[English] 日本語
 Yorodumi
Yorodumi- PDB-3lbk: Structure of human MDM2 protein in complex with a small molecule ... -
+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 3lbk | ||||||
|---|---|---|---|---|---|---|---|
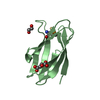
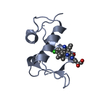
| Title | Structure of human MDM2 protein in complex with a small molecule inhibitor | ||||||
 Components Components | E3 ubiquitin-protein ligase Mdm2 | ||||||
 Keywords Keywords | LIGASE / MDMX / MDM2 / p53 / inhibitor / Alternative splicing / Cytoplasm / Nucleus / Phosphoprotein / Proto-oncogene / Ubl conjugation / Ubl conjugation pathway / Zinc-finger | ||||||
| Function / homology |  Function and homology information Function and homology informationcellular response to vitamin B1 / response to formaldehyde / response to water-immersion restraint stress / response to ether / traversing start control point of mitotic cell cycle / atrial septum development / fibroblast activation / regulation of protein catabolic process at postsynapse, modulating synaptic transmission / Trafficking of AMPA receptors / receptor serine/threonine kinase binding ...cellular response to vitamin B1 / response to formaldehyde / response to water-immersion restraint stress / response to ether / traversing start control point of mitotic cell cycle / atrial septum development / fibroblast activation / regulation of protein catabolic process at postsynapse, modulating synaptic transmission / Trafficking of AMPA receptors / receptor serine/threonine kinase binding / negative regulation of intrinsic apoptotic signaling pathway by p53 class mediator / negative regulation of protein processing / positive regulation of vascular associated smooth muscle cell migration / response to steroid hormone / SUMO transferase activity / peroxisome proliferator activated receptor binding / atrioventricular valve morphogenesis / AKT phosphorylates targets in the cytosol / response to iron ion / NEDD8 ligase activity / endocardial cushion morphogenesis / cellular response to peptide hormone stimulus / ventricular septum development / positive regulation of muscle cell differentiation / cardiac septum morphogenesis / regulation of postsynaptic neurotransmitter receptor internalization / SUMOylation of ubiquitinylation proteins / cellular response to alkaloid / blood vessel development / ligase activity / Constitutive Signaling by AKT1 E17K in Cancer / negative regulation of DNA damage response, signal transduction by p53 class mediator / cellular response to antibiotic / SUMOylation of transcription factors / negative regulation of signal transduction by p53 class mediator / regulation of protein catabolic process / cellular response to UV-C / cellular response to estrogen stimulus / protein sumoylation / response to magnesium ion / blood vessel remodeling / ribonucleoprotein complex binding / protein localization to nucleus / protein autoubiquitination / positive regulation of vascular associated smooth muscle cell proliferation / NPAS4 regulates expression of target genes / transcription repressor complex / positive regulation of mitotic cell cycle / regulation of heart rate / : / positive regulation of protein export from nucleus / response to cocaine / ubiquitin binding / DNA damage response, signal transduction by p53 class mediator / establishment of protein localization / sperm end piece / Stabilization of p53 / Regulation of RUNX3 expression and activity / cellular response to gamma radiation / RING-type E3 ubiquitin transferase / Oncogene Induced Senescence / Regulation of TP53 Activity through Methylation / Degradation of CDH1 / cellular response to growth factor stimulus / protein destabilization / response to toxic substance / centriolar satellite / cellular response to hydrogen peroxide / disordered domain specific binding / protein polyubiquitination / p53 binding / ubiquitin-protein transferase activity / endocytic vesicle membrane / Signaling by ALK fusions and activated point mutants / Regulation of TP53 Degradation / ubiquitin protein ligase activity / positive regulation of proteasomal ubiquitin-dependent protein catabolic process / negative regulation of neuron projection development / sperm principal piece / 5S rRNA binding / protein-containing complex assembly / sperm midpiece / Oxidative Stress Induced Senescence / cellular response to hypoxia / Regulation of TP53 Activity through Phosphorylation / amyloid fibril formation / ubiquitin-dependent protein catabolic process / proteasome-mediated ubiquitin-dependent protein catabolic process / regulation of cell cycle / Ub-specific processing proteases / postsynaptic density / protein ubiquitination / response to xenobiotic stimulus / protein domain specific binding / response to antibiotic / negative regulation of DNA-templated transcription / apoptotic process / positive regulation of cell population proliferation / ubiquitin protein ligase binding / positive regulation of gene expression Similarity search - Function | ||||||
| Biological species |  Homo sapiens (human) Homo sapiens (human) | ||||||
| Method |  X-RAY DIFFRACTION / X-RAY DIFFRACTION /  SYNCHROTRON / SYNCHROTRON /  MOLECULAR REPLACEMENT / Resolution: 2.3 Å MOLECULAR REPLACEMENT / Resolution: 2.3 Å | ||||||
 Authors Authors | Popowicz, G.M. / Czarna, A. / Wolf, S. / Holak, T.A. | ||||||
 Citation Citation |  Journal: Cell Cycle / Year: 2010 Journal: Cell Cycle / Year: 2010Title: Structures of low molecular weight inhibitors bound to MDMX and MDM2 reveal new approaches for p53-MDMX/MDM2 antagonist drug discovery Authors: Popowicz, G.M. / Czarna, A. / Wolf, S. / Wang, K. / Wang, W. / Domling, A. / Holak, T.A. | ||||||
| History |
|
- Structure visualization
Structure visualization
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  3lbk.cif.gz 3lbk.cif.gz | 32.5 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb3lbk.ent.gz pdb3lbk.ent.gz | 21.1 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  3lbk.json.gz 3lbk.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/lb/3lbk https://data.pdbj.org/pub/pdb/validation_reports/lb/3lbk ftp://data.pdbj.org/pub/pdb/validation_reports/lb/3lbk ftp://data.pdbj.org/pub/pdb/validation_reports/lb/3lbk | HTTPS FTP |
|---|
-Related structure data
| Related structure data |  3lbjC  3lblC  1rv1S C: citing same article ( S: Starting model for refinement |
|---|---|
| Similar structure data |
- Links
Links
- Assembly
Assembly
| Deposited unit | 
| ||||||||
|---|---|---|---|---|---|---|---|---|---|
| 1 |
| ||||||||
| 2 | 
| ||||||||
| Unit cell |
|
- Components
Components
| #1: Protein | Mass: 11159.074 Da / Num. of mol.: 1 / Fragment: p53 binding domain / Mutation: L33E Source method: isolated from a genetically manipulated source Source: (gene. exp.)  Homo sapiens (human) / Gene: MDM2 / Plasmid: pET-46 Ek/LIC / Production host: Homo sapiens (human) / Gene: MDM2 / Plasmid: pET-46 Ek/LIC / Production host:  References: UniProt: Q00987, Ligases; Forming carbon-nitrogen bonds; Acid-amino-acid ligases (peptide synthases) |
|---|---|
| #2: Chemical | ChemComp-K23 / |
| #3: Chemical | ChemComp-SO4 / |
| #4: Water | ChemComp-HOH / |
-Experimental details
-Experiment
| Experiment | Method:  X-RAY DIFFRACTION / Number of used crystals: 1 X-RAY DIFFRACTION / Number of used crystals: 1 |
|---|
- Sample preparation
Sample preparation
| Crystal | Density Matthews: 1.97 Å3/Da / Density % sol: 37.48 % |
|---|---|
| Crystal grow | Temperature: 300 K / Method: vapor diffusion, sitting drop / pH: 4.5 Details: 0.1M sodium-acetate-trihydrate, 2M ammonium sulfate, pH 4.5, VAPOR DIFFUSION, SITTING DROP, temperature 300K |
-Data collection
| Diffraction | Mean temperature: 90 K |
|---|---|
| Diffraction source | Source:  SYNCHROTRON / Site: SYNCHROTRON / Site:  SLS SLS  / Beamline: X10SA / Wavelength: 1 Å / Beamline: X10SA / Wavelength: 1 Å |
| Detector | Type: MAR CCD 165 mm / Detector: CCD / Date: May 25, 2009 / Details: monochromator, mirror |
| Radiation | Monochromator: Si / Protocol: SINGLE WAVELENGTH / Monochromatic (M) / Laue (L): M / Scattering type: x-ray |
| Radiation wavelength | Wavelength: 1 Å / Relative weight: 1 |
| Reflection | Resolution: 2.2→30 Å / Num. all: 7896 / Num. obs: 6949 / % possible obs: 88.6 % / Observed criterion σ(F): 2 / Observed criterion σ(I): 2 / Rmerge(I) obs: 0.079 / Net I/σ(I): 12.1 |
| Reflection shell | Resolution: 2.2→2.3 Å / Rmerge(I) obs: 0.502 / Mean I/σ(I) obs: 1.8 / % possible all: 80.4 |
- Processing
Processing
| Software |
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Refinement | Method to determine structure:  MOLECULAR REPLACEMENT MOLECULAR REPLACEMENTStarting model: 1RV1 Resolution: 2.3→20 Å / Cor.coef. Fo:Fc: 0.948 / Cor.coef. Fo:Fc free: 0.926 / SU B: 6.726 / SU ML: 0.157 / Cross valid method: THROUGHOUT / σ(F): 2 / ESU R: 0.231 / ESU R Free: 0.206 / Stereochemistry target values: MAXIMUM LIKELIHOOD / Details: HYDROGENS HAVE BEEN ADDED IN THE RIDING POSITIONS
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Solvent computation | Ion probe radii: 0.8 Å / Shrinkage radii: 0.8 Å / VDW probe radii: 1.2 Å / Solvent model: MASK | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Displacement parameters | Biso mean: 39.097 Å2
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refinement step | Cycle: LAST / Resolution: 2.3→20 Å
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refine LS restraints |
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| LS refinement shell | Resolution: 2.3→2.359 Å / Total num. of bins used: 20
|
 Movie
Movie Controller
Controller











 PDBj
PDBj















