+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 2lr5 | ||||||
|---|---|---|---|---|---|---|---|
| Title | 1H chemical shift assignments for micasin | ||||||
 Components Components | micasin | ||||||
 Keywords Keywords | ANTIMICROBIAL PROTEIN | ||||||
| Biological species |  Arthroderma otae (fungus) Arthroderma otae (fungus) | ||||||
| Method | SOLUTION NMR / torsion angle dynamics | ||||||
| Model details | lowest energy, model 1 | ||||||
 Authors Authors | Harvey, P.J. / Craik, D.J. / Zhu, S. | ||||||
 Citation Citation |  Journal: Proc.Natl.Acad.Sci.USA / Year: 2012 Journal: Proc.Natl.Acad.Sci.USA / Year: 2012Title: Dermatophytic defensin with antiinfective potential Authors: Zhu, S. / Gao, B. / Harvey, P.J. / Craik, D.J. | ||||||
| History |
|
- Structure visualization
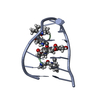
Structure visualization
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  2lr5.cif.gz 2lr5.cif.gz | 214 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb2lr5.ent.gz pdb2lr5.ent.gz | 177.4 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  2lr5.json.gz 2lr5.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Summary document |  2lr5_validation.pdf.gz 2lr5_validation.pdf.gz | 513.7 KB | Display |  wwPDB validaton report wwPDB validaton report |
|---|---|---|---|---|
| Full document |  2lr5_full_validation.pdf.gz 2lr5_full_validation.pdf.gz | 658.2 KB | Display | |
| Data in XML |  2lr5_validation.xml.gz 2lr5_validation.xml.gz | 20.3 KB | Display | |
| Data in CIF |  2lr5_validation.cif.gz 2lr5_validation.cif.gz | 30.3 KB | Display | |
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/lr/2lr5 https://data.pdbj.org/pub/pdb/validation_reports/lr/2lr5 ftp://data.pdbj.org/pub/pdb/validation_reports/lr/2lr5 ftp://data.pdbj.org/pub/pdb/validation_reports/lr/2lr5 | HTTPS FTP |
-Related structure data
| Similar structure data | |
|---|---|
| Other databases |
- Links
Links
- Assembly
Assembly
| Deposited unit | 
| |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| 1 |
| |||||||||
| NMR ensembles |
|
- Components
Components
| #1: Protein/peptide | Mass: 4067.702 Da / Num. of mol.: 1 / Source method: obtained synthetically / Source: (synth.)  Arthroderma otae (fungus) Arthroderma otae (fungus) |
|---|---|
| Has protein modification | Y |
| Sequence details | THE SEQUENCE OF THIS PROTEIN WAS NOT AVAILABLE AT THE UNIPROT KNOWLEDGEBASE DATABASE (UNIPROTKB) AT ...THE SEQUENCE OF THIS PROTEIN WAS NOT AVAILABLE AT THE UNIPROT KNOWLEDGEB |
-Experimental details
-Experiment
| Experiment | Method: SOLUTION NMR | ||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| NMR experiment |
|
- Sample preparation
Sample preparation
| Details |
| |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Sample |
| |||||||||
| Sample conditions | pH: 3 / Pressure: ambient / Temperature: 298 K |
-NMR measurement
| NMR spectrometer | Type: Bruker Avance / Manufacturer: Bruker / Model: AVANCE / Field strength: 600 MHz |
|---|
- Processing
Processing
| NMR software |
| ||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Refinement | Method: torsion angle dynamics / Software ordinal: 1 | ||||||||||||||||||||||||||||
| NMR constraints | NOE constraints total: 261 / NOE intraresidue total count: 79 / NOE long range total count: 29 / NOE medium range total count: 49 / NOE sequential total count: 93 / Protein chi angle constraints total count: 7 / Protein phi angle constraints total count: 28 / Protein psi angle constraints total count: 19 | ||||||||||||||||||||||||||||
| NMR representative | Selection criteria: lowest energy | ||||||||||||||||||||||||||||
| NMR ensemble | Conformer selection criteria: structures with the lowest energy Conformers calculated total number: 50 / Conformers submitted total number: 20 / Representative conformer: 1 |
 Movie
Movie Controller
Controller










 PDBj
PDBj HSQC
HSQC