[English] 日本語
 Yorodumi
Yorodumi- PDB-3zf5: Phage dUTPases control transfer of virulence genes by a proto-onc... -
+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 3zf5 | ||||||
|---|---|---|---|---|---|---|---|
| Title | Phage dUTPases control transfer of virulence genes by a proto-oncogenic G protein-like mechanism. (Staphylococcus bacteriophage 80alpha dUTPase Y84F mutant with dUpNHpp). | ||||||
 Components Components | DUTPASE | ||||||
 Keywords Keywords | HYDROLASE / PATHOGENICITY ISLAND / SAPI INDUCTION / GENE TRANSFER / MOONLIGHTING PROTEINS / G-PROTEIN / P-LOOP | ||||||
| Function / homology |  Function and homology information Function and homology informationdUTP catabolic process / dUMP biosynthetic process / dUTP diphosphatase / dUTP diphosphatase activity / magnesium ion binding Similarity search - Function | ||||||
| Biological species |  STAPHYLOCOCCUS PHAGE 80ALPHA (virus) STAPHYLOCOCCUS PHAGE 80ALPHA (virus) | ||||||
| Method |  X-RAY DIFFRACTION / X-RAY DIFFRACTION /  MOLECULAR REPLACEMENT / Resolution: 3.2 Å MOLECULAR REPLACEMENT / Resolution: 3.2 Å | ||||||
 Authors Authors | Tormo-Mas, M.A. / Donderis, J. / Garcia-Caballer, M. / Alt, A. / Mir-Sanchis, I. / Marina, A. / Penades, J.R. | ||||||
 Citation Citation |  Journal: Mol.Cell / Year: 2013 Journal: Mol.Cell / Year: 2013Title: Phage Dutpases Control Transfer of Virulence Genes by a Proto-Oncogenic G Protein-Like Mechanism. Authors: Tormo-Mas, M.A. / Donderis, J. / Garcia-Caballer, M. / Alt, A. / Mir-Sanchis, I. / Marina, A. / Penades, J.R. #1: Journal: Nature / Year: 2010 Title: Moonlighting Bacteriophage Proteins Derepress Staphylococcal Pathogenicity Islands. Authors: Tormo-Mas, M.A. / Mir, I. / Shrestha, A. / Tallent, S.M. / Campoy, S. / Lasa, I. / Barbe, J. / Novick, R.P. / Christie, G.E. / Penades, J.R. | ||||||
| History |
|
- Structure visualization
Structure visualization
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  3zf5.cif.gz 3zf5.cif.gz | 50.2 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb3zf5.ent.gz pdb3zf5.ent.gz | 34 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  3zf5.json.gz 3zf5.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/zf/3zf5 https://data.pdbj.org/pub/pdb/validation_reports/zf/3zf5 ftp://data.pdbj.org/pub/pdb/validation_reports/zf/3zf5 ftp://data.pdbj.org/pub/pdb/validation_reports/zf/3zf5 | HTTPS FTP |
|---|
-Related structure data
| Related structure data |  3zezSC  3zf0C  3zf1C  3zf2C  3zf3C  3zf4C  3zf6C S: Starting model for refinement C: citing same article ( |
|---|---|
| Similar structure data |
- Links
Links
- Assembly
Assembly
| Deposited unit | 
| |||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | 
| |||||||||||||||
| Unit cell |
| |||||||||||||||
| Components on special symmetry positions |
|
- Components
Components
| #1: Protein | Mass: 22651.627 Da / Num. of mol.: 1 / Mutation: YES Source method: isolated from a genetically manipulated source Details: STRUCTURE IN PRESENCE OF DUMP / Source: (gene. exp.)  STAPHYLOCOCCUS PHAGE 80ALPHA (virus) / Production host: STAPHYLOCOCCUS PHAGE 80ALPHA (virus) / Production host:  | ||||
|---|---|---|---|---|---|
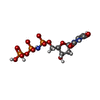
| #2: Chemical | ChemComp-DUP / | ||||
| #3: Chemical | | #4: Chemical | ChemComp-MG / | #5: Water | ChemComp-HOH / | |
-Experimental details
-Experiment
| Experiment | Method:  X-RAY DIFFRACTION X-RAY DIFFRACTION |
|---|
- Sample preparation
Sample preparation
| Crystal | Density Matthews: 2.5 Å3/Da / Density % sol: 50.75 % / Description: NONE |
|---|---|
| Crystal grow | Details: 2-8% TERT-BUTANOL, 0.1M TRIS (PH 8.5), 30-50% MPD PR PEG400. |
-Data collection
| Diffraction | Mean temperature: 110 K |
|---|---|
| Diffraction source | Source:  ROTATING ANODE / Wavelength: 1.54 ROTATING ANODE / Wavelength: 1.54 |
| Radiation | Protocol: SINGLE WAVELENGTH / Monochromatic (M) / Laue (L): M / Scattering type: x-ray |
| Radiation wavelength | Wavelength: 1.54 Å / Relative weight: 1 |
| Reflection | Resolution: 3→14.9 Å / Num. obs: 4719 / % possible obs: 98.5 % / Observed criterion σ(I): 2 / Redundancy: 4.4 % / Rmerge(I) obs: 0.17 / Net I/σ(I): 0.35 |
| Reflection shell | Resolution: 3→3.16 Å / Redundancy: 4.4 % / Rmerge(I) obs: 0.35 / Mean I/σ(I) obs: 2.3 / % possible all: 100 |
- Processing
Processing
| Software |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Refinement | Method to determine structure:  MOLECULAR REPLACEMENT MOLECULAR REPLACEMENTStarting model: PDB ENTRY 3ZEZ Resolution: 3.2→14.71 Å / Cor.coef. Fo:Fc: 0.878 / Cor.coef. Fo:Fc free: 0.807 / SU B: 15.769 / SU ML: 0.267 / Cross valid method: THROUGHOUT / ESU R Free: 0.126 / Stereochemistry target values: MAXIMUM LIKELIHOOD Details: HYDROGENS HAVE BEEN ADDED IN THE RIDING POSITIONS. INITIAL MET IS NOT TRACED. RESIDUES F115, K117, E118 AND E159 ARE TRACED AS A BECAUSE OF LACK OF ELECTRON DENSITY.
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Solvent computation | Ion probe radii: 0.8 Å / Shrinkage radii: 0.8 Å / VDW probe radii: 1.2 Å / Solvent model: MASK | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Displacement parameters | Biso mean: 29.684 Å2
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refinement step | Cycle: LAST / Resolution: 3.2→14.71 Å
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refine LS restraints |
|
 Movie
Movie Controller
Controller









 PDBj
PDBj