+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 1fn6 | ||||||
|---|---|---|---|---|---|---|---|
| Title | CRYSTAL STRUCTURE OF PORCINE BETA TRYPSIN WITH 0.1% POLYDOCANOL | ||||||
 Components Components | TRYPSIN | ||||||
 Keywords Keywords | HYDROLASE / SERINE PROTEASE | ||||||
| Function / homology |  Function and homology information Function and homology informationtrypsin / digestion / serine-type endopeptidase activity / proteolysis / : / metal ion binding Similarity search - Function | ||||||
| Biological species |  | ||||||
| Method |  X-RAY DIFFRACTION / X-RAY DIFFRACTION /  MOLECULAR REPLACEMENT / Resolution: 1.8 Å MOLECULAR REPLACEMENT / Resolution: 1.8 Å | ||||||
 Authors Authors | Deepthi, S. / Johnson, A. / Pattabhi, V. | ||||||
 Citation Citation |  Journal: Acta Crystallogr.,Sect.D / Year: 2001 Journal: Acta Crystallogr.,Sect.D / Year: 2001Title: Structures of porcine beta-trypsin-detergent complexes: the stabilization of proteins through hydrophilic binding of polydocanol. Authors: Deepthi, S. / Johnson, A. / Pattabhi, V. #1:  Journal: BIOCHIM.BIOPHYS.ACTA / Year: 1999 Journal: BIOCHIM.BIOPHYS.ACTA / Year: 1999Title: Crystal Structure at 1.63 Angstroms Resolution of the Native Form of Porcine Beta Trypsin : Revealing an Acetate Ion Binding Site and Functional Water Network Authors: Johnson, A. / Gautham, N. / Pattabhi, V. #2:  Journal: Acta Crystallogr.,Sect.D / Year: 1997 Journal: Acta Crystallogr.,Sect.D / Year: 1997Title: The First Structure at 1.8 Angstroms Resolution of an Active Autolysate Form of Porcine Alpha Trypsin Authors: Johnson, A. / Krishnaswamy, S. / Sundaram, P.V. / Pattabhi, V. | ||||||
| History |
|
- Structure visualization
Structure visualization
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  1fn6.cif.gz 1fn6.cif.gz | 59.5 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb1fn6.ent.gz pdb1fn6.ent.gz | 42.4 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  1fn6.json.gz 1fn6.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/fn/1fn6 https://data.pdbj.org/pub/pdb/validation_reports/fn/1fn6 ftp://data.pdbj.org/pub/pdb/validation_reports/fn/1fn6 ftp://data.pdbj.org/pub/pdb/validation_reports/fn/1fn6 | HTTPS FTP |
|---|
-Related structure data
| Related structure data |  1fmgC  1fniC  1qquS S: Starting model for refinement C: citing same article ( |
|---|---|
| Similar structure data |
- Links
Links
- Assembly
Assembly
| Deposited unit | 
| ||||||||
|---|---|---|---|---|---|---|---|---|---|
| 1 |
| ||||||||
| Unit cell |
|
- Components
Components
-Protein , 1 types, 1 molecules A
| #1: Protein | Mass: 23491.525 Da / Num. of mol.: 1 / Source method: isolated from a natural source / Source: (natural)  |
|---|
-Non-polymers , 5 types, 161 molecules 


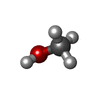





| #2: Chemical | ChemComp-CA / | ||||
|---|---|---|---|---|---|
| #3: Chemical | ChemComp-SO4 / | ||||
| #4: Chemical | | #5: Chemical | ChemComp-MOH / | #6: Water | ChemComp-HOH / | |
-Details
| Has protein modification | Y |
|---|---|
| Sequence details | THE 223 AMINO ACIDS OF TRYPSIN ARE IDENTIFIED |
-Experimental details
-Experiment
| Experiment | Method:  X-RAY DIFFRACTION / Number of used crystals: 1 X-RAY DIFFRACTION / Number of used crystals: 1 |
|---|
- Sample preparation
Sample preparation
| Crystal | Density Matthews: 2.12 Å3/Da / Density % sol: 41.86 % | ||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Crystal grow | Method: vapor diffusion, hanging drop / pH: 6.7 / Details: pH 6.70, VAPOR DIFFUSION, HANGING DROP | ||||||||||||||||||||||||||||||||||||||||||
| Crystal grow | *PLUS Temperature: 293 K / pH: 6.7 | ||||||||||||||||||||||||||||||||||||||||||
| Components of the solutions | *PLUS
|
-Data collection
| Diffraction | Mean temperature: 293 K |
|---|---|
| Diffraction source | Source:  ROTATING ANODE / Type: OTHER / Wavelength: 1.54 ROTATING ANODE / Type: OTHER / Wavelength: 1.54 |
| Detector | Type: MARRESEARCH / Detector: IMAGE PLATE / Date: Mar 16, 1998 |
| Radiation | Protocol: SINGLE WAVELENGTH / Monochromatic (M) / Laue (L): M / Scattering type: x-ray |
| Radiation wavelength | Wavelength: 1.54 Å / Relative weight: 1 |
| Reflection | Resolution: 1.8→26.26 Å / Num. obs: 17686 / % possible obs: 92.63 % / Observed criterion σ(I): 0 / Redundancy: 3.84 % / Rmerge(I) obs: 0.0677 / Net I/σ(I): 14.61 |
| Reflection shell | Resolution: 1.8→1.9 Å / Redundancy: 3.3 % / Rmerge(I) obs: 0.4495 / Mean I/σ(I) obs: 1.35 / % possible all: 88.4 |
| Reflection | *PLUS Num. measured all: 67986 |
| Reflection shell | *PLUS Num. unique obs: 4514 / Rmerge(I) obs: 0.361 |
- Processing
Processing
| Software |
| ||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Refinement | Method to determine structure:  MOLECULAR REPLACEMENT MOLECULAR REPLACEMENTStarting model: 1QQU Resolution: 1.8→8 Å / Cross valid method: FREE R / σ(F): 0
| ||||||||||||||||||||||||||||||||||||
| Refinement step | Cycle: LAST / Resolution: 1.8→8 Å
| ||||||||||||||||||||||||||||||||||||
| Software | *PLUS Name: X-PLOR,REFMAC / Classification: refinement | ||||||||||||||||||||||||||||||||||||
| Refinement | *PLUS Highest resolution: 1.8 Å / % reflection Rfree: 4.85 % / Rfactor all: 0.162 / Rfactor obs: 0.157 / Rfactor Rfree: 0.238 | ||||||||||||||||||||||||||||||||||||
| Solvent computation | *PLUS | ||||||||||||||||||||||||||||||||||||
| Displacement parameters | *PLUS | ||||||||||||||||||||||||||||||||||||
| Refine LS restraints | *PLUS
|
 Movie
Movie Controller
Controller























 PDBj
PDBj






