[English] 日本語
 Yorodumi
Yorodumi- PDB-3nu9: Crystal Structure of HIV-1 Protease Mutant I84V with Antiviral Dr... -
+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 3nu9 | ||||||
|---|---|---|---|---|---|---|---|
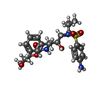
| Title | Crystal Structure of HIV-1 Protease Mutant I84V with Antiviral Drug Amprenavir | ||||||
 Components Components | Protease | ||||||
 Keywords Keywords | HYDROLASE/HYDROLASE INHIBITOR / enzyme inhibition / aspartic protease / HIV/AIDS / conformational change / Amprenavir / HYDROLASE / HYDROLASE-HYDROLASE INHIBITOR complex | ||||||
| Function / homology |  Function and homology information Function and homology informationHIV-1 retropepsin / symbiont-mediated activation of host apoptosis / retroviral ribonuclease H / exoribonuclease H / exoribonuclease H activity / DNA integration / viral genome integration into host DNA / establishment of integrated proviral latency / RNA-directed DNA polymerase / RNA stem-loop binding ...HIV-1 retropepsin / symbiont-mediated activation of host apoptosis / retroviral ribonuclease H / exoribonuclease H / exoribonuclease H activity / DNA integration / viral genome integration into host DNA / establishment of integrated proviral latency / RNA-directed DNA polymerase / RNA stem-loop binding / viral penetration into host nucleus / host multivesicular body / RNA-directed DNA polymerase activity / RNA-DNA hybrid ribonuclease activity / Transferases; Transferring phosphorus-containing groups; Nucleotidyltransferases / host cell / viral nucleocapsid / DNA recombination / DNA-directed DNA polymerase / aspartic-type endopeptidase activity / Hydrolases; Acting on ester bonds / DNA-directed DNA polymerase activity / symbiont-mediated suppression of host gene expression / viral translational frameshifting / symbiont entry into host cell / lipid binding / host cell plasma membrane / host cell nucleus / virion membrane / structural molecule activity / proteolysis / DNA binding / zinc ion binding Similarity search - Function | ||||||
| Biological species |   Human immunodeficiency virus 1 Human immunodeficiency virus 1 | ||||||
| Method |  X-RAY DIFFRACTION / X-RAY DIFFRACTION /  SYNCHROTRON / SYNCHROTRON /  MOLECULAR REPLACEMENT / Resolution: 1.85 Å MOLECULAR REPLACEMENT / Resolution: 1.85 Å | ||||||
 Authors Authors | Shen, C.H. / Weber, I.T. | ||||||
 Citation Citation |  Journal: Febs J. / Year: 2010 Journal: Febs J. / Year: 2010Title: Amprenavir complexes with HIV-1 protease and its drug-resistant mutants altering hydrophobic clusters. Authors: Shen, C.H. / Wang, Y.F. / Kovalevsky, A.Y. / Harrison, R.W. / Weber, I.T. | ||||||
| History |
|
- Structure visualization
Structure visualization
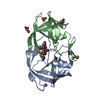
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  3nu9.cif.gz 3nu9.cif.gz | 56.1 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb3nu9.ent.gz pdb3nu9.ent.gz | 39.1 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  3nu9.json.gz 3nu9.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/nu/3nu9 https://data.pdbj.org/pub/pdb/validation_reports/nu/3nu9 ftp://data.pdbj.org/pub/pdb/validation_reports/nu/3nu9 ftp://data.pdbj.org/pub/pdb/validation_reports/nu/3nu9 | HTTPS FTP |
|---|
-Related structure data
| Related structure data | 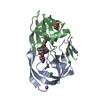 3nu3C  3nu4C  3nu5C  3nu6C  3nujC  3nuoC  2f8gS C: citing same article ( S: Starting model for refinement |
|---|---|
| Similar structure data |
- Links
Links
- Assembly
Assembly
| Deposited unit | 
| ||||||||
|---|---|---|---|---|---|---|---|---|---|
| 1 |
| ||||||||
| Unit cell |
|
- Components
Components
| #1: Protein | Mass: 10726.650 Da / Num. of mol.: 2 / Fragment: residues 501-599 / Mutation: Q7K, L33I, L63I, C67A, I84V, C95A Source method: isolated from a genetically manipulated source Source: (gene. exp.)   Human immunodeficiency virus 1 / Genus: Lentivirus / Gene: gag, pol / Plasmid: PET11A / Species (production host): Escherichia coli / Production host: Human immunodeficiency virus 1 / Genus: Lentivirus / Gene: gag, pol / Plasmid: PET11A / Species (production host): Escherichia coli / Production host:  #2: Chemical | ChemComp-478 / { | #3: Chemical | ChemComp-IOD / #4: Water | ChemComp-HOH / | |
|---|
-Experimental details
-Experiment
| Experiment | Method:  X-RAY DIFFRACTION / Number of used crystals: 1 X-RAY DIFFRACTION / Number of used crystals: 1 |
|---|
- Sample preparation
Sample preparation
| Crystal | Density Matthews: 2.74 Å3/Da / Density % sol: 55.08 % |
|---|---|
| Crystal grow | Temperature: 298 K / Method: vapor diffusion, hanging drop / pH: 4 Details: Crystal was grown by the hanging-drop vapor-diffusion method at room temperature, from a 2.2 mg/ml protein solution at pH 4.0 with 0.13M NaI, 0.1M NaOAc. The inhibitor was mixed with ...Details: Crystal was grown by the hanging-drop vapor-diffusion method at room temperature, from a 2.2 mg/ml protein solution at pH 4.0 with 0.13M NaI, 0.1M NaOAc. The inhibitor was mixed with protease in a ratio 5:1 , VAPOR DIFFUSION, HANGING DROP, temperature 298K |
-Data collection
| Diffraction | Mean temperature: 100 K |
|---|---|
| Diffraction source | Source:  SYNCHROTRON / Site: SYNCHROTRON / Site:  APS APS  / Beamline: 22-ID / Wavelength: 1 Å / Beamline: 22-ID / Wavelength: 1 Å |
| Detector | Type: MARMOSAIC 300 mm CCD / Detector: CCD / Date: Mar 28, 2008 |
| Radiation | Protocol: SINGLE WAVELENGTH / Monochromatic (M) / Laue (L): M / Scattering type: x-ray |
| Radiation wavelength | Wavelength: 1 Å / Relative weight: 1 |
| Reflection | Resolution: 1.85→50 Å / % possible obs: 93.2 % / Observed criterion σ(F): 0 / Observed criterion σ(I): 0 / Redundancy: 4 % / Biso Wilson estimate: 19.6 Å2 / Rmerge(I) obs: 0.097 |
| Reflection shell | Resolution: 1.85→1.9 Å / Redundancy: 3.6 % / Rmerge(I) obs: 0.345 / Mean I/σ(I) obs: 2.7 / Num. unique all: 1552 / % possible all: 76.6 |
- Processing
Processing
| Software |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Refinement | Method to determine structure:  MOLECULAR REPLACEMENT MOLECULAR REPLACEMENTStarting model: 2F8G Resolution: 1.85→9.97 Å / Cor.coef. Fo:Fc: 0.943 / Cor.coef. Fo:Fc free: 0.913 / SU B: 4.721 / SU ML: 0.082 / Cross valid method: THROUGHOUT / σ(F): 0 / ESU R Free: 0.138 / Stereochemistry target values: MAXIMUM LIKELIHOOD / Details: HYDROGENS HAVE BEEN ADDED IN THE RIDING POSITIONS
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Solvent computation | Ion probe radii: 0.8 Å / Shrinkage radii: 0.8 Å / VDW probe radii: 1.2 Å / Solvent model: MASK | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Displacement parameters | Biso mean: 27.34 Å2
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refinement step | Cycle: LAST / Resolution: 1.85→9.97 Å
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refine LS restraints |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| LS refinement shell | Resolution: 1.853→1.9 Å / Total num. of bins used: 20
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refinement TLS params. | Method: refined / Origin x: 15.386 Å / Origin y: 17.143 Å / Origin z: 18.907 Å
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refinement TLS group |
|
 Movie
Movie Controller
Controller


















 PDBj
PDBj