[English] 日本語
 Yorodumi
Yorodumi- PDB-1ep2: CRYSTAL STRUCTURE OF LACTOCOCCUS LACTIS DIHYDROOROTATE DEHYDROGEN... -
+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 1ep2 | ||||||
|---|---|---|---|---|---|---|---|
| Title | CRYSTAL STRUCTURE OF LACTOCOCCUS LACTIS DIHYDROOROTATE DEHYDROGENASE B COMPLEXED WITH OROTATE | ||||||
 Components Components | (DIHYDROOROTATE DEHYDROGENASE B ...) x 2 | ||||||
 Keywords Keywords | OXIDOREDUCTASE / Heterotetramer | ||||||
| Function / homology |  Function and homology information Function and homology informationdihydroorotate dehydrogenase (NAD+) / dihydroorotate dehydrogenase (NAD+) activity / 'de novo' UMP biosynthetic process / 'de novo' pyrimidine nucleobase biosynthetic process / 2 iron, 2 sulfur cluster binding / flavin adenine dinucleotide binding / electron transfer activity / oxidoreductase activity / metal ion binding / cytoplasm Similarity search - Function | ||||||
| Biological species |  Lactococcus lactis (lactic acid bacteria) Lactococcus lactis (lactic acid bacteria) | ||||||
| Method |  X-RAY DIFFRACTION / Resolution: 2.4 Å X-RAY DIFFRACTION / Resolution: 2.4 Å | ||||||
 Authors Authors | Rowland, P. / Norager, S. / Jensen, K.F. / Larsen, S. | ||||||
 Citation Citation |  Journal: Structure Fold.Des. / Year: 2000 Journal: Structure Fold.Des. / Year: 2000Title: Structure of dihydroorotate dehydrogenase B: electron transfer between two flavin groups bridged by an iron-sulphur cluster. Authors: Rowland, P. / Norager, S. / Jensen, K.F. / Larsen, S. | ||||||
| History |
|
- Structure visualization
Structure visualization
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  1ep2.cif.gz 1ep2.cif.gz | 126.1 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb1ep2.ent.gz pdb1ep2.ent.gz | 96.3 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  1ep2.json.gz 1ep2.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/ep/1ep2 https://data.pdbj.org/pub/pdb/validation_reports/ep/1ep2 ftp://data.pdbj.org/pub/pdb/validation_reports/ep/1ep2 ftp://data.pdbj.org/pub/pdb/validation_reports/ep/1ep2 | HTTPS FTP |
|---|
-Related structure data
- Links
Links
- Assembly
Assembly
| Deposited unit | 
| |||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | 
| |||||||||||||||
| Unit cell |
| |||||||||||||||
| Components on special symmetry positions |
| |||||||||||||||
| Details | The biological assembly is a heterotetramer. There is a crystallographic two-fold axis through the center of the heterotetramer, passing through a small hydrophobic cavity between the two chain A molecules of the heterotetramer at approximate coordinates (47.61, 34.32, 13.52). The heterotetramer is held together by the two chain A molecules, with the two chain B molecules making few close contacts with each other. |
- Components
Components
-DIHYDROOROTATE DEHYDROGENASE B ... , 2 types, 2 molecules AB
| #1: Protein | Mass: 32977.289 Da / Num. of mol.: 1 Source method: isolated from a genetically manipulated source Source: (gene. exp.)  Lactococcus lactis (lactic acid bacteria) Lactococcus lactis (lactic acid bacteria)Production host:  |
|---|---|
| #2: Protein | Mass: 28559.100 Da / Num. of mol.: 1 Source method: isolated from a genetically manipulated source Source: (gene. exp.)  Lactococcus lactis (lactic acid bacteria) Lactococcus lactis (lactic acid bacteria)Production host:  |
-Non-polymers , 5 types, 148 molecules 
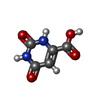







| #3: Chemical | ChemComp-FMN / |
|---|---|
| #4: Chemical | ChemComp-ORO / |
| #5: Chemical | ChemComp-FAD / |
| #6: Chemical | ChemComp-FES / |
| #7: Water | ChemComp-HOH / |
-Experimental details
-Experiment
| Experiment | Method:  X-RAY DIFFRACTION / Number of used crystals: 1 X-RAY DIFFRACTION / Number of used crystals: 1 |
|---|
- Sample preparation
Sample preparation
| Crystal | Density Matthews: 2.59 Å3/Da / Density % sol: 52.5 % | ||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Crystal grow | Temperature: 298 K / Method: vapor diffusion, hanging drop / pH: 4.6 Details: 2.4 M ammonium sulfate, 0.1 M sodium acetate, pH 4.6, VAPOR DIFFUSION, HANGING DROP, temperature 298K | ||||||||||||||||||||||||||||||
| Crystal grow | *PLUS pH: 6 Details: Rowland, P., (1997) Acta Crystallogr., Sect.D, 53, 802. | ||||||||||||||||||||||||||||||
| Components of the solutions | *PLUS
|
-Data collection
| Diffraction | Mean temperature: 288 K |
|---|---|
| Diffraction source | Source:  ROTATING ANODE / Type: RIGAKU RU200 / Wavelength: 1.54 ROTATING ANODE / Type: RIGAKU RU200 / Wavelength: 1.54 |
| Detector | Type: RIGAKU RAXIS II / Detector: IMAGE PLATE / Date: Jan 31, 1997 |
| Radiation | Protocol: SINGLE WAVELENGTH / Monochromatic (M) / Laue (L): M / Scattering type: x-ray |
| Radiation wavelength | Wavelength: 1.54 Å / Relative weight: 1 |
| Reflection | Resolution: 2.4→38.5 Å / Num. all: 89776 / Num. obs: 21335 / % possible obs: 86 % / Observed criterion σ(I): -3 / Redundancy: 4.2 % / Biso Wilson estimate: 34 Å2 / Rmerge(I) obs: 0.099 / Net I/σ(I): 12.4 |
| Reflection shell | Resolution: 2.4→2.44 Å / Redundancy: 4 % / Rmerge(I) obs: 0.497 / Num. unique all: 1107 / % possible all: 90.1 |
| Reflection | *PLUS Num. measured all: 89776 |
- Processing
Processing
| Software |
| |||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Refinement | Resolution: 2.4→38.5 Å / Cross valid method: THROUGHOUT / Stereochemistry target values: Engh & Huber
| |||||||||||||||||||||||||
| Refinement step | Cycle: LAST / Resolution: 2.4→38.5 Å
| |||||||||||||||||||||||||
| Refine LS restraints |
| |||||||||||||||||||||||||
| Software | *PLUS Name:  X-PLOR / Version: 3.1 / Classification: refinement X-PLOR / Version: 3.1 / Classification: refinement | |||||||||||||||||||||||||
| Refinement | *PLUS Lowest resolution: 38.5 Å / % reflection Rfree: 5 % / Rfactor obs: 0.19 / Rfactor Rwork: 0.19 | |||||||||||||||||||||||||
| Solvent computation | *PLUS | |||||||||||||||||||||||||
| Displacement parameters | *PLUS |
 Movie
Movie Controller
Controller













 PDBj
PDBj













