[English] 日本語
 Yorodumi
Yorodumi- PDB-1tn3: THE C-TYPE LECTIN CARBOHYDRATE RECOGNITION DOMAIN OF HUMAN TETRANECTIN -
+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 1tn3 | ||||||
|---|---|---|---|---|---|---|---|
| Title | THE C-TYPE LECTIN CARBOHYDRATE RECOGNITION DOMAIN OF HUMAN TETRANECTIN | ||||||
 Components Components | TETRANECTIN | ||||||
 Keywords Keywords | LECTIN / TETRANECTIN / PLASMINOGEN BINDING / KRINGLE 4 / C-TYPE LECTIN / CARBOHYDRATE RECOGNITION DOMAIN | ||||||
| Function / homology |  Function and homology information Function and homology informationkringle domain binding / granular component / platelet dense granule lumen / positive regulation of plasminogen activation / bone mineralization / ossification / Platelet degranulation / heparin binding / carbohydrate binding / extracellular matrix ...kringle domain binding / granular component / platelet dense granule lumen / positive regulation of plasminogen activation / bone mineralization / ossification / Platelet degranulation / heparin binding / carbohydrate binding / extracellular matrix / calcium ion binding / : / extracellular exosome / extracellular region / cytoplasm Similarity search - Function | ||||||
| Biological species |  Homo sapiens (human) Homo sapiens (human) | ||||||
| Method |  X-RAY DIFFRACTION / X-RAY DIFFRACTION /  SYNCHROTRON / SYNCHROTRON /  MOLECULAR REPLACEMENT / Resolution: 2 Å MOLECULAR REPLACEMENT / Resolution: 2 Å | ||||||
 Authors Authors | Kastrup, J.S. / Nielsen, B.B. / Rasmussen, H. / Holtet, T.L. / Graversen, J.H. / Etzerodt, M. / Thoegersen, H.C. / Larsen, I.K. | ||||||
 Citation Citation |  Journal: Acta Crystallogr.,Sect.D / Year: 1998 Journal: Acta Crystallogr.,Sect.D / Year: 1998Title: Structure of the C-type lectin carbohydrate recognition domain of human tetranectin. Authors: Kastrup, J.S. / Nielsen, B.B. / Rasmussen, H. / Holtet, T.L. / Graversen, J.H. / Etzerodt, M. / Thogersen, H.C. / Larsen, I.K. #1:  Journal: Acta Crystallogr.,Sect.D / Year: 1997 Journal: Acta Crystallogr.,Sect.D / Year: 1997Title: Human Plasminogen Binding Protein Tetranectin: Crystallization and Preliminary X-Ray Analysis of the C-Type Lectin Crd and the Full-Length Protein Authors: Kastrup, J.S. / Rasmussen, H. / Nielsen, B.B. / Larsen, I.K. / Holtet, T.L. / Graversen, J.H. / Etzerodt, M. / Thogersen, H.C. #2:  Journal: FEBS Lett. / Year: 1997 Journal: FEBS Lett. / Year: 1997Title: Crystal Structure of Tetranectin, a Trimeric Plasminogen-Binding Protein with an Alpha-Helical Coiled Coil Authors: Nielsen, B.B. / Kastrup, J.S. / Rasmussen, H. / Holtet, T.L. / Graversen, J.H. / Etzerodt, M. / Thogersen, H.C. / Larsen, I.K. #3:  Journal: Protein Sci. / Year: 1997 Journal: Protein Sci. / Year: 1997Title: Tetranectin, a Trimeric Plasminogen-Binding C-Type Lectin Authors: Holtet, T.L. / Graversen, J.H. / Clemmensen, I. / Thogersen, H.C. / Etzerodt, M. #4:  Journal: FEBS Lett. / Year: 1992 Journal: FEBS Lett. / Year: 1992Title: The Gene Structure of Tetranectin, a Plasminogen Binding Protein Authors: Berglund, L. / Petersen, T.E. | ||||||
| History |
|
- Structure visualization
Structure visualization
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  1tn3.cif.gz 1tn3.cif.gz | 44 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb1tn3.ent.gz pdb1tn3.ent.gz | 29.7 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  1tn3.json.gz 1tn3.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/tn/1tn3 https://data.pdbj.org/pub/pdb/validation_reports/tn/1tn3 ftp://data.pdbj.org/pub/pdb/validation_reports/tn/1tn3 ftp://data.pdbj.org/pub/pdb/validation_reports/tn/1tn3 | HTTPS FTP |
|---|
-Related structure data
| Related structure data |  2msbS S: Starting model for refinement |
|---|---|
| Similar structure data |
- Links
Links
- Assembly
Assembly
| Deposited unit | 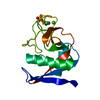
| |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| 1 |
| |||||||||
| Unit cell |
| |||||||||
| Components on special symmetry positions |
| |||||||||
| Details | THE CARBOHYDRATE RECOGNITION DOMAIN OF TETRANECTIN FORMS A DIMER IN CRYSTALS, WHEREAS FULL-LENGTH TETRANECTIN IS A HOMOTRIMER IN CRYSTALS AS WELL AS IN SOLUTION. TWO DIFFERENT SIDE CHAIN CONFORMATIONS HAVE BEEN IDENTIFIED FOR THR 48, VAL 49, AND MET 58. IN ADDITION, THE AMIDE MOIETIES LINKING THR 138 AND GLU 139 AND THR 141 AND ALA 142, RESPECTIVELY, ARE OBSERVED IN TWO DISTINCT CONFORMATIONS. THE SIDE CHAINS OF NINE RESIDUES (GLN 66, ARG 101, GLU 107, ARG 130, LYS 134, GLU 139, ARG 169, ILE 180, VAL 181) ARE LESS WELL-DEFINED. |
- Components
Components
| #1: Protein | Mass: 15136.991 Da / Num. of mol.: 1 / Fragment: RESIDUES 45 - 181 Source method: isolated from a genetically manipulated source Source: (gene. exp.)  Homo sapiens (human) / Plasmid: PT7H6 / Production host: Homo sapiens (human) / Plasmid: PT7H6 / Production host:  | ||||||||
|---|---|---|---|---|---|---|---|---|---|
| #2: Chemical | | #3: Chemical | ChemComp-SO4 / | #4: Chemical | ChemComp-EOH / | #5: Water | ChemComp-HOH / | Has protein modification | Y | |
-Experimental details
-Experiment
| Experiment | Method:  X-RAY DIFFRACTION / Number of used crystals: 2 X-RAY DIFFRACTION / Number of used crystals: 2 |
|---|
- Sample preparation
Sample preparation
| Crystal | Density Matthews: 2.6 Å3/Da / Density % sol: 53 % | |||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Crystal grow | Method: vapor diffusion, hanging drop / pH: 8 Details: THE PROTEIN WAS CRYSTALLIZED BY THE HANGING DROP METHOD BY MIXING 4 MICROLITER OF A SOLUTION CONTAINING 5 MG/ML PROTEIN, 0.02 M CACL2, 0.05 M NACL, 0.10 M TRIS-HCL, PH 8.0, WITH 3 MICROLITER ...Details: THE PROTEIN WAS CRYSTALLIZED BY THE HANGING DROP METHOD BY MIXING 4 MICROLITER OF A SOLUTION CONTAINING 5 MG/ML PROTEIN, 0.02 M CACL2, 0.05 M NACL, 0.10 M TRIS-HCL, PH 8.0, WITH 3 MICROLITER RESERVOIR SOLUTION CONTAINING 2.1 M AMMONIUM SULFATE, 5 % ETHANOL., vapor diffusion - hanging drop | |||||||||||||||||||||||||||||||||||
| Crystal | *PLUS Density % sol: 60 % | |||||||||||||||||||||||||||||||||||
| Crystal grow | *PLUS Temperature: 293 K / Method: vapor diffusion, hanging dropDetails: Kastrup, J.S., (1997) Acta Crystallogr.,Sect.D, 53, 108. | |||||||||||||||||||||||||||||||||||
| Components of the solutions | *PLUS
|
-Data collection
| Diffraction | Mean temperature: 277 K |
|---|---|
| Diffraction source | Source:  SYNCHROTRON / Site: SYNCHROTRON / Site:  EMBL/DESY, HAMBURG EMBL/DESY, HAMBURG  / Beamline: X11 / Wavelength: 0.927 / Beamline: X11 / Wavelength: 0.927 |
| Detector | Type: MARRESEARCH / Detector: IMAGE PLATE / Date: May 1, 1995 |
| Radiation | Monochromatic (M) / Laue (L): M / Scattering type: x-ray |
| Radiation wavelength | Wavelength: 0.927 Å / Relative weight: 1 |
| Reflection | Resolution: 2→15 Å / Num. obs: 10723 / % possible obs: 97.2 % / Observed criterion σ(I): 0 / Redundancy: 4.7 % / Biso Wilson estimate: 23 Å2 / Rsym value: 0.062 / Net I/σ(I): 11.4 |
| Reflection shell | Resolution: 2→2.05 Å / Redundancy: 5 % / Mean I/σ(I) obs: 3.4 / Rsym value: 0.221 / % possible all: 98.3 |
| Reflection | *PLUS Rmerge(I) obs: 0.062 |
| Reflection shell | *PLUS Rmerge(I) obs: 0.221 |
- Processing
Processing
| Software |
| ||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Refinement | Method to determine structure:  MOLECULAR REPLACEMENT MOLECULAR REPLACEMENTStarting model: PDB ENTRY 2MSB, ONE MONOMER Resolution: 2→25 Å / Isotropic thermal model: TNT BCORREL 1.0 / Cross valid method: A POSTERIORI / σ(F): 0 / Stereochemistry target values: TNT PROTGEO Details: THE STRUCTURE WAS INITIALLY REFINED TO AN R-VALUE OF 34.7 % USING X-PLOR.
| ||||||||||||||||||||||||||||||||||||||||||||||||||
| Solvent computation | Solvent model: MOEWS AND KRETSINGER (J.MOL.BIOL. (1995) 91, 201-228) Bsol: 292 Å2 / ksol: 0.87 e/Å3 | ||||||||||||||||||||||||||||||||||||||||||||||||||
| Refinement step | Cycle: LAST / Resolution: 2→25 Å
| ||||||||||||||||||||||||||||||||||||||||||||||||||
| Refine LS restraints |
| ||||||||||||||||||||||||||||||||||||||||||||||||||
| Software | *PLUS Name: TNT / Version: 5E / Classification: refinement | ||||||||||||||||||||||||||||||||||||||||||||||||||
| Refinement | *PLUS Rfactor Rfree: 0.267 | ||||||||||||||||||||||||||||||||||||||||||||||||||
| Solvent computation | *PLUS | ||||||||||||||||||||||||||||||||||||||||||||||||||
| Displacement parameters | *PLUS | ||||||||||||||||||||||||||||||||||||||||||||||||||
| Refine LS restraints | *PLUS
|
 Movie
Movie Controller
Controller












 PDBj
PDBj
















