+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 2j85 | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Title | B116 of Sulfolobus turreted icosahedral virus (STIV) | |||||||||
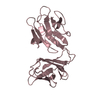
 Components Components | STIV B116 | |||||||||
 Keywords Keywords | VIRAL PROTEIN / ARCHAEAL VIRUS / CRENARCHAEAL VIRUS / B116 / STIV / ARCHAEA / SULFOLOBUS / CRENARCHAEA / SULFOLOBUS TURRETED ICOSAHEDRAL VIRUS / HYPOTHETICAL PROTEIN | |||||||||
| Function / homology | Uncharacterised protein PF08960, DUF1874 / STIV B116-like / STIV B116-like superfamily / STIV B116-like / Rossmann fold / 3-Layer(aba) Sandwich / Alpha Beta / Uncharacterized protein Function and homology information Function and homology information | |||||||||
| Biological species |    SULFOLOBUS TURRETED ICOSAHEDRAL VIRUS SULFOLOBUS TURRETED ICOSAHEDRAL VIRUS | |||||||||
| Method |  X-RAY DIFFRACTION / X-RAY DIFFRACTION /  SYNCHROTRON / SYNCHROTRON /  SAD / Resolution: 2.39 Å SAD / Resolution: 2.39 Å | |||||||||
 Authors Authors | Larson, E.T. / Reiter, D. / Young, M.J. / Lawrence, C.M. | |||||||||
 Citation Citation |  Journal: Virology / Year: 2007 Journal: Virology / Year: 2007Title: A New DNA Binding Protein Highly Conserved in Diverse Crenarchaeal Viruses Authors: Larson, E.T. / Eilers, B.J. / Reiter, D. / Ortmann, A.C. / Young, M.J. / Lawrence, C.M. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  2j85.cif.gz 2j85.cif.gz | 60.7 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb2j85.ent.gz pdb2j85.ent.gz | 46.4 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  2j85.json.gz 2j85.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/j8/2j85 https://data.pdbj.org/pub/pdb/validation_reports/j8/2j85 ftp://data.pdbj.org/pub/pdb/validation_reports/j8/2j85 ftp://data.pdbj.org/pub/pdb/validation_reports/j8/2j85 | HTTPS FTP |
|---|
-Related structure data
| Similar structure data |
|---|
- Links
Links
- Assembly
Assembly
| Deposited unit | 
| ||||||||
|---|---|---|---|---|---|---|---|---|---|
| 1 |
| ||||||||
| Unit cell |
|
- Components
Components
| #1: Protein | Mass: 14409.745 Da / Num. of mol.: 2 Source method: isolated from a genetically manipulated source Source: (gene. exp.)    SULFOLOBUS TURRETED ICOSAHEDRAL VIRUS / Strain: ISOLATE YNPRC179 SULFOLOBUS TURRETED ICOSAHEDRAL VIRUS / Strain: ISOLATE YNPRC179Description: STIV WAS ISOLATED FROM SULFOLOBUS SPECIES IN ACIDIC HOT SPRINGS (PH 2.9-3.9, 72-92 DEGREES C) IN THE RABBIT CREEK THERMAL AREA WITHIN MIDWAY GEYSER BASIN INYELLOWSTONE NATIONAL PARK ...Description: STIV WAS ISOLATED FROM SULFOLOBUS SPECIES IN ACIDIC HOT SPRINGS (PH 2.9-3.9, 72-92 DEGREES C) IN THE RABBIT CREEK THERMAL AREA WITHIN MIDWAY GEYSER BASIN INYELLOWSTONE NATIONAL PARK (RICE, ET AL. PNAS. MAY 18, 2004 VOL. 101 NO. 20. PP. 7716-7720.) Plasmid: PEXP14-STIVB116 / Production host:  #2: Water | ChemComp-HOH / | Has protein modification | Y | Sequence details | C-TERMINUS 6XHIS TAG WAS ADDED DURING CLONING. | |
|---|
-Experimental details
-Experiment
| Experiment | Method:  X-RAY DIFFRACTION / Number of used crystals: 1 X-RAY DIFFRACTION / Number of used crystals: 1 |
|---|
- Sample preparation
Sample preparation
| Crystal | Density Matthews: 3.3 Å3/Da / Density % sol: 62.4 % Description: DATA WERE COLLECTED DURING RAPIDATA 2004 COURSE AT NSLS. |
|---|---|
| Crystal grow | Temperature: 290 K / Method: vapor diffusion, hanging drop / pH: 8 Details: 6.5 MG/ML PROTEIN WAS CRYSTALLIZED BY HANGING DROP VAPOR DIFFUSION IN 0.1 M TRIS-HCL (PH 8.0), 1.95 M NH4H2PO4 AT 17 DEGREES C. |
-Data collection
| Diffraction | Mean temperature: 100 K |
|---|---|
| Diffraction source | Source:  SYNCHROTRON / Site: SYNCHROTRON / Site:  NSLS NSLS  / Beamline: X9B / Wavelength: 0.97946 / Beamline: X9B / Wavelength: 0.97946 |
| Detector | Type: ADSC QUANTUM 4 / Detector: CCD / Date: Apr 27, 2004 / Details: MIRRORS |
| Radiation | Monochromator: CRYSTAL / Protocol: SINGLE WAVELENGTH / Monochromatic (M) / Laue (L): M / Scattering type: x-ray |
| Radiation wavelength | Wavelength: 0.97946 Å / Relative weight: 1 |
| Reflection | Resolution: 2.4→30 Å / Num. obs: 14911 / % possible obs: 99.9 % / Observed criterion σ(I): 3 / Redundancy: 5.1 % / Rmerge(I) obs: 0.07 / Net I/σ(I): 22 |
| Reflection shell | Resolution: 2.4→2.49 Å / Redundancy: 5.3 % / Rmerge(I) obs: 0.26 / Mean I/σ(I) obs: 5 / % possible all: 100 |
- Processing
Processing
| Software |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Refinement | Method to determine structure:  SAD / Resolution: 2.39→29.81 Å / Cor.coef. Fo:Fc: 0.93 / Cor.coef. Fo:Fc free: 0.918 / SU B: 12.526 / SU ML: 0.152 / TLS residual ADP flag: LIKELY RESIDUAL / Cross valid method: THROUGHOUT / ESU R: 0.283 / ESU R Free: 0.218 / Stereochemistry target values: MAXIMUM LIKELIHOOD SAD / Resolution: 2.39→29.81 Å / Cor.coef. Fo:Fc: 0.93 / Cor.coef. Fo:Fc free: 0.918 / SU B: 12.526 / SU ML: 0.152 / TLS residual ADP flag: LIKELY RESIDUAL / Cross valid method: THROUGHOUT / ESU R: 0.283 / ESU R Free: 0.218 / Stereochemistry target values: MAXIMUM LIKELIHOODDetails: HYDROGENS HAVE BEEN ADDED IN THE RIDING POSITIONS. TLS GROUP SELECTIONS WERE MADE WITH THE AID OF TLS MOTION DETERMINATION SERVER. PAINTER AND MERRITT. 2005. ACTA CRYST D61, 465-471. CHAIN A ...Details: HYDROGENS HAVE BEEN ADDED IN THE RIDING POSITIONS. TLS GROUP SELECTIONS WERE MADE WITH THE AID OF TLS MOTION DETERMINATION SERVER. PAINTER AND MERRITT. 2005. ACTA CRYST D61, 465-471. CHAIN A AND CHAIN B TOGETHER MAKE UP THE BIOLOGICAL MOLECULE.
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Solvent computation | Ion probe radii: 0.8 Å / Shrinkage radii: 0.8 Å / VDW probe radii: 1.2 Å / Solvent model: BABINET MODEL WITH MASK | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Displacement parameters | Biso mean: 28.3 Å2
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refinement step | Cycle: LAST / Resolution: 2.39→29.81 Å
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refine LS restraints |
|
 Movie
Movie Controller
Controller











 PDBj
PDBj