[English] 日本語
 Yorodumi
Yorodumi- PDB-1joc: EEA1 homodimer of C-terminal FYVE domain bound to inositol 1,3-di... -
+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 1joc | ||||||
|---|---|---|---|---|---|---|---|
| Title | EEA1 homodimer of C-terminal FYVE domain bound to inositol 1,3-diphosphate | ||||||
 Components Components | Early Endosomal Autoantigen 1 | ||||||
 Keywords Keywords | MEMBRANE PROTEIN / FYVE domain / inositol 3-phosphate binding | ||||||
| Function / homology |  Function and homology information Function and homology informationsynaptic vesicle to endosome fusion / postsynaptic early endosome / 1-phosphatidylinositol binding / Toll Like Receptor 9 (TLR9) Cascade / chemical synaptic transmission, postsynaptic / axonal spine / presynaptic endosome / host-mediated perturbation of viral process / early endosome to late endosome transport / vesicle fusion ...synaptic vesicle to endosome fusion / postsynaptic early endosome / 1-phosphatidylinositol binding / Toll Like Receptor 9 (TLR9) Cascade / chemical synaptic transmission, postsynaptic / axonal spine / presynaptic endosome / host-mediated perturbation of viral process / early endosome to late endosome transport / vesicle fusion / GTP-dependent protein binding / recycling endosome / Schaffer collateral - CA1 synapse / endocytosis / early endosome membrane / calmodulin binding / early endosome / glutamatergic synapse / protein homodimerization activity / extracellular exosome / zinc ion binding / cytosol Similarity search - Function | ||||||
| Biological species |  Homo sapiens (human) Homo sapiens (human) | ||||||
| Method |  X-RAY DIFFRACTION / X-RAY DIFFRACTION /  SYNCHROTRON / SYNCHROTRON /  MAD / Resolution: 2.2 Å MAD / Resolution: 2.2 Å | ||||||
 Authors Authors | Dumas, J.J. / Merithew, E. / Rajamani, D. / Hayes, S. / Lawe, D. / Corvera, S. / Lambright, D.G. | ||||||
 Citation Citation |  Journal: Mol.Cell / Year: 2001 Journal: Mol.Cell / Year: 2001Title: Multivalent endosome targeting by homodimeric EEA1. Authors: Dumas, J.J. / Merithew, E. / Sudharshan, E. / Rajamani, D. / Hayes, S. / Lawe, D. / Corvera, S. / Lambright, D.G. | ||||||
| History |
|
- Structure visualization
Structure visualization
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  1joc.cif.gz 1joc.cif.gz | 62.2 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb1joc.ent.gz pdb1joc.ent.gz | 46.3 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  1joc.json.gz 1joc.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/jo/1joc https://data.pdbj.org/pub/pdb/validation_reports/jo/1joc ftp://data.pdbj.org/pub/pdb/validation_reports/jo/1joc ftp://data.pdbj.org/pub/pdb/validation_reports/jo/1joc | HTTPS FTP |
|---|
-Related structure data
| Similar structure data |
|---|
- Links
Links
- Assembly
Assembly
| Deposited unit | 
| ||||||||
|---|---|---|---|---|---|---|---|---|---|
| 1 |
| ||||||||
| Unit cell |
|
- Components
Components
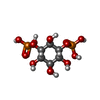
| #1: Protein | Mass: 14139.077 Da / Num. of mol.: 2 / Fragment: C-terminal domain, FYVE domain Source method: isolated from a genetically manipulated source Source: (gene. exp.)  Homo sapiens (human) / Plasmid: pET15b / Species (production host): Escherichia coli / Production host: Homo sapiens (human) / Plasmid: pET15b / Species (production host): Escherichia coli / Production host:  #2: Chemical | ChemComp-ZN / #3: Chemical | #4: Water | ChemComp-HOH / | |
|---|
-Experimental details
-Experiment
| Experiment | Method:  X-RAY DIFFRACTION / Number of used crystals: 1 X-RAY DIFFRACTION / Number of used crystals: 1 |
|---|
- Sample preparation
Sample preparation
| Crystal | Density Matthews: 2.42 Å3/Da / Density % sol: 49.15 % | ||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Crystal grow | Method: vapor diffusion, hanging drop / pH: 7 Details: 11% PEG 4000, 50 mM HEPES, 60 mM ammonium acetate, 10% glycerol, and 1.5 mM Ins(1,3)P2, pH 7.0, VAPOR DIFFUSION, HANGING DROP | ||||||||||||||||||||||||||||||||||||
| Crystal grow | *PLUS Temperature: 4 ℃ | ||||||||||||||||||||||||||||||||||||
| Components of the solutions | *PLUS
|
-Data collection
| Diffraction | Mean temperature: 100 K | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Diffraction source | Source:  SYNCHROTRON / Site: SYNCHROTRON / Site:  NSLS NSLS  / Beamline: X12C / Wavelength: 1.28322, 1.28356, 1.23985 / Beamline: X12C / Wavelength: 1.28322, 1.28356, 1.23985 | ||||||||||||
| Detector | Type: BRANDEIS - B1 / Detector: CCD / Date: Mar 20, 2001 | ||||||||||||
| Radiation | Protocol: MAD / Monochromatic (M) / Laue (L): M / Scattering type: x-ray | ||||||||||||
| Radiation wavelength |
|
- Processing
Processing
| Software |
| |||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Refinement | Method to determine structure:  MAD / Resolution: 2.2→20 Å / Rfactor Rfree: 0.281 / Rfactor Rwork: 0.221 / Stereochemistry target values: Engh & Huber MAD / Resolution: 2.2→20 Å / Rfactor Rfree: 0.281 / Rfactor Rwork: 0.221 / Stereochemistry target values: Engh & HuberDetails: Residues 1287 and 1288 were missing from the electron density. | |||||||||||||||
| Refinement step | Cycle: LAST / Resolution: 2.2→20 Å
| |||||||||||||||
| Refine LS restraints |
| |||||||||||||||
| Software | *PLUS Name:  X-PLOR / Version: 3.1 / Classification: refinement X-PLOR / Version: 3.1 / Classification: refinement | |||||||||||||||
| Refinement | *PLUS Highest resolution: 2.2 Å / Lowest resolution: 20 Å / % reflection Rfree: 5 % / Rfactor obs: 0.221 | |||||||||||||||
| Solvent computation | *PLUS | |||||||||||||||
| Displacement parameters | *PLUS | |||||||||||||||
| Refine LS restraints | *PLUS Type: x_angle_deg / Dev ideal: 1.1 |
 Movie
Movie Controller
Controller








 PDBj
PDBj