[English] 日本語
 Yorodumi
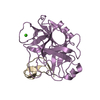
Yorodumi- PDB-1ppe: THE REFINED 2.0 ANGSTROMS X-RAY CRYSTAL STRUCTURE OF THE COMPLEX ... -
+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 1ppe | ||||||
|---|---|---|---|---|---|---|---|
| Title | THE REFINED 2.0 ANGSTROMS X-RAY CRYSTAL STRUCTURE OF THE COMPLEX FORMED BETWEEN BOVINE BETA-TRYPSIN AND CMTI-I, A TRYPSIN INHIBITOR FROM SQUASH SEEDS (CUCURBITA MAXIMA): TOPOLOGICAL SIMILARITY OF THE SQUASH SEED INHIBITORS WITH THE CARBOXYPEPTIDASE A INHIBITOR FROM POTATOES | ||||||
 Components Components |
| ||||||
 Keywords Keywords | HYDROLASE/HYDROLASE INHIBITOR / SERINE PROTEINASE / HYDROLASE-HYDROLASE INHIBITOR COMPLEX | ||||||
| Function / homology |  Function and homology information Function and homology informationtrypsin / serpin family protein binding / serine protease inhibitor complex / digestion / serine-type endopeptidase inhibitor activity / endopeptidase activity / serine-type endopeptidase activity / proteolysis / : / extracellular region / metal ion binding Similarity search - Function | ||||||
| Biological species |  | ||||||
| Method |  X-RAY DIFFRACTION / Resolution: 2 Å X-RAY DIFFRACTION / Resolution: 2 Å | ||||||
 Authors Authors | Bode, W. / Huber, R. | ||||||
 Citation Citation |  Journal: FEBS Lett. / Year: 1989 Journal: FEBS Lett. / Year: 1989Title: The refined 2.0 A X-ray crystal structure of the complex formed between bovine beta-trypsin and CMTI-I, a trypsin inhibitor from squash seeds (Cucurbita maxima). Topological similarity of the ...Title: The refined 2.0 A X-ray crystal structure of the complex formed between bovine beta-trypsin and CMTI-I, a trypsin inhibitor from squash seeds (Cucurbita maxima). Topological similarity of the squash seed inhibitors with the carboxypeptidase A inhibitor from potatoes Authors: Bode, W. / Greyling, H.J. / Huber, R. / Otlewski, J. / Wilusz, T. #1:  Journal: Curr.Opin.Struct.Biol. / Year: 1991 Journal: Curr.Opin.Struct.Biol. / Year: 1991Title: Ligand Binding: Proteinase-Protein Inhibitor Interactions Authors: Bode, W. / Huber, R. #2:  Journal: J.Mol.Biol. / Year: 1989 Journal: J.Mol.Biol. / Year: 1989Title: Nuclear Magnetic Resonance Solution and X-Ray Structures of Squash Trypsin Inhibitor Exhibit the Same Conformation of the Proteinase Binding Loop Authors: Holak, T.A. / Bode, W. / Huber, R. / Otlewski, J. / Wilusz, T. #3:  Journal: Biochem.Biophys.Res.Commun. / Year: 1985 Journal: Biochem.Biophys.Res.Commun. / Year: 1985Title: The Squash Family of Serine Proteinase Inhibitors. Amino Acid Sequences and Association Equilibrium Constants of Inhibitors from Squash, Summer Squash, Zucchini, and Cucumber Seeds Authors: Wieczorek, M. / Otlewski, J. / Cook, J. / Parks, K. / Leluk, J. / Wilimowska-Pelc, A. / Polanowski, A. / Wilusz, T. / Laskowski Junior, M. #4:  Journal: Hoppe-Seyler's Z.Physiol.Chem. / Year: 1983 Journal: Hoppe-Seyler's Z.Physiol.Chem. / Year: 1983Title: Amino-Acid Sequence of Two Trypsin Isoinhibitors, Itd I and Itd III from Squash Seeds (Cucurbita Maxima) Authors: Wilusz, T. / Wieczorek, M. / Polanowski, A. / Denton, A. / Cook, J. / Laskowski Junior, M. #5:  Journal: J.Mol.Biol. / Year: 1975 Journal: J.Mol.Biol. / Year: 1975Title: The Refined Crystal Structure of Bovine Beta-Trypsin at 1.8 Angstroms Resolution. Crystallographic Refinement, Calcium Binding Site, Benzamidine Binding Site and Active Site at Ph 7.0 Authors: Bode, W. / Schwager, P. | ||||||
| History |
|
- Structure visualization
Structure visualization
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  1ppe.cif.gz 1ppe.cif.gz | 62.7 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb1ppe.ent.gz pdb1ppe.ent.gz | 45.1 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  1ppe.json.gz 1ppe.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/pp/1ppe https://data.pdbj.org/pub/pdb/validation_reports/pp/1ppe ftp://data.pdbj.org/pub/pdb/validation_reports/pp/1ppe ftp://data.pdbj.org/pub/pdb/validation_reports/pp/1ppe | HTTPS FTP |
|---|
-Related structure data
| Similar structure data |
|---|
- Links
Links
- Assembly
Assembly
| Deposited unit | 
| ||||||||
|---|---|---|---|---|---|---|---|---|---|
| 1 |
| ||||||||
| Unit cell |
| ||||||||
| Atom site foot note | 1: GLU E 70 - ASP E 71 OMEGA ANGLE = 149.885 PEPTIDE BOND DEVIATES SIGNIFICANTLY FROM TRANS CONFORMATION |
- Components
Components
| #1: Protein | Mass: 23324.287 Da / Num. of mol.: 1 Source method: isolated from a genetically manipulated source Source: (gene. exp.)  |
|---|---|
| #2: Protein/peptide | Mass: 3279.919 Da / Num. of mol.: 1 Source method: isolated from a genetically manipulated source References: UniProt: P01074 |
| #3: Water | ChemComp-HOH / |
| Has protein modification | Y |
-Experimental details
-Experiment
| Experiment | Method:  X-RAY DIFFRACTION X-RAY DIFFRACTION |
|---|
- Sample preparation
Sample preparation
| Crystal | Density Matthews: 2.3 Å3/Da / Density % sol: 46.62 % | |||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Crystal grow | *PLUS Temperature: 20 ℃ / Method: vapor diffusion, hanging drop / PH range low: 5 / PH range high: 4 | |||||||||||||||
| Components of the solutions | *PLUS
|
-Data collection
| Radiation | Scattering type: x-ray |
|---|---|
| Radiation wavelength | Relative weight: 1 |
| Reflection | *PLUS Highest resolution: 2 Å / Num. obs: 14191 / % possible obs: 81 % / Num. measured all: 54864 / Rmerge(I) obs: 0.073 |
- Processing
Processing
| Software |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Refinement | Resolution: 2→6 Å / Rfactor Rwork: 0.151 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refinement step | Cycle: LAST / Resolution: 2→6 Å
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refine LS restraints |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Software | *PLUS Name: EREF / Classification: refinement | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refinement | *PLUS Highest resolution: 2 Å / Lowest resolution: 6 Å / Num. reflection obs: 13077 / Rfactor obs: 0.151 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Solvent computation | *PLUS | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Displacement parameters | *PLUS Biso mean: 15 Å2 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refine LS restraints | *PLUS Type: o_angle_d / Dev ideal: 2.7 |
 Movie
Movie Controller
Controller











 PDBj
PDBj


