[English] 日本語
 Yorodumi
Yorodumi- PDB-1tc2: TERNARY SUBSTRATE COMPLEX OF THE HYPOXANTHINE PHOSPHORIBOSYLTRANS... -
+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 1tc2 | ||||||
|---|---|---|---|---|---|---|---|
| Title | TERNARY SUBSTRATE COMPLEX OF THE HYPOXANTHINE PHOSPHORIBOSYLTRANSFERASE FROM TRYPANOSOMA CRUZI | ||||||
 Components Components | PROTEIN (HYPOXANTHINE PHOSPHORIBOSYLTRANSFERASE) | ||||||
 Keywords Keywords | TRANSFERASE / GLYCOSYLTRANSFERASE / PHOSPHORIBOSYLTRANSFERASE / NUCLEOTIDE METABOLISM / PURINE SALVAGE / TERNARY COMPLEX / NEAR TRANSITION STATE | ||||||
| Function / homology |  Function and homology information Function and homology informationhypoxanthine phosphoribosyltransferase / guanine salvage / hypoxanthine metabolic process / hypoxanthine phosphoribosyltransferase activity / GMP salvage / IMP salvage / purine ribonucleoside salvage / nucleotide binding / magnesium ion binding / cytosol Similarity search - Function | ||||||
| Biological species |  | ||||||
| Method |  X-RAY DIFFRACTION / X-RAY DIFFRACTION /  SYNCHROTRON / SYNCHROTRON /  MOLECULAR REPLACEMENT / Resolution: 1.81 Å MOLECULAR REPLACEMENT / Resolution: 1.81 Å | ||||||
 Authors Authors | Focia, P.J. / Craig III, S.P. / Eakin, A.E. | ||||||
 Citation Citation |  Journal: Biochemistry / Year: 1998 Journal: Biochemistry / Year: 1998Title: Approaching the transition state in the crystal structure of a phosphoribosyltransferase. Authors: Focia, P.J. / Craig III, S.P. / Eakin, A.E. | ||||||
| History |
|
- Structure visualization
Structure visualization
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  1tc2.cif.gz 1tc2.cif.gz | 95.6 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb1tc2.ent.gz pdb1tc2.ent.gz | 71.4 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  1tc2.json.gz 1tc2.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/tc/1tc2 https://data.pdbj.org/pub/pdb/validation_reports/tc/1tc2 ftp://data.pdbj.org/pub/pdb/validation_reports/tc/1tc2 ftp://data.pdbj.org/pub/pdb/validation_reports/tc/1tc2 | HTTPS FTP |
|---|
-Related structure data
| Related structure data |  1tc1S S: Starting model for refinement |
|---|---|
| Similar structure data |
- Links
Links
- Assembly
Assembly
| Deposited unit | 
| ||||||||
|---|---|---|---|---|---|---|---|---|---|
| 1 |
| ||||||||
| Unit cell |
| ||||||||
| Noncrystallographic symmetry (NCS) | NCS oper: (Code: given Matrix: (0.865547, 0.408421, 0.289862), Vector: |
- Components
Components
-Protein / Sugars , 2 types, 4 molecules AB

| #1: Protein | Mass: 25595.404 Da / Num. of mol.: 2 Source method: isolated from a genetically manipulated source Source: (gene. exp.)   References: UniProt: Q27796, UniProt: Q4DRC4*PLUS, hypoxanthine phosphoribosyltransferase #4: Sugar | |
|---|
-Non-polymers , 4 types, 199 molecules 
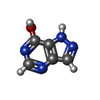





| #2: Chemical | | #3: Chemical | #5: Chemical | ChemComp-MN / | #6: Water | ChemComp-HOH / | |
|---|
-Experimental details
-Experiment
| Experiment | Method:  X-RAY DIFFRACTION / Number of used crystals: 1 X-RAY DIFFRACTION / Number of used crystals: 1 |
|---|
- Sample preparation
Sample preparation
| Crystal | Density Matthews: 2.03 Å3/Da / Density % sol: 39.35 % | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Crystal grow | pH: 4.6 / Details: pH 4.6 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Crystal grow | *PLUS Temperature: 4 ℃ / pH: 6.8 / Method: vapor diffusion, hanging drop | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Components of the solutions | *PLUS
|
-Data collection
| Diffraction | Mean temperature: 100 K |
|---|---|
| Diffraction source | Source:  SYNCHROTRON / Site: SYNCHROTRON / Site:  SSRL SSRL  / Beamline: BL7-1 / Wavelength: 1.08 / Beamline: BL7-1 / Wavelength: 1.08 |
| Detector | Type: MARRESEARCH / Detector: IMAGE PLATE / Date: Jul 4, 1997 |
| Radiation | Protocol: SINGLE WAVELENGTH / Monochromatic (M) / Laue (L): M / Scattering type: x-ray |
| Radiation wavelength | Wavelength: 1.08 Å / Relative weight: 1 |
| Reflection | Resolution: 1.81→20.6 Å / Num. obs: 33026 / % possible obs: 88.8 % / Observed criterion σ(I): -3 / Redundancy: 3.6 % / Rsym value: 0.051 / Net I/σ(I): 20.8 |
| Reflection shell | Resolution: 1.81→1.85 Å / Mean I/σ(I) obs: 3.8 / Rsym value: 0.32 / % possible all: 80.1 |
| Reflection | *PLUS Rmerge(I) obs: 0.051 |
| Reflection shell | *PLUS % possible obs: 80.1 % / Rmerge(I) obs: 0.32 |
- Processing
Processing
| Software |
| ||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Refinement | Method to determine structure:  MOLECULAR REPLACEMENT MOLECULAR REPLACEMENTStarting model: PDB ENTRY 1TC1 Resolution: 1.81→6 Å / Cross valid method: THROUGHOUT / σ(F): 1 Details: PATCH STATEMENTS WERE USED FOR CIS PEPTIDES AND METAL-OXYGEN BONDS SER 88 IS POORLY ORDERED IN SUBUNIT A THE ENERGIES FOR METAL-OXYGEN BOND LENGTHS AND ANGLES WERE SET ARTIFICIALLY LOW ...Details: PATCH STATEMENTS WERE USED FOR CIS PEPTIDES AND METAL-OXYGEN BONDS SER 88 IS POORLY ORDERED IN SUBUNIT A THE ENERGIES FOR METAL-OXYGEN BOND LENGTHS AND ANGLES WERE SET ARTIFICIALLY LOW DURING REFINEMENT IN ORDER TO ALLOW THESE ATOMS MORE FREEDOM TO ADJUST
| ||||||||||||||||||||||||
| Refinement step | Cycle: LAST / Resolution: 1.81→6 Å
| ||||||||||||||||||||||||
| Refine LS restraints |
| ||||||||||||||||||||||||
| LS refinement shell | Resolution: 1.81→1.85 Å / Total num. of bins used: 15
| ||||||||||||||||||||||||
| Xplor file |
| ||||||||||||||||||||||||
| Software | *PLUS Name:  X-PLOR / Version: 3.851 / Classification: refinement X-PLOR / Version: 3.851 / Classification: refinement | ||||||||||||||||||||||||
| Refine LS restraints | *PLUS
|
 Movie
Movie Controller
Controller












 PDBj
PDBj




