+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 1in1 | ||||||
|---|---|---|---|---|---|---|---|
| Title | NMR STRUCTURE OF HUMAN DNA LIGASE IIIALPHA BRCT DOMAIN | ||||||
 Components Components | DNA LIGASE III | ||||||
 Keywords Keywords | LIGASE / parallel beta sheet | ||||||
| Function / homology |  Function and homology information Function and homology informationDNA ligase III-XRCC1 complex / negative regulation of mitochondrial DNA replication / DNA ligase activity / DNA ligase (ATP) / DNA ligase (ATP) activity / Strand-asynchronous mitochondrial DNA replication / double-strand break repair via alternative nonhomologous end joining / lagging strand elongation / HDR through MMEJ (alt-NHEJ) / Resolution of AP sites via the single-nucleotide replacement pathway ...DNA ligase III-XRCC1 complex / negative regulation of mitochondrial DNA replication / DNA ligase activity / DNA ligase (ATP) / DNA ligase (ATP) activity / Strand-asynchronous mitochondrial DNA replication / double-strand break repair via alternative nonhomologous end joining / lagging strand elongation / HDR through MMEJ (alt-NHEJ) / Resolution of AP sites via the single-nucleotide replacement pathway / mitochondrial DNA repair / DNA biosynthetic process / APEX1-Independent Resolution of AP Sites via the Single Nucleotide Replacement Pathway / base-excision repair, gap-filling / Gap-filling DNA repair synthesis and ligation in GG-NER / mitochondrion organization / base-excision repair / double-strand break repair via homologous recombination / Gap-filling DNA repair synthesis and ligation in TC-NER / double-strand break repair / mitochondrial matrix / cell division / mitochondrion / DNA binding / zinc ion binding / nucleoplasm / ATP binding / nucleus Similarity search - Function | ||||||
| Biological species |  Homo sapiens (human) Homo sapiens (human) | ||||||
| Method | SOLUTION NMR / distance geometry, simulated annealing, torsion angle dynamics | ||||||
 Authors Authors | Krishnan, V.V. / Thornton, K.H. / Thelen, M.P. / Cosman, M. | ||||||
 Citation Citation |  Journal: Biochemistry / Year: 2001 Journal: Biochemistry / Year: 2001Title: Solution structure and backbone dynamics of the human DNA ligase IIIalpha BRCT domain Authors: Krishnan, V.V. / Thornton, K.H. / Thelen, M.P. / Cosman, M. #1:  Journal: Protein Expr.Purif. / Year: 2001 Journal: Protein Expr.Purif. / Year: 2001Title: Expression, purification, and biophysical characterization of the BRCT domain of human DNA ligase III alpha Authors: Cosman, M. / Krishnan, V.V. / Thornton, K.H. / Thelen, M.P. | ||||||
| History |
|
- Structure visualization
Structure visualization
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  1in1.cif.gz 1in1.cif.gz | 559.2 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb1in1.ent.gz pdb1in1.ent.gz | 448.4 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  1in1.json.gz 1in1.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/in/1in1 https://data.pdbj.org/pub/pdb/validation_reports/in/1in1 ftp://data.pdbj.org/pub/pdb/validation_reports/in/1in1 ftp://data.pdbj.org/pub/pdb/validation_reports/in/1in1 | HTTPS FTP |
|---|
-Related structure data
- Links
Links
- Assembly
Assembly
| Deposited unit | 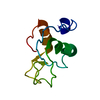
| |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| 1 |
| |||||||||
| NMR ensembles |
|
- Components
Components
| #1: Protein | Mass: 9985.422 Da / Num. of mol.: 1 / Fragment: BRCT DOMAIN Source method: isolated from a genetically manipulated source Source: (gene. exp.)  Homo sapiens (human) / Plasmid: BL21(DE3) / Production host: Homo sapiens (human) / Plasmid: BL21(DE3) / Production host:  |
|---|
-Experimental details
-Experiment
| Experiment | Method: SOLUTION NMR | ||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| NMR experiment |
| ||||||||||||||||||||||||
| NMR details | Text: The structures were determined using triple-resonance NMR spectroscopy |
- Sample preparation
Sample preparation
| Details | Contents: 50 mM NaH2PO4, 150 mM NaCl, 25 mM d10-DTT / Solvent system: 90-95% H2O and 10-5% D2O |
|---|---|
| Sample conditions | Ionic strength: 0.4-1.0 mM / pH: 6.6 / Pressure: ambient / Temperature: 288 K |
| Crystal grow | *PLUS Method: other / Details: NMR |
-NMR measurement
| NMR spectrometer | Type: Varian INOVA / Manufacturer: Varian / Model: INOVA / Field strength: 600 MHz |
|---|
- Processing
Processing
| NMR software |
| ||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Refinement | Method: distance geometry, simulated annealing, torsion angle dynamics Software ordinal: 1 Details: the structures are based on a total of 1072 restraints, 979 NOE-derived distance constraints, 25 dihedral angle restraints, 54 H bond restraints from hydrogen bonds | ||||||||||||||||||||
| NMR representative | Selection criteria: ensemble of 20 structures | ||||||||||||||||||||
| NMR ensemble | Conformer selection criteria: structures with the least restraint violations, target function Conformers calculated total number: 299 / Conformers submitted total number: 20 |
 Movie
Movie Controller
Controller













 PDBj
PDBj





 NMRPipe
NMRPipe