[English] 日本語
 Yorodumi
Yorodumi- PDB-3nfl: Crystal structure of the PTPN4 PDZ domain complexed with the C-te... -
+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 3nfl | ||||||
|---|---|---|---|---|---|---|---|
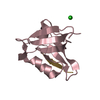
| Title | Crystal structure of the PTPN4 PDZ domain complexed with the C-terminus of the GluN2A NMDA receptor subunit | ||||||
 Components Components |
| ||||||
 Keywords Keywords | PROTEIN BINDING / PDZ-PDZ-binding site complex | ||||||
| Function / homology |  Function and homology information Function and homology informationexcitatory chemical synaptic transmission / Toll Like Receptor 4 (TLR4) Cascade / directional locomotion / Synaptic adhesion-like molecules / serotonin metabolic process / protein localization to postsynaptic membrane / Interleukin-37 signaling / sleep / Assembly and cell surface presentation of NMDA receptors / Neurexins and neuroligins ...excitatory chemical synaptic transmission / Toll Like Receptor 4 (TLR4) Cascade / directional locomotion / Synaptic adhesion-like molecules / serotonin metabolic process / protein localization to postsynaptic membrane / Interleukin-37 signaling / sleep / Assembly and cell surface presentation of NMDA receptors / Neurexins and neuroligins / protein dephosphorylation / regulation of monoatomic cation transmembrane transport / NMDA glutamate receptor activity / NMDA selective glutamate receptor complex / glutamate receptor signaling pathway / calcium ion transmembrane import into cytosol / startle response / dopamine metabolic process / non-membrane spanning protein tyrosine phosphatase activity / Negative regulation of NMDA receptor-mediated neuronal transmission / Unblocking of NMDA receptors, glutamate binding and activation / monoatomic cation transmembrane transport / Long-term potentiation / regulation of neuronal synaptic plasticity / glutamate receptor binding / MECP2 regulates neuronal receptors and channels / positive regulation of synaptic transmission, glutamatergic / cytoskeletal protein binding / protein-tyrosine-phosphatase / glutamate-gated calcium ion channel activity / protein tyrosine phosphatase activity / neurogenesis / sensory perception of pain / ionotropic glutamate receptor signaling pathway / cytoplasmic vesicle membrane / positive regulation of excitatory postsynaptic potential / sodium ion transmembrane transport / response to amphetamine / protein catabolic process / synaptic membrane / synaptic transmission, glutamatergic / excitatory postsynaptic potential / transmitter-gated monoatomic ion channel activity involved in regulation of postsynaptic membrane potential / negative regulation of protein catabolic process / brain development / visual learning / regulation of synaptic plasticity / response to wounding / postsynaptic density membrane / cytoplasmic side of plasma membrane / calcium ion transmembrane transport / memory / synaptic vesicle / long-term synaptic potentiation / amyloid-beta binding / presynaptic membrane / dendritic spine / chemical synaptic transmission / cytoskeleton / response to ethanol / learning or memory / postsynaptic membrane / postsynaptic density / positive regulation of apoptotic process / response to xenobiotic stimulus / endoplasmic reticulum membrane / glutamatergic synapse / cell surface / zinc ion binding / nucleoplasm / plasma membrane / cytoplasm / cytosol Similarity search - Function | ||||||
| Biological species |  Homo sapiens (human) Homo sapiens (human) | ||||||
| Method |  X-RAY DIFFRACTION / X-RAY DIFFRACTION /  SYNCHROTRON / SYNCHROTRON /  MOLECULAR REPLACEMENT / Resolution: 1.91 Å MOLECULAR REPLACEMENT / Resolution: 1.91 Å | ||||||
 Authors Authors | Babault, N. / Cordier, F. / Lafage, M. / Cockburn, J. / Haouz, A. / Rey, F.A. / Delepierre, M. / Buc, H. / Lafon, M. / Wolff, N. | ||||||
 Citation Citation |  Journal: Structure / Year: 2011 Journal: Structure / Year: 2011Title: Peptides Targeting the PDZ Domain of PTPN4 Are Efficient Inducers of Glioblastoma Cell Death. Authors: Babault, N. / Cordier, F. / Lafage, M. / Cockburn, J. / Haouz, A. / Prehaud, C. / Rey, F.A. / Delepierre, M. / Buc, H. / Lafon, M. / Wolff, N. | ||||||
| History |
|
- Structure visualization
Structure visualization
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  3nfl.cif.gz 3nfl.cif.gz | 166.3 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb3nfl.ent.gz pdb3nfl.ent.gz | 133.4 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  3nfl.json.gz 3nfl.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/nf/3nfl https://data.pdbj.org/pub/pdb/validation_reports/nf/3nfl ftp://data.pdbj.org/pub/pdb/validation_reports/nf/3nfl ftp://data.pdbj.org/pub/pdb/validation_reports/nf/3nfl | HTTPS FTP |
|---|
-Related structure data
| Related structure data |  3nfkC  2vphS S: Starting model for refinement C: citing same article ( |
|---|---|
| Similar structure data |
- Links
Links
- Assembly
Assembly
| Deposited unit | 
| ||||||||
|---|---|---|---|---|---|---|---|---|---|
| 1 | 
| ||||||||
| 2 | 
| ||||||||
| 3 | 
| ||||||||
| 4 | 
| ||||||||
| 5 | 
| ||||||||
| 6 | 
| ||||||||
| Unit cell |
|
- Components
Components
| #1: Protein | Mass: 11694.363 Da / Num. of mol.: 4 / Fragment: PDZ domain (UNP residues 499-604) Source method: isolated from a genetically manipulated source Source: (gene. exp.)  Homo sapiens (human) / Gene: PTPN4 / Plasmid: pDEST15 / Production host: Homo sapiens (human) / Gene: PTPN4 / Plasmid: pDEST15 / Production host:  #2: Protein/peptide | Mass: 1913.204 Da / Num. of mol.: 4 / Fragment: UNP residues 1449-1464 / Source method: obtained synthetically Details: NR2A16 peptide has been chemically synthetized. The sequence ocuurs naturally in Homo sapiens (Human) Source: (synth.)  Homo sapiens (human) / References: UniProt: Q12879 Homo sapiens (human) / References: UniProt: Q12879#3: Water | ChemComp-HOH / | Has protein modification | Y | |
|---|
-Experimental details
-Experiment
| Experiment | Method:  X-RAY DIFFRACTION / Number of used crystals: 1 X-RAY DIFFRACTION / Number of used crystals: 1 |
|---|
- Sample preparation
Sample preparation
| Crystal | Density Matthews: 2.34 Å3/Da / Density % sol: 47.38 % |
|---|---|
| Crystal grow | Temperature: 291 K / Method: vapor diffusion, hanging drop Details: 24% PEG 4000, 0.2M ammonium sulfate, VAPOR DIFFUSION, HANGING DROP, temperature 291K |
-Data collection
| Diffraction | Mean temperature: 100 K |
|---|---|
| Diffraction source | Source:  SYNCHROTRON / Site: SYNCHROTRON / Site:  SLS SLS  / Beamline: X06SA / Wavelength: 0.9795 Å / Beamline: X06SA / Wavelength: 0.9795 Å |
| Detector | Type: PSI PILATUS 6M / Detector: PIXEL / Date: Aug 8, 2009 |
| Radiation | Monochromator: LN2 cooled fixed-exit Si(111) Dynamically bendable mirror Protocol: SINGLE WAVELENGTH / Monochromatic (M) / Laue (L): M / Scattering type: x-ray |
| Radiation wavelength | Wavelength: 0.9795 Å / Relative weight: 1 |
| Reflection | Resolution: 1.91→95.45 Å / Num. all: 40560 / Num. obs: 38978 / % possible obs: 96.1 % / Redundancy: 4.2 % / Biso Wilson estimate: 37.4 Å2 / Rmerge(I) obs: 0.043 / Net I/σ(I): 21.1 |
| Reflection shell | Resolution: 1.91→2.01 Å / Redundancy: 2.1 % / Rmerge(I) obs: 0.309 / Mean I/σ(I) obs: 2 / Num. unique all: 4287 / % possible all: 74.7 |
- Processing
Processing
| Software |
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Refinement | Method to determine structure:  MOLECULAR REPLACEMENT MOLECULAR REPLACEMENTStarting model: PDB ENTRY 2VPH Resolution: 1.91→49.88 Å / Cor.coef. Fo:Fc: 0.9235 / Cor.coef. Fo:Fc free: 0.9025 / SU R Cruickshank DPI: 0.144 / Cross valid method: THROUGHOUT / σ(F): 0
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Displacement parameters | Biso mean: 54.07 Å2
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refine analyze | Luzzati coordinate error obs: 0.387 Å | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refinement step | Cycle: LAST / Resolution: 1.91→49.88 Å
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refine LS restraints |
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| LS refinement shell | Resolution: 1.91→1.96 Å / Total num. of bins used: 20
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refinement TLS params. | Method: refined / Refine-ID: X-RAY DIFFRACTION
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refinement TLS group |
|
 Movie
Movie Controller
Controller











 PDBj
PDBj
















