+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 2hqh | ||||||
|---|---|---|---|---|---|---|---|
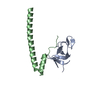
| Title | Crystal structure of p150Glued and CLIP-170 | ||||||
 Components Components |
| ||||||
 Keywords Keywords | STRUCTURAL PROTEIN / PROTEIN BINDING / beta/beta structure / zinc finger motif | ||||||
| Function / homology |  Function and homology information Function and homology informationcell cortex region / centriolar subdistal appendage / centriole-centriole cohesion / positive regulation of neuromuscular junction development / microtubule anchoring at centrosome / maintenance of synapse structure / ventral spinal cord development / mitotic nuclear membrane disassembly / dynein complex / microtubule plus-end ...cell cortex region / centriolar subdistal appendage / centriole-centriole cohesion / positive regulation of neuromuscular junction development / microtubule anchoring at centrosome / maintenance of synapse structure / ventral spinal cord development / mitotic nuclear membrane disassembly / dynein complex / microtubule plus-end / XBP1(S) activates chaperone genes / positive regulation of microtubule nucleation / melanosome transport / microtubule plus-end binding / microtubule bundle formation / non-motile cilium assembly / retrograde transport, endosome to Golgi / Signaling by LTK in cancer / COPI-independent Golgi-to-ER retrograde traffic / microtubule associated complex / intermediate filament / nuclear migration / perinuclear theca / neuromuscular process / establishment of mitotic spindle orientation / neuromuscular junction development / motor behavior / cell leading edge / RHO GTPases activate IQGAPs / intercellular bridge / positive regulation of microtubule polymerization / COPI-mediated anterograde transport / ruffle / cytoplasmic microtubule organization / neuron projection maintenance / Amplification of signal from unattached kinetochores via a MAD2 inhibitory signal / Loss of Nlp from mitotic centrosomes / Loss of proteins required for interphase microtubule organization from the centrosome / Recruitment of mitotic centrosome proteins and complexes / MHC class II antigen presentation / Recruitment of NuMA to mitotic centrosomes / Anchoring of the basal body to the plasma membrane / Mitotic Prometaphase / HSP90 chaperone cycle for steroid hormone receptors (SHR) in the presence of ligand / EML4 and NUDC in mitotic spindle formation / cytoplasmic vesicle membrane / acrosomal vesicle / AURKA Activation by TPX2 / Resolution of Sister Chromatid Cohesion / regulation of mitotic spindle organization / centriole / tubulin binding / RHO GTPases Activate Formins / neuron cellular homeostasis / kinetochore / tau protein binding / spindle / spindle pole / mitotic spindle / Signaling by ALK fusions and activated point mutants / Separation of Sister Chromatids / Regulation of PLK1 Activity at G2/M Transition / nuclear envelope / nervous system development / mitotic cell cycle / microtubule cytoskeleton / cell cortex / microtubule binding / microtubule / neuron projection / cilium / ciliary basal body / axon / cell division / neuronal cell body / centrosome / protein kinase binding / zinc ion binding / membrane / identical protein binding / nucleus / plasma membrane / cytoplasm / cytosol Similarity search - Function | ||||||
| Biological species |  Homo sapiens (human) Homo sapiens (human) | ||||||
| Method |  X-RAY DIFFRACTION / X-RAY DIFFRACTION /  SYNCHROTRON / SYNCHROTRON /  MAD / Resolution: 1.8 Å MAD / Resolution: 1.8 Å | ||||||
 Authors Authors | Hayashi, I. / Ikura, M. | ||||||
 Citation Citation |  Journal: Nat.Struct.Mol.Biol. / Year: 2007 Journal: Nat.Struct.Mol.Biol. / Year: 2007Title: CLIP170 autoinhibition mimics intermolecular interactions with p150Glued or EB1. Authors: Hayashi, I. / Plevin, M.J. / Ikura, M. | ||||||
| History |
|
- Structure visualization
Structure visualization
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  2hqh.cif.gz 2hqh.cif.gz | 95.6 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb2hqh.ent.gz pdb2hqh.ent.gz | 73.8 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  2hqh.json.gz 2hqh.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/hq/2hqh https://data.pdbj.org/pub/pdb/validation_reports/hq/2hqh ftp://data.pdbj.org/pub/pdb/validation_reports/hq/2hqh ftp://data.pdbj.org/pub/pdb/validation_reports/hq/2hqh | HTTPS FTP |
|---|
-Related structure data
| Related structure data | |
|---|---|
| Similar structure data |
- Links
Links
- Assembly
Assembly
| Deposited unit | 
| ||||||||
|---|---|---|---|---|---|---|---|---|---|
| 1 | 
| ||||||||
| 2 | 
| ||||||||
| 3 | 
| ||||||||
| 4 | 
| ||||||||
| 5 | 
| ||||||||
| Unit cell |
| ||||||||
| Components on special symmetry positions |
| ||||||||
| Details | The biological assembly is a dimer of chain A and E, or B and F, or C and G, or D and H |
- Components
Components
| #1: Protein | Mass: 9928.076 Da / Num. of mol.: 4 / Fragment: CAP-Gly domain, residues 15-107 Source method: isolated from a genetically manipulated source Source: (gene. exp.)  Homo sapiens (human) / Gene: DCTN1 / Plasmid: pET15b / Species (production host): Escherichia coli / Production host: Homo sapiens (human) / Gene: DCTN1 / Plasmid: pET15b / Species (production host): Escherichia coli / Production host:  #2: Protein/peptide | Mass: 2928.173 Da / Num. of mol.: 4 / Fragment: second zinc finger domain, residues 1405-1427 Source method: isolated from a genetically manipulated source Source: (gene. exp.)  Homo sapiens (human) / Gene: RSN, CYLN1 / Plasmid: pGEX-4T1 / Species (production host): Escherichia coli / Production host: Homo sapiens (human) / Gene: RSN, CYLN1 / Plasmid: pGEX-4T1 / Species (production host): Escherichia coli / Production host:  #3: Chemical | ChemComp-ZN / #4: Water | ChemComp-HOH / | |
|---|
-Experimental details
-Experiment
| Experiment | Method:  X-RAY DIFFRACTION / Number of used crystals: 1 X-RAY DIFFRACTION / Number of used crystals: 1 |
|---|
- Sample preparation
Sample preparation
| Crystal | Density Matthews: 2.54 Å3/Da / Density % sol: 51.48 % |
|---|---|
| Crystal grow | Temperature: 298 K / Method: vapor diffusion, sitting drop / pH: 7 Details: 4M sodium formate, pH 7, VAPOR DIFFUSION, SITTING DROP, temperature 298K |
-Data collection
| Diffraction | Mean temperature: 100 K | |||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Diffraction source | Source:  SYNCHROTRON / Site: SYNCHROTRON / Site:  APS APS  / Beamline: 19-BM / Wavelength: 0.9791, 1.2826, 1.2830, 1.2694 / Beamline: 19-BM / Wavelength: 0.9791, 1.2826, 1.2830, 1.2694 | |||||||||||||||
| Detector | Type: CUSTOM-MADE / Detector: CCD / Date: Jun 5, 2005 | |||||||||||||||
| Radiation | Protocol: MAD / Monochromatic (M) / Laue (L): M / Scattering type: x-ray | |||||||||||||||
| Radiation wavelength |
| |||||||||||||||
| Reflection | Resolution: 1.8→45.7 Å / Num. all: 48867 / Num. obs: 49011 / % possible obs: 100 % / Observed criterion σ(F): 0 / Observed criterion σ(I): 0 / Biso Wilson estimate: 14 Å2 | |||||||||||||||
| Reflection shell | Resolution: 1.8→1.86 Å / % possible all: 100 |
- Processing
Processing
| Software |
| |||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Refinement | Method to determine structure:  MAD / Resolution: 1.8→45.68 Å / σ(F): 0 / σ(I): 0 / Stereochemistry target values: Engh & Huber MAD / Resolution: 1.8→45.68 Å / σ(F): 0 / σ(I): 0 / Stereochemistry target values: Engh & Huber
| |||||||||||||||||||||||||
| Displacement parameters | Biso mean: 22.6 Å2
| |||||||||||||||||||||||||
| Refinement step | Cycle: LAST / Resolution: 1.8→45.68 Å
|
 Movie
Movie Controller
Controller













 PDBj
PDBj























