Entry Database : PDB / ID : 1zzqTitle Rat nNOS D597N mutant with L-N(omega)-Nitroarginine-(4R)-amino-L-proline amide bound Nitric-oxide synthase, brain Keywords / Function / homology Function Domain/homology Component
/ / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / Biological species Rattus norvegicus (Norway rat)Method / / / Resolution : 1.9 Å Authors Li, H. / Flinspach, M.L. / Igarashi, J. / Jamal, J. / Yang, W. / Gomez-Vidal, J.A. / Litzinger, E.A. / Silverman, R.B. / Poulos, T.L. Journal : Biochemistry / Year : 2005Title : Exploring the Binding Conformations of Bulkier Dipeptide Amide Inhibitors in Constitutive Nitric Oxide Synthases.
Authors :
Li, H. / Flinspach, M.L. / Igarashi, J. / Jamal, J. / Yang, W. / Litzinger, E.A. / Huang, H. / Erdal, E.P. / Silverman, R.B. / Poulos, T.L. #1: Journal : Nat.Struct.Mol.Biol. / Year : 2004Title : Structural Basis for Dipeptide Amide Isoform-Selective Inhibition of Neuronal Nitric Oxide Synthase
Authors :
Flinspach, M.L. / Li, H. / Jamal, J. / Yang, W. / Huang, H. / Hah, J.M. / Gomez-Vidal, J.A. / Litzinger, E.A. / Silverman, R.B. / Poulos, T.L. History Deposition Jun 14, 2005 Deposition site / Processing site Revision 1.0 Dec 6, 2005 Provider / Type Revision 1.1 Apr 30, 2008 Group Revision 1.2 Jul 13, 2011 Group Revision 1.3 Oct 20, 2021 Group / Derived calculationsCategory database_2 / pdbx_struct_conn_angle ... database_2 / pdbx_struct_conn_angle / struct_conn / struct_ref_seq_dif / struct_site Item _database_2.pdbx_DOI / _database_2.pdbx_database_accession ... _database_2.pdbx_DOI / _database_2.pdbx_database_accession / _pdbx_struct_conn_angle.ptnr1_auth_asym_id / _pdbx_struct_conn_angle.ptnr1_auth_seq_id / _pdbx_struct_conn_angle.ptnr1_label_asym_id / _pdbx_struct_conn_angle.ptnr1_label_seq_id / _pdbx_struct_conn_angle.ptnr3_auth_asym_id / _pdbx_struct_conn_angle.ptnr3_auth_seq_id / _pdbx_struct_conn_angle.ptnr3_label_asym_id / _pdbx_struct_conn_angle.ptnr3_label_seq_id / _pdbx_struct_conn_angle.value / _struct_conn.pdbx_dist_value / _struct_conn.ptnr1_auth_comp_id / _struct_conn.ptnr1_auth_seq_id / _struct_conn.ptnr1_label_asym_id / _struct_conn.ptnr1_label_atom_id / _struct_conn.ptnr1_label_comp_id / _struct_conn.ptnr1_label_seq_id / _struct_conn.ptnr2_auth_asym_id / _struct_conn.ptnr2_auth_comp_id / _struct_conn.ptnr2_auth_seq_id / _struct_conn.ptnr2_label_asym_id / _struct_conn.ptnr2_label_atom_id / _struct_conn.ptnr2_label_comp_id / _struct_conn.ptnr2_label_seq_id / _struct_ref_seq_dif.details / _struct_site.pdbx_auth_asym_id / _struct_site.pdbx_auth_comp_id / _struct_site.pdbx_auth_seq_id Revision 1.4 Feb 14, 2024 Group / Category / chem_comp_bond
Show all Show less
 Yorodumi
Yorodumi Open data
Open data Basic information
Basic information Components
Components Keywords
Keywords Function and homology information
Function and homology information
 X-RAY DIFFRACTION /
X-RAY DIFFRACTION /  SYNCHROTRON /
SYNCHROTRON /  FOURIER SYNTHESIS / Resolution: 1.9 Å
FOURIER SYNTHESIS / Resolution: 1.9 Å  Authors
Authors Citation
Citation Journal: Biochemistry / Year: 2005
Journal: Biochemistry / Year: 2005 Journal: Nat.Struct.Mol.Biol. / Year: 2004
Journal: Nat.Struct.Mol.Biol. / Year: 2004 Structure visualization
Structure visualization Molmil
Molmil Jmol/JSmol
Jmol/JSmol Downloads & links
Downloads & links Download
Download 1zzq.cif.gz
1zzq.cif.gz PDBx/mmCIF format
PDBx/mmCIF format pdb1zzq.ent.gz
pdb1zzq.ent.gz PDB format
PDB format 1zzq.json.gz
1zzq.json.gz PDBx/mmJSON format
PDBx/mmJSON format Other downloads
Other downloads https://data.pdbj.org/pub/pdb/validation_reports/zz/1zzq
https://data.pdbj.org/pub/pdb/validation_reports/zz/1zzq ftp://data.pdbj.org/pub/pdb/validation_reports/zz/1zzq
ftp://data.pdbj.org/pub/pdb/validation_reports/zz/1zzq Links
Links Assembly
Assembly
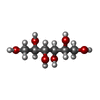
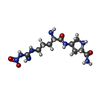
 Components
Components












 X-RAY DIFFRACTION / Number of used crystals: 1
X-RAY DIFFRACTION / Number of used crystals: 1  Sample preparation
Sample preparation SYNCHROTRON / Site:
SYNCHROTRON / Site:  SSRL
SSRL  / Beamline: BL9-1 / Wavelength: 0.9537 Å
/ Beamline: BL9-1 / Wavelength: 0.9537 Å Processing
Processing FOURIER SYNTHESIS / Resolution: 1.9→35.89 Å / Rfactor Rfree error: 0.004 / Data cutoff high absF: 2500416.39 / Data cutoff low absF: 0 / Isotropic thermal model: RESTRAINED / Cross valid method: THROUGHOUT / σ(F): 0 / Stereochemistry target values: Engh & Huber
FOURIER SYNTHESIS / Resolution: 1.9→35.89 Å / Rfactor Rfree error: 0.004 / Data cutoff high absF: 2500416.39 / Data cutoff low absF: 0 / Isotropic thermal model: RESTRAINED / Cross valid method: THROUGHOUT / σ(F): 0 / Stereochemistry target values: Engh & Huber Movie
Movie Controller
Controller



























 PDBj
PDBj









