[English] 日本語
 Yorodumi
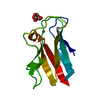
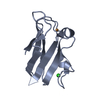
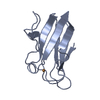
Yorodumi- PDB-2pcy: THE CRYSTAL STRUCTURE OF POPLAR APOPLASTOCYANIN AT 1.8-ANGSTROMS ... -
+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 2pcy | ||||||
|---|---|---|---|---|---|---|---|
| Title | THE CRYSTAL STRUCTURE OF POPLAR APOPLASTOCYANIN AT 1.8-ANGSTROMS RESOLUTION. THE GEOMETRY OF THE COPPER-BINDING SITE IS CREATED BY THE POLYPEPTIDE | ||||||
 Components Components | PLASTOCYANIN | ||||||
 Keywords Keywords | ELECTRON TRANSPORT PROTEIN(CUPROPROTEIN) | ||||||
| Function / homology |  Function and homology information Function and homology informationchloroplast thylakoid lumen / chloroplast thylakoid membrane / electron transfer activity / copper ion binding Similarity search - Function | ||||||
| Biological species |  | ||||||
| Method |  X-RAY DIFFRACTION / Resolution: 1.8 Å X-RAY DIFFRACTION / Resolution: 1.8 Å | ||||||
 Authors Authors | Garrett, T.P.J. / Guss, J.M. / Freeman, H.C. | ||||||
 Citation Citation |  Journal: J.Biol.Chem. / Year: 1984 Journal: J.Biol.Chem. / Year: 1984Title: The crystal structure of poplar apoplastocyanin at 1.8-A resolution. The geometry of the copper-binding site is created by the polypeptide Authors: Garrett, T.P. / Clingeleffer, D.J. / Guss, J.M. / Rogers, S.J. / Freeman, H.C. #1:  Journal: J.Mol.Biol. / Year: 1983 Journal: J.Mol.Biol. / Year: 1983Title: Structure of Oxidized Poplar Plastocyanin at 1.6 Angstroms Resolution Authors: Guss, J.M. / Freeman, H.C. #2:  Journal: Nature / Year: 1978 Journal: Nature / Year: 1978Title: X-Ray Crystal Structure Analysis of Plastocyanin at 2.7 Angstroms Resolution Authors: Colman, P.M. / Freeman, H.C. / Guss, J.M. / Murata, M. / Norris, V.A. / Ramshaw, J.A.M. / Venkatappa, M.P. | ||||||
| History |
|
- Structure visualization
Structure visualization
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  2pcy.cif.gz 2pcy.cif.gz | 29 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb2pcy.ent.gz pdb2pcy.ent.gz | 19.5 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  2pcy.json.gz 2pcy.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/pc/2pcy https://data.pdbj.org/pub/pdb/validation_reports/pc/2pcy ftp://data.pdbj.org/pub/pdb/validation_reports/pc/2pcy ftp://data.pdbj.org/pub/pdb/validation_reports/pc/2pcy | HTTPS FTP |
|---|
-Related structure data
| Similar structure data |
|---|
- Links
Links
- Assembly
Assembly
| Deposited unit | 
| ||||||||
|---|---|---|---|---|---|---|---|---|---|
| 1 |
| ||||||||
| Unit cell |
| ||||||||
| Atom site foot note | 1: RESIDUES 16 AND 36 ARE CIS-PROLINES. |
- Components
Components
| #1: Protein | Mass: 10493.607 Da / Num. of mol.: 1 Source method: isolated from a genetically manipulated source Source: (gene. exp.)  |
|---|---|
| #2: Water | ChemComp-HOH / |
-Experimental details
-Experiment
| Experiment | Method:  X-RAY DIFFRACTION X-RAY DIFFRACTION |
|---|
- Sample preparation
Sample preparation
| Crystal | Density Matthews: 1.91 Å3/Da / Density % sol: 35.5 % |
|---|---|
| Crystal grow | *PLUS Method: other / Details: Chapman, G.V., (1977) J. Mol. Biol., 110, 187. |
-Data collection
| Reflection | *PLUS Highest resolution: 1.8 Å / Num. all: 7891 / Num. obs: 7103 / Observed criterion σ(I): 1 / Rmerge(I) obs: 0.029 |
|---|
- Processing
Processing
| Software | Name: RESTRAINED / Version: RECIPROCAL-SPACE LEAST-SQUARES REFINEMENT / Classification: refinement | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Refinement | Resolution: 1.8→7 Å / Rfactor Rwork: 0.16 / σ(I): 1 | ||||||||||||
| Refinement step | Cycle: LAST / Resolution: 1.8→7 Å
|
 Movie
Movie Controller
Controller









 PDBj
PDBj
