[English] 日本語
 Yorodumi
Yorodumi- PDB-1yub: SOLUTION STRUCTURE OF AN RRNA METHYLTRANSFERASE (ERMAM) THAT CONF... -
+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 1yub | ||||||
|---|---|---|---|---|---|---|---|
| Title | SOLUTION STRUCTURE OF AN RRNA METHYLTRANSFERASE (ERMAM) THAT CONFERS MACROLIDE-LINCOSAMIDE-STREPTOGRAMIN ANTIBIOTIC RESISTANCE, NMR, MINIMIZED AVERAGE STRUCTURE | ||||||
 Components Components | RRNA METHYLTRANSFERASE | ||||||
 Keywords Keywords | METHYLTRANSFERASE / ERM / ERMAM / MLS ANTIBIOTICS / RRNA | ||||||
| Function / homology |  Function and homology information Function and homology information23S rRNA (adenine2085-N6)-dimethyltransferase / 23S rRNA (adenine(2085)-N(6))-dimethyltransferase activity / rRNA (adenine-N6,N6-)-dimethyltransferase activity / response to antibiotic / RNA binding / cytosol Similarity search - Function | ||||||
| Biological species |  | ||||||
| Method | SOLUTION NMR / DG, SA | ||||||
 Authors Authors | Yu, L. / Petros, A.M. / Schnuchel, A. / Zhong, P. / Severin, J.M. / Walter, K. / Holzman, T.F. / Fesik, S.W. | ||||||
 Citation Citation |  Journal: Nat.Struct.Biol. / Year: 1997 Journal: Nat.Struct.Biol. / Year: 1997Title: Solution structure of an rRNA methyltransferase (ErmAM) that confers macrolide-lincosamide-streptogramin antibiotic resistance. Authors: Yu, L. / Petros, A.M. / Schnuchel, A. / Zhong, P. / Severin, J.M. / Walter, K. / Holzman, T.F. / Fesik, S.W. #1:  Journal: Nat.Struct.Biol. / Year: 1997 Journal: Nat.Struct.Biol. / Year: 1997Title: Erratum. Solution Structure of an Rrna Methyltransferase (Ermam) that Confers Macrolide-Lincosamide-Streptogramin Antibiotic Resistance Authors: Yu, L. / Petros, A.M. / Schnuchel, A. / Zhong, P. / Severin, J.M. / Walter, K. / Holzman, T.F. / Fesik, S.W. | ||||||
| History |
|
- Structure visualization
Structure visualization
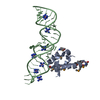
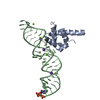
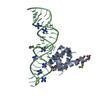
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  1yub.cif.gz 1yub.cif.gz | 102.5 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb1yub.ent.gz pdb1yub.ent.gz | 80.7 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  1yub.json.gz 1yub.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/yu/1yub https://data.pdbj.org/pub/pdb/validation_reports/yu/1yub ftp://data.pdbj.org/pub/pdb/validation_reports/yu/1yub ftp://data.pdbj.org/pub/pdb/validation_reports/yu/1yub | HTTPS FTP |
|---|
-Related structure data
| Similar structure data |
|---|
- Links
Links
- Assembly
Assembly
| Deposited unit | 
| |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| 1 |
| |||||||||
| NMR ensembles |
|
- Components
Components
| #1: Protein | Mass: 28862.568 Da / Num. of mol.: 1 / Mutation: I75T, S100N, H118R Source method: isolated from a genetically manipulated source Source: (gene. exp.)  Strain: 5728, A CLINICAL ISOLATE FROM ABBOTT CULTURE COLLECTION Cell line: BL21 / Gene: ERM / Plasmid: PET-24(+) / Species (production host): Escherichia coli / Gene (production host): ERM / Production host:  |
|---|
-Experimental details
-Experiment
| Experiment | Method: SOLUTION NMR | ||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| NMR experiment |
|
- Sample preparation
Sample preparation
| Sample conditions | Ionic strength: 50 mM SODIUM PHOSPHATE, 100 mM NACL / pH: 6.5 / Temperature: 303 K |
|---|---|
| Crystal grow | *PLUS Method: other / Details: NMR |
-NMR measurement
| NMR spectrometer |
|
|---|
- Processing
Processing
| Software |
| ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| NMR software |
| ||||||||||||
| Refinement | Method: DG, SA / Software ordinal: 1 Details: THE TOTAL NUMBER OF DISTANCE RESTRAINTS USED WAS 3259. THE TOTAL NUMBER OF TORSIONAL RESTRAINTS USED WAS 63. | ||||||||||||
| NMR ensemble | Conformer selection criteria: LOWEST ENERGY AND LEAST RESTRAINT VIOLATION Conformers calculated total number: 17 / Conformers submitted total number: 1 |
 Movie
Movie Controller
Controller









 PDBj
PDBj X-PLOR
X-PLOR