[English] 日本語
 Yorodumi
Yorodumi- PDB-1o53: Solution structure of the N-terminal membrane anchor of E. coli e... -
+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 1o53 | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Title | Solution structure of the N-terminal membrane anchor of E. coli enzyme IIA(Glucose) | |||||||||
 Components Components | PTS system, glucose-specific IIA component | |||||||||
 Keywords Keywords | TRANSFERASE / amphipathic helix | |||||||||
| Function / homology |  Function and homology information Function and homology informationnegative regulation of carbohydrate metabolic process / phosphoenolpyruvate-dependent sugar phosphotransferase system / extrinsic component of cytoplasmic side of plasma membrane / kinase activity / membrane / metal ion binding / cytoplasm / cytosol Similarity search - Function | |||||||||
| Method | SOLUTION NMR / simulated annealing | |||||||||
 Authors Authors | Wang, G. / Keifer, P.A. / Peterkofsky, A. | |||||||||
 Citation Citation |  Journal: Protein Sci. / Year: 2003 Journal: Protein Sci. / Year: 2003Title: Solution structure of the N-terminal amphitropic domain of Escherichia coli glucose-specific enzyme IIA in membrane-mimetic micelles Authors: Wang, G. / Keifer, P.A. / Peterkofsky, A. | |||||||||
| History |
|
- Structure visualization
Structure visualization
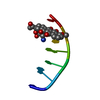
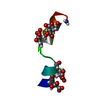
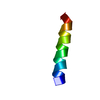
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  1o53.cif.gz 1o53.cif.gz | 89.8 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb1o53.ent.gz pdb1o53.ent.gz | 62.7 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  1o53.json.gz 1o53.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/o5/1o53 https://data.pdbj.org/pub/pdb/validation_reports/o5/1o53 ftp://data.pdbj.org/pub/pdb/validation_reports/o5/1o53 ftp://data.pdbj.org/pub/pdb/validation_reports/o5/1o53 | HTTPS FTP |
|---|
-Related structure data
| Related structure data | |
|---|---|
| Similar structure data |
- Links
Links
- Assembly
Assembly
| Deposited unit | 
| |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| 1 |
| |||||||||
| NMR ensembles |
|
- Components
Components
| #1: Protein/peptide | Mass: 1696.983 Da / Num. of mol.: 1 Fragment: N-terminal membrane anchor, residues 1-15 of enzyme IIA(Glucose) Source method: obtained synthetically Details: The peptide was synthesized using the solid-phase method and purified by HPLC. The sequence of the peptide is naturally found in Escherichia coli (bacteria). References: UniProt: P08837, UniProt: P0A284*PLUS, protein-Npi-phosphohistidine-sugar phosphotransferase |
|---|
-Experimental details
-Experiment
| Experiment | Method: SOLUTION NMR |
|---|---|
| NMR experiment | Type: 2D NOESY, TOCSY, DQF-COSY |
| NMR details | Text: This structure was determined using standard 2D homonuclear NMR techniques. |
- Sample preparation
Sample preparation
| Details | Contents: natural abundance synthetic peptide 5 mM peptide and 50 mM dihexanoyl phosphatidylglycerol Solvent system: 90% H2O and 10% D2O |
|---|---|
| Sample conditions | Ionic strength: no buffer / pH: 5.4 / Pressure: ambient / Temperature: 298 K |
| Crystal grow | *PLUS Method: other / Details: NMR |
-NMR measurement
| NMR spectrometer | Type: Varian INOVA / Manufacturer: Varian / Model: INOVA / Field strength: 600 MHz |
|---|
- Processing
Processing
| NMR software |
| ||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Refinement | Method: simulated annealing / Software ordinal: 1 Details: The structures are based on 146 distances derived from the NOESY spectra. No dihedral angles or hydrogen-bond restraints were applied. | ||||||||||||||||||||
| NMR representative | Selection criteria: most resemble the average structure. | ||||||||||||||||||||
| NMR ensemble | Conformer selection criteria: No NOE violations greater than 0.20 A; rms difference for bond deviations from ideality less than 0.01 A; rms difference for angle deviations from ideality less than 2 ...Conformer selection criteria: No NOE violations greater than 0.20 A; rms difference for bond deviations from ideality less than 0.01 A; rms difference for angle deviations from ideality less than 2 degrees; Structures with the lowerest energies in the ensemble; Structures most resemble the average structure. Conformers calculated total number: 100 / Conformers submitted total number: 20 |
 Movie
Movie Controller
Controller









 PDBj
PDBj XPLOR-NIH
XPLOR-NIH