[English] 日本語
 Yorodumi
Yorodumi- PDB-1ovd: THE K136E MUTANT OF LACTOCOCCUS LACTIS DIHYDROOROTATE DEHYDROGENA... -
+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 1ovd | ||||||
|---|---|---|---|---|---|---|---|
| Title | THE K136E MUTANT OF LACTOCOCCUS LACTIS DIHYDROOROTATE DEHYDROGENASE A IN COMPLEX WITH OROTATE | ||||||
 Components Components | DIHYDROOROTATE DEHYDROGENASE A | ||||||
 Keywords Keywords | OXIDOREDUCTASE / HOMODIMER / ALPHA-BETA BARREL / FLAVOPROTEIN / OROTATE COMPLEX / MUTANT ENZYME | ||||||
| Function / homology |  Function and homology information Function and homology informationdihydroorotate dehydrogenase (fumarate) / dihydroorotate dehydrogenase (fumarate) activity / 'de novo' UMP biosynthetic process / 'de novo' pyrimidine nucleobase biosynthetic process / cytoplasm Similarity search - Function | ||||||
| Biological species |  Lactococcus lactis (lactic acid bacteria) Lactococcus lactis (lactic acid bacteria) | ||||||
| Method |  X-RAY DIFFRACTION / X-RAY DIFFRACTION /  SYNCHROTRON / SYNCHROTRON /  MOLECULAR REPLACEMENT / Resolution: 2.25 Å MOLECULAR REPLACEMENT / Resolution: 2.25 Å | ||||||
 Authors Authors | Norager, S. / Arent, S. / Bjornberg, O. / Ottosen, M. / Lo Leggio, L. / Jensen, K.F. / Larsen, S. | ||||||
 Citation Citation |  Journal: J.Biol.Chem. / Year: 2003 Journal: J.Biol.Chem. / Year: 2003Title: Lactococcus lactis dihydroorotate dehydrogenase A mutants reveal important facets of the enzymatic function. Authors: Norager, S. / Arent, S. / Bjornberg, O. / Ottesen, M. / Lo Leggio, L. / Jensen, K.F. / Larsen, S. #1:  Journal: Biochemistry / Year: 1997 Journal: Biochemistry / Year: 1997Title: Active Site of Dihydroorotate Dehydrogenase A from Lactococcus Lactis Investigated by Chemical Modification and Mutagenesis Authors: Bjornberg, O. / Rowland, P. / Larsen, S. / Jensen, K.F. #2:  Journal: Protein Sci. / Year: 1998 Journal: Protein Sci. / Year: 1998Title: The Crystals Structure of Lactococcus Lactis Dihydroorotate Dehydrogenase A Complexed with the Enzyme Reaction Product Throws Light on its Enzymatic Function Authors: Rowland, P. / Bjornberg, O. / Nielsen, F.S. / Jensen, K.F. / Larsen, S. #3:  Journal: Structure / Year: 1997 Journal: Structure / Year: 1997Title: The Crystals Structure of the Flavin Containing Enzyme Dihydroorotate Dehydrogenase A from Lactococcus Lactis Authors: Rowland, P. / Nielsen, F.S. / Jensen, K.F. / Larsen, S. #4:  Journal: Protein Sci. / Year: 1996 Journal: Protein Sci. / Year: 1996Title: Purification and Characterisation of Dihydroorotate Dehydrogenase A from Lactococcus Lactis, Crystallisation and Preliminary X-Ray Diffraction Studies of the Enzyme Authors: Nielsen, F.S. / Rowland, P. / Larsen, S. / Jensen, K.F. | ||||||
| History |
|
- Structure visualization
Structure visualization
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  1ovd.cif.gz 1ovd.cif.gz | 146.7 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb1ovd.ent.gz pdb1ovd.ent.gz | 112.6 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  1ovd.json.gz 1ovd.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/ov/1ovd https://data.pdbj.org/pub/pdb/validation_reports/ov/1ovd ftp://data.pdbj.org/pub/pdb/validation_reports/ov/1ovd ftp://data.pdbj.org/pub/pdb/validation_reports/ov/1ovd | HTTPS FTP |
|---|
-Related structure data
| Related structure data |  1jqvC  1jqxC  1jrbC  1jrcC  1jubC  1jueC  1dorS S: Starting model for refinement C: citing same article ( |
|---|---|
| Similar structure data |
- Links
Links
- Assembly
Assembly
| Deposited unit | 
| ||||||||
|---|---|---|---|---|---|---|---|---|---|
| 1 |
| ||||||||
| Unit cell |
| ||||||||
| Details | The asymmetric unit contains the biological homodimer |
- Components
Components
-Protein , 1 types, 2 molecules AB
| #1: Protein | Mass: 34242.102 Da / Num. of mol.: 2 / Mutation: K136E Source method: isolated from a genetically manipulated source Source: (gene. exp.)  Lactococcus lactis (lactic acid bacteria) Lactococcus lactis (lactic acid bacteria)Gene: PYRDA / Plasmid: PUHE23 / Production host:  References: UniProt: P54321, UniProt: A2RJT9*PLUS, EC: 1.3.3.1 |
|---|
-Non-polymers , 5 types, 498 molecules 

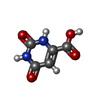






| #2: Chemical | | #3: Chemical | #4: Chemical | #5: Chemical | ChemComp-GOL / #6: Water | ChemComp-HOH / | |
|---|
-Experimental details
-Experiment
| Experiment | Method:  X-RAY DIFFRACTION / Number of used crystals: 1 X-RAY DIFFRACTION / Number of used crystals: 1 |
|---|
- Sample preparation
Sample preparation
| Crystal | Density Matthews: 2.41 Å3/Da / Density % sol: 48.61 % | |||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Crystal grow | Temperature: 293 K / Method: vapor diffusion, hanging drop / pH: 8.5 Details: PEG 6K, Sodium acetate, Tris-HCl, pH 8.5, VAPOR DIFFUSION, HANGING DROP, temperature 293K | |||||||||||||||||||||||||||||||||||
| Crystal grow | *PLUS pH: 7 / Method: vapor diffusion | |||||||||||||||||||||||||||||||||||
| Components of the solutions | *PLUS
|
-Data collection
| Diffraction | Mean temperature: 100 K |
|---|---|
| Diffraction source | Source:  SYNCHROTRON / Site: SYNCHROTRON / Site:  MAX II MAX II  / Beamline: I711 / Wavelength: 1.098 Å / Beamline: I711 / Wavelength: 1.098 Å |
| Detector | Type: MARRESEARCH / Detector: CCD / Date: Sep 26, 2001 / Details: Bent mirror |
| Radiation | Monochromator: Si(III) single crystal / Protocol: SINGLE WAVELENGTH / Monochromatic (M) / Laue (L): M / Scattering type: x-ray |
| Radiation wavelength | Wavelength: 1.098 Å / Relative weight: 1 |
| Reflection | Resolution: 2.25→12 Å / Num. all: 34158 / Num. obs: 33990 / % possible obs: 100 % / Observed criterion σ(F): 0 / Observed criterion σ(I): -3 / Redundancy: 4.3 % / Biso Wilson estimate: 6.2 Å2 / Rmerge(I) obs: 0.089 / Net I/σ(I): 15.3 |
| Reflection shell | Resolution: 2.25→2.29 Å / Rmerge(I) obs: 0.282 / Mean I/σ(I) obs: 4.93 / Num. unique all: 1709 / % possible all: 100 |
| Reflection | *PLUS Num. obs: 34158 / % possible obs: 100 % / Num. measured all: 146256 |
| Reflection shell | *PLUS % possible obs: 100 % / Mean I/σ(I) obs: 4.9 |
- Processing
Processing
| Software |
| ||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Refinement | Method to determine structure:  MOLECULAR REPLACEMENT MOLECULAR REPLACEMENTStarting model: Lactococcus lactis DHODA PDB ID 1DOR Resolution: 2.25→12 Å / Rfactor Rfree error: 0.005 / Data cutoff high absF: 156760.05 / Data cutoff high rms absF: 156760.05 / Data cutoff low absF: 0 / Isotropic thermal model: isotropic / Cross valid method: THROUGHOUT / σ(F): 0 / Stereochemistry target values: ENGH & HUBER
| ||||||||||||||||||||||||||||||||||||
| Solvent computation | Solvent model: FLAT / Bsol: 42.7672 Å2 / ksol: 0.353933 e/Å3 | ||||||||||||||||||||||||||||||||||||
| Displacement parameters | Biso mean: 19.5 Å2
| ||||||||||||||||||||||||||||||||||||
| Refine analyze |
| ||||||||||||||||||||||||||||||||||||
| Refinement step | Cycle: LAST / Resolution: 2.25→12 Å
| ||||||||||||||||||||||||||||||||||||
| Refine LS restraints |
| ||||||||||||||||||||||||||||||||||||
| LS refinement shell | Resolution: 2.25→2.39 Å / Rfactor Rfree error: 0.015 / Total num. of bins used: 10
| ||||||||||||||||||||||||||||||||||||
| Xplor file |
| ||||||||||||||||||||||||||||||||||||
| Refine LS restraints | *PLUS
|
 Movie
Movie Controller
Controller















 PDBj
PDBj



