[English] 日本語
 Yorodumi
Yorodumi- PDB-3qz4: Crystal structure of an Endo-1,4-beta-xylanase D (BT_3675) from B... -
+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 3qz4 | ||||||
|---|---|---|---|---|---|---|---|
| Title | Crystal structure of an Endo-1,4-beta-xylanase D (BT_3675) from Bacteroides thetaiotaomicron VPI-5482 at 1.74 A resolution | ||||||
 Components Components | Endo-1,4-beta-xylanase D | ||||||
 Keywords Keywords | HYDROLASE / 5-BLADED BETA-PROPELLER FOLD / XYLAN DEGRADATION / STRUCTURAL GENOMICS / JOINT CENTER FOR STRUCTURAL GENOMICS / JCSG / PROTEIN STRUCTURE INITIATIVE / PSI-BIOLOGY | ||||||
| Function / homology |  Function and homology information Function and homology informationxylan catabolic process / hydrolase activity, hydrolyzing O-glycosyl compounds Similarity search - Function | ||||||
| Biological species |  Bacteroides thetaiotaomicron (bacteria) Bacteroides thetaiotaomicron (bacteria) | ||||||
| Method |  X-RAY DIFFRACTION / X-RAY DIFFRACTION /  SYNCHROTRON / SYNCHROTRON /  MAD / Resolution: 1.74 Å MAD / Resolution: 1.74 Å | ||||||
 Authors Authors | Joint Center for Structural Genomics (JCSG) | ||||||
 Citation Citation |  Journal: To be published Journal: To be publishedTitle: Crystal structure of an Endo-1,4-beta-xylanase D (BT_3675) from Bacteroides thetaiotaomicron VPI-5482 at 1.74 A resolution Authors: Joint Center for Structural Genomics (JCSG) | ||||||
| History |
|
- Structure visualization
Structure visualization
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  3qz4.cif.gz 3qz4.cif.gz | 276.5 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb3qz4.ent.gz pdb3qz4.ent.gz | 223.6 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  3qz4.json.gz 3qz4.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/qz/3qz4 https://data.pdbj.org/pub/pdb/validation_reports/qz/3qz4 ftp://data.pdbj.org/pub/pdb/validation_reports/qz/3qz4 ftp://data.pdbj.org/pub/pdb/validation_reports/qz/3qz4 | HTTPS FTP |
|---|
-Related structure data
| Similar structure data | |
|---|---|
| Other databases |
- Links
Links
- Assembly
Assembly
| Deposited unit | 
| ||||||||
|---|---|---|---|---|---|---|---|---|---|
| 1 | 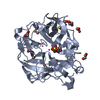
| ||||||||
| 2 | 
| ||||||||
| Unit cell |
| ||||||||
| Details | ANALYTICAL SIZE EXCLUSION CHROMATOGRAPHY PROVIDES SUPPORTING EVIDENCE THAT A MONOMER IS A SIGNIFICANT OLIGOMERIZATION STATE IN SOLUTION. |
- Components
Components
| #1: Protein | Mass: 35159.863 Da / Num. of mol.: 2 / Fragment: sequence database residues 24-333 Source method: isolated from a genetically manipulated source Source: (gene. exp.)  Bacteroides thetaiotaomicron (bacteria) Bacteroides thetaiotaomicron (bacteria)Gene: BT_3675 / Plasmid: SpeedET / Production host:  #2: Chemical | ChemComp-UNX / #3: Chemical | #4: Chemical | ChemComp-EDO / #5: Water | ChemComp-HOH / | Has protein modification | Y | Sequence details | THE CONSTRUCT WAS EXPRESSED WITH A PURIFICATION TAG MGSDKIHHHHHHENLYFQG. THE TAG WAS REMOVED WITH ...THE CONSTRUCT WAS EXPRESSED WITH A PURIFICATI | |
|---|
-Experimental details
-Experiment
| Experiment | Method:  X-RAY DIFFRACTION / Number of used crystals: 1 X-RAY DIFFRACTION / Number of used crystals: 1 |
|---|
- Sample preparation
Sample preparation
| Crystal | Density Matthews: 2.32 Å3/Da / Density % sol: 47.09 % |
|---|---|
| Crystal grow | Temperature: 277 K / Method: vapor diffusion, sitting drop / pH: 8 Details: 20.40% polyethylene glycol 8000, 0.1M HEPES pH 8.0, NANODROP, VAPOR DIFFUSION, SITTING DROP, temperature 277K |
-Data collection
| Diffraction | Mean temperature: 100 K | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Diffraction source | Source:  SYNCHROTRON / Site: SYNCHROTRON / Site:  SSRL SSRL  / Beamline: BL14-1 / Wavelength: 0.95369,0.97915,0.97901 / Beamline: BL14-1 / Wavelength: 0.95369,0.97915,0.97901 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Detector | Type: MARMOSAIC 325 mm CCD / Detector: CCD / Date: Jul 25, 2010 Details: Vertical focusing mirror; double crystal Si(111) monochromator | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Radiation | Monochromator: double crystal Si(111) / Protocol: MAD / Monochromatic (M) / Laue (L): M / Scattering type: x-ray | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Radiation wavelength |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Reflection | Resolution: 1.74→29.865 Å / Num. all: 65872 / Num. obs: 65872 / % possible obs: 100 % / Redundancy: 4.2 % / Biso Wilson estimate: 22.863 Å2 / Rsym value: 0.084 / Net I/σ(I): 8.9 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Reflection shell | Diffraction-ID: 1
|
-Phasing
| Phasing | Method:  MAD MAD |
|---|
- Processing
Processing
| Software |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Refinement | Method to determine structure:  MAD / Resolution: 1.74→29.865 Å / Cor.coef. Fo:Fc: 0.9622 / Cor.coef. Fo:Fc free: 0.9419 / Occupancy max: 1 / Occupancy min: 0.5 / Cross valid method: THROUGHOUT / σ(F): 0 MAD / Resolution: 1.74→29.865 Å / Cor.coef. Fo:Fc: 0.9622 / Cor.coef. Fo:Fc free: 0.9419 / Occupancy max: 1 / Occupancy min: 0.5 / Cross valid method: THROUGHOUT / σ(F): 0 Details: 1. A MET-INHIBITION PROTOCOL WAS USED FOR SELENOMETHIONINE INCORPORATION DURING PROTEIN EXPRESSION. THE OCCUPANCY OF THE SE ATOMS IN THE MSE RESIDUES WAS REDUCED TO 0.75 TO ACCOUNT FOR THE ...Details: 1. A MET-INHIBITION PROTOCOL WAS USED FOR SELENOMETHIONINE INCORPORATION DURING PROTEIN EXPRESSION. THE OCCUPANCY OF THE SE ATOMS IN THE MSE RESIDUES WAS REDUCED TO 0.75 TO ACCOUNT FOR THE REDUCED SCATTERING POWER DUE TO PARTIAL S-MET INCORPORATION. 2. ATOM RECORD CONTAINS SUM OF TLS AND RESIDUAL B FACTORS. ANISOU RECORD CONTAINS SUM OF TLS AND RESIDUAL U FACTORS. 3. ETHYLENE GLYCOLS (EDO) AND HEPES (EPE) FROM THE CRYSTALLIZATION/CRYO CONDITIONS HAVE BEEN MODELED IN THE STRUCTURE. 4. THE FIRST FIVE RESIDUES OF EACH PROTOMER ARE DISODERED AND WERE NOT MODELED. 5. TWO UNKNOWN IONS (UNX) HAVE BEEN MODELED IN EACH PROTOMER. X-RAY FLUORESCENCE EMISSION SPECTRA, COORDINATION GEOMETRY ANDANOMALOUS SIGNAL AT THE MAD WAVELENGTHS WERE INCONCLUSIVE AS TO THE ION IDENTITIES. FOR THE SITE NEAR HIS-279, CALCIUM IS MODELED IN THE 1WL7 HOMOLOG AND A CHLORIDE IS MODELED IN THE 1GYH/1GYE HOMOLOG WITH A COMMENT IN THE PAPER THAT CHLORIDE IS ALSO A POSSIBILITY. THE UNKNOWN ION UNX-334 SHOWS COORDINATION DISTANCES LONGER THAN IS TYPICAL FOR CALCIUM BUT SHORTER THAN WOULD BE EXPECTED FOR CHLORIDE. THE UNKNOWN IONS UNX-335 MIGHT BE MAGNESIUM OR CALCIUM. HOWEVER, NEITHER ION WAS PRESENT IN THE PROTEIN BUFFER OR CRYSTALLIZATION REAGENT. 6. THE REFINEMENT WAS RESTRAINED AGAINST THE MAD PHASES.
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Displacement parameters | Biso max: 124.55 Å2 / Biso mean: 39.0161 Å2 / Biso min: 12.78 Å2
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refinement step | Cycle: LAST / Resolution: 1.74→29.865 Å
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refine LS restraints |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| LS refinement shell | Resolution: 1.74→1.78 Å / Total num. of bins used: 20
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refinement TLS params. | Method: refined / Refine-ID: X-RAY DIFFRACTION
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refinement TLS group |
|
 Movie
Movie Controller
Controller












 PDBj
PDBj






