[English] 日本語
 Yorodumi
Yorodumi- PDB-3cmp: Crystal structure of Siderocalin (NGAL, Lipocalin 2) K125A mutant... -
+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 3cmp | ||||||
|---|---|---|---|---|---|---|---|
| Title | Crystal structure of Siderocalin (NGAL, Lipocalin 2) K125A mutant complexed with Ferric Enterobactin | ||||||
 Components Components | Neutrophil gelatinase-associated lipocalin | ||||||
 Keywords Keywords | ANTIMICROBIAL PROTEIN / Lipocalin / Siderocalin / Enterobactin / Siderophore / Iron / Glycoprotein / Pyrrolidone carboxylic acid / Secreted | ||||||
| Function / homology |  Function and homology information Function and homology informationpositive regulation of iron ion import across plasma membrane / positive regulation of hippocampal neuron apoptotic process / positive regulation of endothelial tube morphogenesis / negative regulation of hippocampal neuron apoptotic process / positive regulation of cell projection organization / Metal sequestration by antimicrobial proteins / response to kainic acid / response to mycotoxin / siderophore transport / cellular response to increased oxygen levels ...positive regulation of iron ion import across plasma membrane / positive regulation of hippocampal neuron apoptotic process / positive regulation of endothelial tube morphogenesis / negative regulation of hippocampal neuron apoptotic process / positive regulation of cell projection organization / Metal sequestration by antimicrobial proteins / response to kainic acid / response to mycotoxin / siderophore transport / cellular response to increased oxygen levels / response to blue light / cellular response to X-ray / response to fructose / short-term memory / cellular response to interleukin-6 / iron ion sequestering activity / enterobactin binding / response to herbicide / response to iron(II) ion / positive regulation of reactive oxygen species biosynthetic process / cellular response to interleukin-1 / long-term memory / extrinsic apoptotic signaling pathway in absence of ligand / positive regulation of endothelial cell migration / cellular response to nutrient levels / acute-phase response / cellular response to nerve growth factor stimulus / Iron uptake and transport / specific granule lumen / cellular response to tumor necrosis factor / cellular response to amyloid-beta / response to virus / cellular response to hydrogen peroxide / positive regulation of cold-induced thermogenesis / cellular response to lipopolysaccharide / protease binding / Interleukin-4 and Interleukin-13 signaling / cellular response to hypoxia / defense response to bacterium / iron ion binding / response to xenobiotic stimulus / innate immune response / Neutrophil degranulation / positive regulation of gene expression / : / extracellular exosome / extracellular region / identical protein binding Similarity search - Function | ||||||
| Biological species |  Homo sapiens (human) Homo sapiens (human) | ||||||
| Method |  X-RAY DIFFRACTION / X-RAY DIFFRACTION /  SYNCHROTRON / SYNCHROTRON /  MOLECULAR REPLACEMENT / Resolution: 2.8 Å MOLECULAR REPLACEMENT / Resolution: 2.8 Å | ||||||
 Authors Authors | Clifton, M.C. / Strong, R.K. | ||||||
 Citation Citation |  Journal: To be Published Journal: To be PublishedTitle: Parsing the functional specificity of Siderocalin / Lipocalin 2 / NGAL for siderophores and related small-molecule ligands Authors: Clifton, M.C. / Rupert, P.B. / Hoette, T.M. / Raymond, K.N. / Abergel, R.J. / Strong, R.K. | ||||||
| History |
|
- Structure visualization
Structure visualization
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  3cmp.cif.gz 3cmp.cif.gz | 119.5 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb3cmp.ent.gz pdb3cmp.ent.gz | 90.4 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  3cmp.json.gz 3cmp.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/cm/3cmp https://data.pdbj.org/pub/pdb/validation_reports/cm/3cmp ftp://data.pdbj.org/pub/pdb/validation_reports/cm/3cmp ftp://data.pdbj.org/pub/pdb/validation_reports/cm/3cmp | HTTPS FTP |
|---|
-Related structure data
| Related structure data |  3hwdC  3hweC  3hwfC  3hwgC  3i0aC  3k3lC  3tf6C  3tnyC  3tzsC  1l6mS S: Starting model for refinement C: citing same article ( |
|---|---|
| Similar structure data |
- Links
Links
- Assembly
Assembly
| Deposited unit | 
| ||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | 
| ||||||||||||||||||
| 2 | 
| ||||||||||||||||||
| 3 | 
| ||||||||||||||||||
| Unit cell |
| ||||||||||||||||||
| Noncrystallographic symmetry (NCS) | NCS domain:
NCS domain segments: Component-ID: 1 / Ens-ID: 1 / Beg auth comp-ID: SER / Beg label comp-ID: SER / End auth comp-ID: ASP / End label comp-ID: ASP / Refine code: 4 / Auth seq-ID: 5 - 177 / Label seq-ID: 25 - 197
| ||||||||||||||||||
| Details | C87S mutation disrupts dimer formation |
- Components
Components
-Protein , 1 types, 3 molecules ABC
| #1: Protein | Mass: 22539.861 Da / Num. of mol.: 3 / Mutation: C87S, K125A Source method: isolated from a genetically manipulated source Source: (gene. exp.)  Homo sapiens (human) / Gene: LCN2, HNL, NGAL / Plasmid: pGEX-4T3 / Production host: Homo sapiens (human) / Gene: LCN2, HNL, NGAL / Plasmid: pGEX-4T3 / Production host:  |
|---|
-Non-polymers , 7 types, 113 molecules 


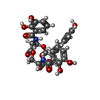









| #2: Chemical | | #3: Chemical | ChemComp-NA / #4: Chemical | #5: Chemical | #6: Chemical | #7: Chemical | #8: Water | ChemComp-HOH / | |
|---|
-Details
| Has protein modification | Y |
|---|
-Experimental details
-Experiment
| Experiment | Method:  X-RAY DIFFRACTION / Number of used crystals: 1 X-RAY DIFFRACTION / Number of used crystals: 1 |
|---|
- Sample preparation
Sample preparation
| Crystal | Density Matthews: 2.9 Å3/Da / Density % sol: 57.59 % |
|---|---|
| Crystal grow | Temperature: 291 K / Method: vapor diffusion, hanging drop / pH: 4.5 Details: 0.2M Ammonium sulfate, 30% PEG4000, pH 4.5, VAPOR DIFFUSION, HANGING DROP, temperature 291K |
-Data collection
| Diffraction | Mean temperature: 100 K |
|---|---|
| Diffraction source | Source:  SYNCHROTRON / Site: SYNCHROTRON / Site:  ALS ALS  / Beamline: 5.0.1 / Beamline: 5.0.1 |
| Detector | Detector: CCD |
| Radiation | Protocol: SINGLE WAVELENGTH / Monochromatic (M) / Laue (L): M / Scattering type: x-ray |
| Radiation wavelength | Relative weight: 1 |
| Reflection | Resolution: 2.8→82.48 Å / Num. all: 20269 / Num. obs: 20066 / % possible obs: 99 % / Redundancy: 9.8 % / Rmerge(I) obs: 0.077 / Net I/σ(I): 29.85 |
| Reflection shell | Resolution: 2.8→2.9 Å / Redundancy: 9.9 % / Rmerge(I) obs: 0.405 / Mean I/σ(I) obs: 5.92 / Num. unique all: 1955 / % possible all: 99.5 |
- Processing
Processing
| Software |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Refinement | Method to determine structure:  MOLECULAR REPLACEMENT MOLECULAR REPLACEMENTStarting model: PDB entry 1L6M Resolution: 2.8→47.96 Å / Cor.coef. Fo:Fc: 0.892 / Cor.coef. Fo:Fc free: 0.823 / SU B: 15.276 / SU ML: 0.307 / Cross valid method: THROUGHOUT / ESU R: 0.947 / ESU R Free: 0.407 / Stereochemistry target values: MAXIMUM LIKELIHOOD / Details: HYDROGENS HAVE BEEN ADDED IN THE RIDING POSITIONS
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Solvent computation | Ion probe radii: 0.8 Å / Shrinkage radii: 0.8 Å / VDW probe radii: 1.4 Å / Solvent model: MASK | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Displacement parameters | Biso mean: 31.185 Å2
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refinement step | Cycle: LAST / Resolution: 2.8→47.96 Å
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refine LS restraints |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refine LS restraints NCS | Dom-ID: 1 / Auth asym-ID: A / Ens-ID: 1 / Number: 2255 / Refine-ID: X-RAY DIFFRACTION
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| LS refinement shell | Resolution: 2.8→2.87 Å / Total num. of bins used: 20
|
 Movie
Movie Controller
Controller












 PDBj
PDBj







