+ Open data
Open data
- Basic information
Basic information
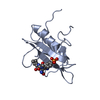
| Entry | Database: PDB / ID: 2sxl | ||||||
|---|---|---|---|---|---|---|---|
| Title | SEX-LETHAL RBD1, NMR, MINIMIZED AVERAGE STRUCTURE | ||||||
 Components Components | SEX-LETHAL PROTEIN | ||||||
 Keywords Keywords | RNA-BINDING DOMAIN / ALTERNATIVE SPLICING / RIKEN Structural Genomics/Proteomics Initiative / RSGI / Structural Genomics | ||||||
| Function / homology |  Function and homology information Function and homology informationsex determination, primary response to X:A ratio / germarium-derived cystoblast division / epithelium regeneration / somatic sex determination / female germ-line sex determination / oocyte differentiation / female sex determination / imaginal disc growth / sex determination / regulation of stem cell division ...sex determination, primary response to X:A ratio / germarium-derived cystoblast division / epithelium regeneration / somatic sex determination / female germ-line sex determination / oocyte differentiation / female sex determination / imaginal disc growth / sex determination / regulation of stem cell division / poly-pyrimidine tract binding / dosage compensation by hyperactivation of X chromosome / negative regulation of RNA export from nucleus / sex differentiation / alternative mRNA splicing, via spliceosome / poly(A) binding / regulation of mRNA splicing, via spliceosome / pre-mRNA binding / positive regulation of smoothened signaling pathway / reciprocal meiotic recombination / poly(U) RNA binding / oogenesis / regulation of alternative mRNA splicing, via spliceosome / negative regulation of mRNA splicing, via spliceosome / negative regulation of translational initiation / mRNA regulatory element binding translation repressor activity / positive regulation of RNA splicing / mRNA 3'-UTR binding / mRNA 5'-UTR binding / negative regulation of translation / protein stabilization / ribonucleoprotein complex / mRNA binding / protein-containing complex / RNA binding / nucleus / cytoplasm / cytosol Similarity search - Function | ||||||
| Biological species |  | ||||||
| Method | SOLUTION NMR / HYBRID DISTANCE GEOMETRY, SIMULATED ANNEALING METHOD | ||||||
 Authors Authors | Inoue, M. / Muto, Y. / Sakamoto, H. / Kigawa, T. / Takio, K. / Shimura, Y. / Yokoyama, S. / RIKEN Structural Genomics/Proteomics Initiative (RSGI) | ||||||
 Citation Citation |  Journal: J.Mol.Biol. / Year: 1997 Journal: J.Mol.Biol. / Year: 1997Title: A characteristic arrangement of aromatic amino acid residues in the solution structure of the amino-terminal RNA-binding domain of Drosophila sex-lethal. Authors: Inoue, M. / Muto, Y. / Sakamoto, H. / Kigawa, T. / Takio, K. / Shimura, Y. / Yokoyama, S. #1:  Journal: Nature / Year: 1994 Journal: Nature / Year: 1994Title: Crystal Structure at 1.92 A Resolution of the RNA-Binding Domain of the U1A Spliceosomal Protein Complexed with an RNA Hairpin Authors: Oubridge, C. / Ito, N. / Evans, P.R. / Teo, C.H. / Nagai, K. #2:  Journal: Biochemistry / Year: 1994 Journal: Biochemistry / Year: 1994Title: Resonance Assignments and Solution Structure of the Second RNA-Binding Domain of Sex-Lethal Determined by Multidimensional Heteronuclear Magnetic Resonance Authors: Lee, A.L. / Kanaar, R. / Rio, D.C. / Wemmer, D.E. #3:  Journal: Biochemistry / Year: 1994 Journal: Biochemistry / Year: 1994Title: Determination of the Secondary Structure and Folding Topology of an RNA Binding Domain of Mammalian Hnrnp A1 Protein Using Three-Dimensional Heteronuclear Magnetic Resonance Spectroscopy Authors: Garrett, D.S. / Lodi, P.J. / Shamoo, Y. / Williams, K.R. / Clore, G.M. / Gronenborn, A.M. #4:  Journal: Biochemistry / Year: 1992 Journal: Biochemistry / Year: 1992Title: 1H, 13C, and 15N NMR Assignments and Global Folding Pattern of the RNA-Binding Domain of the Human Hnrnp C Proteins Authors: Wittekind, M. / Gorlach, M. / Friedrichs, M. / Dreyfuss, G. / Mueller, L. #5:  Journal: Nature / Year: 1990 Journal: Nature / Year: 1990Title: Crystal Structure of the RNA-Binding Domain of the U1 Small Nuclear Ribonucleoprotein A Authors: Nagai, K. / Oubridge, C. / Jessen, T.H. / Li, J. / Evans, P.R. | ||||||
| History |
|
- Structure visualization
Structure visualization
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  2sxl.cif.gz 2sxl.cif.gz | 42.1 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb2sxl.ent.gz pdb2sxl.ent.gz | 30.4 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  2sxl.json.gz 2sxl.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/sx/2sxl https://data.pdbj.org/pub/pdb/validation_reports/sx/2sxl ftp://data.pdbj.org/pub/pdb/validation_reports/sx/2sxl ftp://data.pdbj.org/pub/pdb/validation_reports/sx/2sxl | HTTPS FTP |
|---|
-Related structure data
| Similar structure data | |
|---|---|
| Other databases |
- Links
Links
- Assembly
Assembly
| Deposited unit | 
| |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| 1 |
| |||||||||
| NMR ensembles |
|
- Components
Components
| #1: Protein | Mass: 10061.507 Da / Num. of mol.: 1 / Fragment: RNA-BINDING DOMAIN 1 (RBD1), RESIDUES 122 - 209 / Mutation: F166Y Source method: isolated from a genetically manipulated source Source: (gene. exp.)   |
|---|
-Experimental details
-Experiment
| Experiment | Method: SOLUTION NMR | ||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| NMR experiment |
|
- Sample preparation
Sample preparation
| Sample conditions | pH: 4 / Temperature: 298 K |
|---|---|
| Crystal grow | *PLUS Method: other / Details: NMR |
-NMR measurement
| NMR spectrometer |
|
|---|
- Processing
Processing
| Software |
| ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| NMR software |
| ||||||||||||
| Refinement | Method: HYBRID DISTANCE GEOMETRY, SIMULATED ANNEALING METHOD Software ordinal: 1 Details: REFINEMENT WAS DONE USING THE HYBRID DISTANCE GEOMETRY/SIMULATED ANNEALING METHOD (NILGES, M., CLORE, G.M., & GRONENBORN, A.M. (1988) FEBS LETT 229, 317-324) AS CONTAINED IN X-PLOR PROGRAM ...Details: REFINEMENT WAS DONE USING THE HYBRID DISTANCE GEOMETRY/SIMULATED ANNEALING METHOD (NILGES, M., CLORE, G.M., & GRONENBORN, A.M. (1988) FEBS LETT 229, 317-324) AS CONTAINED IN X-PLOR PROGRAM VERSION 3.1 (BRUNGER, 1992). THE RMSD FOR THE BACKBONE COORDINATES OF THE 20 STRUCTURES ACCEPTED AFTER THE LAST ROUND OF REFINEMENT COMPARED TO THE AVERAGE COORDINATES WAS 0.86 (FOR RESIDUES INVOLVED IN SECONDARY STRUCTURE). | ||||||||||||
| NMR ensemble | Conformer selection criteria: SMALLEST RESIDUAL ENERGY / Conformers calculated total number: 200 / Conformers submitted total number: 1 |
 Movie
Movie Controller
Controller












 PDBj
PDBj HSQC
HSQC