / Jones, D.M. / Smith, A.B. / Debnath, A.K. / Kwong, P.D. | ||||||
| History |
|
- Structure visualization
Structure visualization
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  4dvt.cif.gz 4dvt.cif.gz | 296.5 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb4dvt.ent.gz pdb4dvt.ent.gz | 244 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  4dvt.json.gz 4dvt.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Summary document |  4dvt_validation.pdf.gz 4dvt_validation.pdf.gz | 892.1 KB | Display |  wwPDB validaton report wwPDB validaton report |
|---|---|---|---|---|
| Full document |  4dvt_full_validation.pdf.gz 4dvt_full_validation.pdf.gz | 916.4 KB | Display | |
| Data in XML |  4dvt_validation.xml.gz 4dvt_validation.xml.gz | 31.1 KB | Display | |
| Data in CIF |  4dvt_validation.cif.gz 4dvt_validation.cif.gz | 41.1 KB | Display | |
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/dv/4dvt https://data.pdbj.org/pub/pdb/validation_reports/dv/4dvt ftp://data.pdbj.org/pub/pdb/validation_reports/dv/4dvt ftp://data.pdbj.org/pub/pdb/validation_reports/dv/4dvt | HTTPS FTP |
-Related structure data
| Related structure data |  4dvrC  4dvsC  4dvvC  4dvwC  4dvxC  3tgtS  4dvu C: citing same article ( S: Starting model for refinement |
|---|---|
| Similar structure data |
- Links
Links
- Assembly
Assembly
| Deposited unit | 
| ||||||||
|---|---|---|---|---|---|---|---|---|---|
| 1 | 
| ||||||||
| 2 | 
| ||||||||
| Unit cell |
| ||||||||
| Details | monomer |
- Components
Components
| #1: Protein | Mass: 39160.367 Da / Num. of mol.: 2 / Mutation: S375H Source method: isolated from a genetically manipulated source Source: (gene. exp.)   Human immunodeficiency virus 1 / Gene: HIV-1 Env / Plasmid: pVRC8400 / Cell line (production host): 293F / Production host: Human immunodeficiency virus 1 / Gene: HIV-1 Env / Plasmid: pVRC8400 / Cell line (production host): 293F / Production host:  Homo sapiens (human) / References: UniProt: A0A0M3KKW9 Homo sapiens (human) / References: UniProt: A0A0M3KKW9#2: Sugar | ChemComp-NAG / #3: Chemical | #4: Chemical | #5: Water | ChemComp-HOH / | Has protein modification | Y | Sequence details | THE CRYSTALLIZED SEQUENCE WAS CONSTRUCTED BY DELETION OF N-TERMINAL 1-43, BY REPLACING THE V1/V2 ...THE CRYSTALLIZ | |
|---|
-Experimental details
-Experiment
| Experiment | Method:  X-RAY DIFFRACTION / Number of used crystals: 1 X-RAY DIFFRACTION / Number of used crystals: 1 |
|---|
- Sample preparation
Sample preparation
| Crystal | Density Matthews: 2.66 Å3/Da / Density % sol: 53.73 % |
|---|---|
| Crystal grow | Temperature: 293 K / Method: vapor diffusion, hanging drop / pH: 7.5 Details: 10% PEG 8000, 5% iso-propanol, 0.1M HEPES, pH 7.5, VAPOR DIFFUSION, HANGING DROP, temperature 293K |
-Data collection
| Diffraction | Mean temperature: 100 K |
|---|---|
| Diffraction source | Source:  SYNCHROTRON / Site: SYNCHROTRON / Site:  APS APS  / Beamline: 22-BM / Wavelength: 1 / Beamline: 22-BM / Wavelength: 1 |
| Detector | Type: MAR scanner 300 mm plate / Detector: IMAGE PLATE / Date: Jun 6, 2011 |
| Radiation | Protocol: SINGLE WAVELENGTH / Monochromatic (M) / Laue (L): M / Scattering type: x-ray |
| Radiation wavelength | Wavelength: 1 Å / Relative weight: 1 |
| Reflection | Resolution: 2.4→50 Å / Num. all: 29562 / Num. obs: 27759 / % possible obs: 93.9 % / Observed criterion σ(F): 0 / Observed criterion σ(I): 0 / Redundancy: 3 % / Biso Wilson estimate: 45.16 Å2 / Rmerge(I) obs: 0.071 / Rsym value: 0.085 / Net I/σ(I): 17.6 |
| Reflection shell | Resolution: 2.5→2.54 Å / Redundancy: 1.8 % / Rmerge(I) obs: 0.432 / Mean I/σ(I) obs: 1.46 / Rsym value: 0.485 / % possible all: 60 |
- Processing
Processing
| Software |
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Refinement | Method to determine structure:  MOLECULAR REPLACEMENT MOLECULAR REPLACEMENTStarting model: 3TGT Resolution: 2.4→28.192 Å / Occupancy max: 1 / Occupancy min: 1 / SU ML: 0.33 / σ(F): 1.42 / Phase error: 30.27 / Stereochemistry target values: ML
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Solvent computation | Shrinkage radii: 0.9 Å / VDW probe radii: 1.11 Å / Solvent model: FLAT BULK SOLVENT MODEL | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Displacement parameters | Biso mean: 65.4336 Å2
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refinement step | Cycle: LAST / Resolution: 2.4→28.192 Å
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refine LS restraints |
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| LS refinement shell |
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refinement TLS params. | Method: refined / Refine-ID: X-RAY DIFFRACTION
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refinement TLS group |
|
 Movie
Movie Controller
Controller





















 PDBj
PDBj